This study aims to describe real world palivizumab use and effectiveness in high-risk Latin American infants and young children.
MethodProspective, multicenter observational study with infants at risk for severe RSV infection who received palivizumab according to routine clinical practice. Subjects were followed for one year with monthly visits after the first dose of palivizumab. An infant was considered adherent if receiving all the expected injections or five or fewer injections within appropriate inter-dose intervals. Annual incidence rates and risk factors of lower respiratory tract infection (LRTI) hospitalization were determined through Poisson regression models (α=0.05).
ResultsThe study enrolled 458 children from seven countries in Latin America, from February 2011 to September 2012. The majority (98%) were born <36 weeks gestation. Overall, patients received 83.7% of their expected injections and 86.7% completed one year of follow-up. Of the 61 LRTI hospitalizations, 12 episodes were due to RSV infection. The RSV-associated hospitalization rate was 2.9 per 100 patient-years. Bronchopulmonary dysplasia was identified as an independent risk factor for LRTI hospitalization. A total of 1165 adverse events were recorded during one year of follow-up. One hundred and two patients (22.3%) had a total of 135 serious adverse events, but no events were considered to be related to palivizumab.
ConclusionsThe rate of RSV hospitalization in high-risk infants in Latin America was low and aligned with those observed in randomized control trials and observational studies. Palivizumab prophylaxis appeared effective and had a good safety profile in this population.
Este estudo visa descrever o uso e a eficácia do palivizumabe no mundo real em neonatos e jovens crianças de alto risco latino-americanas.
MétodoEstudo observacional prospectivo multicêntrico com neonatos em risco devido a infecção grave por VSR que receberam palivizumabe de acordo com a prática clínica de rotina. Os indivíduos foram acompanhados por um ano, com visitas mensais após a primeira dose de palivizumabe. Um neonato foi considerado adepto se recebeu todas as injeções esperadas ou ≤5 injeções nos intervalos entre doses adequados. As taxas de incidência anuais e os fatores de risco de internação por infecção do trato respiratório inferior (ITRI) foram determinados por meio dos modelos de regressão de Poisson (α=0,05).
ResultadosO estudo inscreveu 458 crianças de sete países da América Latina, de fevereiro de 2011 a setembro de 2012. A maioria (98%) nasceu com <36 semanas. Em geral, os pacientes receberam 83,7% de suas injeções esperadas, e 86,7% completaram um ano de acompanhamento. Das 61 internações por ITRI, 12 episódios foram devidos a infecção por VSR. A taxa de internação associada ao VSR foi de 2,9 em cada 100 pacientes-anos. A displasia broncopulmonar foi identificada como um fator de risco independente da internação por ITRI. Foram registrados 1165 eventos adversos no total durante um ano de acompanhamento. 122 pacientes (22,3%) apresentaram um total de 135 eventos adversos graves, porém nenhum deles foi considerado relacionado ao palivizumabe.
ConclusõesA taxa de internação por VSR em neonatos de alto risco na América Latina foi baixa e em linha com as observadas em ensaios clínicos controlados randomizados e estudos observacionais. A profilaxia com palivizumabe pareceu eficaz e com bom perfil de segurança nessa população.
Lower respiratory tract infections (LRTIs) are a leading cause of acute illnesses and mortality in infants and children <5 years of age, accounting for approximately 1.4 million deaths in 2010 worldwide.1 Respiratory syncytial virus (RSV) is the most frequent etiology of severe respiratory illness, such as bronchiolitis and/or pneumonia.2,3 One meta-analysis estimated that at least 33.8 million episodes of LRTI and 66,000–199,000 deaths in children <5 years of age were caused by RSV infection in 2005.2 Nearly 99% of RSV deaths occurred in developing countries, where the incidence of RSV-associated LRTI seems to be more than twice that observed in industrialized countries.2
In Latin America, a recent meta-analysis estimated a 41.5% (95% CI, 32.0–41.4) prevalence of RSV in infants aged 0–11 months with LRTI, and that RSV was responsible for 36.5% (95% CI, 28.5–44.9) of hospital admissions due to LRTI among infants aged 0–11 months.4 Moreover, a prospective epidemiological cohort of patients aged <18 years hospitalized due to LRTIs from 2000 to 2013 in Buenos Aires registered a RSV associated fatality rate of 1.9% (74 deaths/3888 cases).5 Although these estimates may imply an urgency for health-related interventions, more studies are needed to quantify the disease burden due to RSV and risk factors associated to LRTI hospitalizations in Latin American countries.2,4
Several host- and social-related factors have been associated with an increased risk of RSV infection, namely age <6 months at the beginning of the RSV season, male sex, multiple birth, household crowding, low socioeconomic status, and parental education, daycare attendance, exposure to indoor tobacco smoke, and abbreviated breastfeeding.6,7 In addition, some clinical conditions are associated with severe RSV disease and hospitalization, including prematurity (infants born at ≤35 weeks of gestational age), bronchopulmonary dysplasia (BPD), and hemodynamically significant congenital heart disease (HSCHD).3,8–10 However, the relative importance of these risk factors may be different in Latin America.
Palivizumab (Synagis®; AbbVie Inc, IL, USA) is a humanized monoclonal antibody approved for the prevention of severe RSV disease in premature infants and in young children with BPD or HSCHD.11 Its safety and efficacy have been assessed in randomized controlled studies, open-label non-comparative trials, and several observational studies.12 Lower rates of RSV hospitalizations and clinical complications have been reported in children who received palivizumab prophylaxis.8,12,13 However, there is a paucity of data about the use and effectiveness of palivizumab in developing countries where rates of RSV infection are higher, specifically in Latin American countries.12,14–17
This study aims to describe palivizumab use in high risk-infants, and to estimate the frequency of RSV hospitalization among infants and young children treated with palivizumab in Latin American countries, during a one-year period of follow-up after the first dose.
MethodsStudy designThis was a prospective, observational, multicenter study in a cohort of infants at risk for severe RSV infection who received palivizumab during local RSV circulation and within the terms of marketing authorization with regard to dose, population, and indication. The study was conducted at 24 sites across seven Latin American countries (Argentina, Chile, Colombia, Ecuador, Mexico, Peru, and Uruguay). Subject recruitment began on February 19, 2011, and concluded on September 6, 2012; each subject was followed for 12 months, with monthly visits during the period of palivizumab administration and, afterwards, with monthly calls.
Eligibility criteriaAt each study site, premature infants (born at ≤35 weeks of gestational age) or young children with a history of BPD or HSCHD who received the first dose of palivizumab within the two weeks prior to the inclusion criteria assessment were eligible to participate in the study. Children excluded from receiving palivizumab as per local practice did not enter into the study. The parents or legal guardians of the infants and young children included in the study provided written informed consent for their child's participation.
Follow-up and data collectionEnrolled subjects were followed for one year after receiving their first dose of palivizumab. Epidemiological and clinical data, information about patient adherence, hospitalizations, and safety data were collected on a monthly basis and according to routine clinical practice, through in-office evaluations during the first four months. After the first four months, there were six and 12-month follow-up visits and follow-up phone calls at months five, seven, eight, nine, 10, and 11 after the subject was included in the study.
Each patient's medical history and exposure to environmental risk factors were collected and a physical examination was performed at each in-office study visit. Adverse events and hospitalizations that occurred since the previous visit were also recorded. LRTI was defined as pneumonia, bronchiolitis, or wheezing, according to clinical diagnosis. Nasal and pharyngeal swabs were collected for RSV testing only when recommended by local clinical guidelines and/or according to physician/medical decision.
Children could receive up to five doses of palivizumab, according to the physician's prescription. Patient adherence to treatment was calculated as the received number of palivizumab doses vs. the expected number of doses. The expected number of doses for each patient was estimated based on seasonality and the current guidelines for each country. An analysis of inter-dose interval (for cases in which the date for every dose was available) was performed, and intervals of 30 (±5) days were considered to be compliant.
Safety analysisAdverse events were classified based on the Medical Dictionary for Regulatory Activities classification system, version 16.0. Serious adverse events were defined as any adverse event that resulted in death, a life-threatening situation, inpatient hospitalization, persistent or significant disability or incapacity, or other clinically relevant events.
Statistical analysisA minimum sample size of 400 patients was estimated based on the potential number of patients receiving palivizumab in each country, and to allow an estimate of the proportion of LRTIs with a 95% confidence interval (CI) and within an estimated precision of 3%. All data analysis was conducted with Stata (StataCorp. 2007. Stata Statistical Software: version 10. TX, USA). Frequencies and percentages of categorical variables, as well as measures of central position and dispersion for quantitative variables, were used to describe the characteristics of sample subjects, the use of palivizumab, and most common adverse events. The 95% CIs were estimated for the proportions or incidence rates of relevant outcomes. Generalized linear models were used to estimate annual LRTI hospitalization rates. Poisson regression models were applied to identify risk factors for LRTI hospitalizations, and baseline characteristics such as age, gender, birth weight, and comorbidities were included as covariates.
This study was conducted in accordance with the Declaration of Helsinki. The study protocol was approved by independent institutional ethical committees before study initiation, and according to national legislation. Written informed consent from at least one parent or legal guardian was obtained for all patients.
ResultsSample characteristicsA total of 464 infants and young children were enrolled at 24 sites in seven countries in Latin America, from which six were excluded due to having no additional follow-up visits (n=4), invalid informed consent (n=1), or having received palivizumab in the previous season (n=1). Hence, 458 infants and young children were included in the study, and 397 (86.7%) completed one year of follow-up. Among the 61 infants and young children that did not complete follow-up, 43 (70.5%) were lost to follow-up, six (9.8%) had study consent withdrawn, three (4.9%) died from non-respiratory disease-associated reasons, and nine (29.4%) did not complete the study for other reasons.
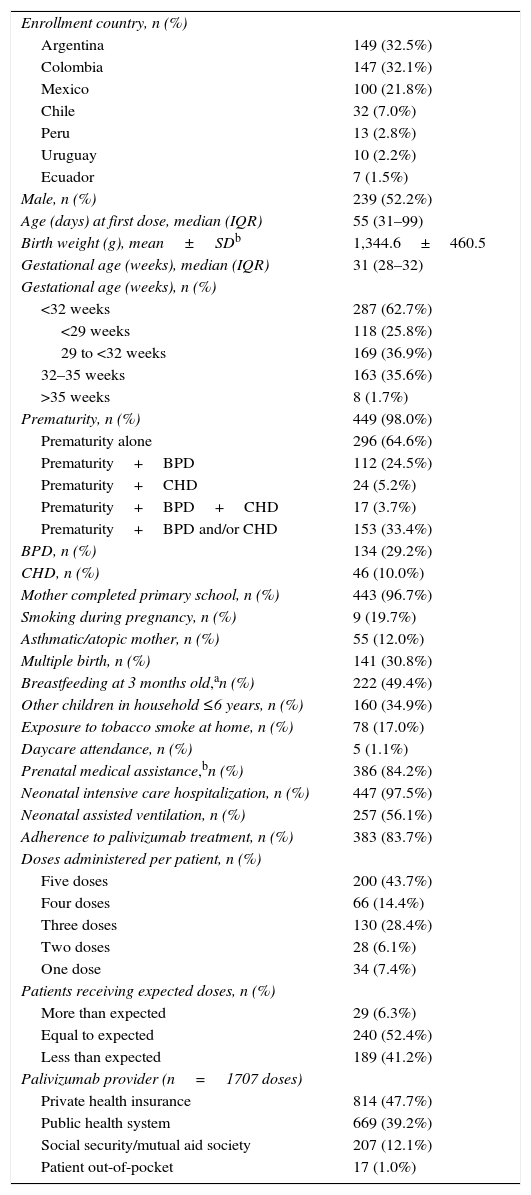
Patient demographics of the included infants and young children are shown in Table 1. Fifty-two percent of the patients were male. The mean birth weight was 1345g (standard deviation [SD], 460), and the median gestational age was 31 weeks (range, 23–39 weeks). Nearly all of the patients in the study (98.2%) were premature; 29.2% of patients had BPD and 10.1% had HSCHD. Furthermore, prematurity with BPD and/or HSCHD were seen in 33.4% of patients.
Demographic, clinical, and treatment characteristics of the study population (n=458).
| Enrollment country, n (%) | |
| Argentina | 149 (32.5%) |
| Colombia | 147 (32.1%) |
| Mexico | 100 (21.8%) |
| Chile | 32 (7.0%) |
| Peru | 13 (2.8%) |
| Uruguay | 10 (2.2%) |
| Ecuador | 7 (1.5%) |
| Male, n (%) | 239 (52.2%) |
| Age (days) at first dose, median (IQR) | 55 (31–99) |
| Birth weight (g), mean±SDb | 1,344.6±460.5 |
| Gestational age (weeks), median (IQR) | 31 (28–32) |
| Gestational age (weeks), n (%) | |
| <32 weeks | 287 (62.7%) |
| <29 weeks | 118 (25.8%) |
| 29 to <32 weeks | 169 (36.9%) |
| 32–35 weeks | 163 (35.6%) |
| >35 weeks | 8 (1.7%) |
| Prematurity, n (%) | 449 (98.0%) |
| Prematurity alone | 296 (64.6%) |
| Prematurity+BPD | 112 (24.5%) |
| Prematurity+CHD | 24 (5.2%) |
| Prematurity+BPD+CHD | 17 (3.7%) |
| Prematurity+BPD and/or CHD | 153 (33.4%) |
| BPD, n (%) | 134 (29.2%) |
| CHD, n (%) | 46 (10.0%) |
| Mother completed primary school, n (%) | 443 (96.7%) |
| Smoking during pregnancy, n (%) | 9 (19.7%) |
| Asthmatic/atopic mother, n (%) | 55 (12.0%) |
| Multiple birth, n (%) | 141 (30.8%) |
| Breastfeeding at 3 months old,an (%) | 222 (49.4%) |
| Other children in household ≤6 years, n (%) | 160 (34.9%) |
| Exposure to tobacco smoke at home, n (%) | 78 (17.0%) |
| Daycare attendance, n (%) | 5 (1.1%) |
| Prenatal medical assistance,bn (%) | 386 (84.2%) |
| Neonatal intensive care hospitalization, n (%) | 447 (97.5%) |
| Neonatal assisted ventilation, n (%) | 257 (56.1%) |
| Adherence to palivizumab treatment, n (%) | 383 (83.7%) |
| Doses administered per patient, n (%) | |
| Five doses | 200 (43.7%) |
| Four doses | 66 (14.4%) |
| Three doses | 130 (28.4%) |
| Two doses | 28 (6.1%) |
| One dose | 34 (7.4%) |
| Patients receiving expected doses, n (%) | |
| More than expected | 29 (6.3%) |
| Equal to expected | 240 (52.4%) |
| Less than expected | 189 (41.2%) |
| Palivizumab provider (n=1707 doses) | |
| Private health insurance | 814 (47.7%) |
| Public health system | 669 (39.2%) |
| Social security/mutual aid society | 207 (12.1%) |
| Patient out-of-pocket | 17 (1.0%) |
BPD, bronchopulmonary dysplasia; CHD, congenital heart disease; IQR, interquartile range; SD, standard deviation.
The most frequent reason for indication of palivizumab was prematurity (by itself or in conjunction with other indications) (98.0%). The total number of doses administered was 1744 in 458 patients, corresponding to 3.8±1.3 doses per patient on average. Overall, patients received 83.7% (95% CI, 80.8–86.5) of their expected doses, based on seasonality and current guidelines for each country, and 78.4% of the 1249 inter-dose intervals were classified as adherent (Table 1). The mean time between starting palivizumab prophylaxis and developing an LRTI due to RSV was 75.7 days, with a median of 44 days.
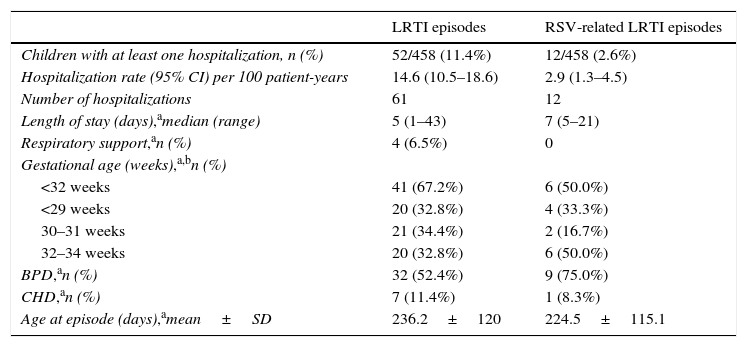
Hospitalizations due to LRTISixty-one episodes of LRTI that required hospitalization occurred in 52 children (Table 2). The hospitalization rate due to LRTI was 14.6 per 100 patient-years (95% CI, 10.5–18.6). RSV infection was confirmed in 12 episodes in 12 infants, and the RSV-associated hospitalization rate was 2.9 per 100 patient-years (95% CI, 1.3–4.5). None of the RSV hospitalizations required respiratory support. From the 12 cases of RSV-associated hospitalizations, all were preterm babies and ten had another risk factor: nine were BPD patients and one was HSCHD.
Characterization of hospitalization episodes due to LRTI.
| LRTI episodes | RSV-related LRTI episodes | |
|---|---|---|
| Children with at least one hospitalization, n (%) | 52/458 (11.4%) | 12/458 (2.6%) |
| Hospitalization rate (95% CI) per 100 patient-years | 14.6 (10.5–18.6) | 2.9 (1.3–4.5) |
| Number of hospitalizations | 61 | 12 |
| Length of stay (days),amedian (range) | 5 (1–43) | 7 (5–21) |
| Respiratory support,an (%) | 4 (6.5%) | 0 |
| Gestational age (weeks),a,bn (%) | ||
| <32 weeks | 41 (67.2%) | 6 (50.0%) |
| <29 weeks | 20 (32.8%) | 4 (33.3%) |
| 30–31 weeks | 21 (34.4%) | 2 (16.7%) |
| 32–34 weeks | 20 (32.8%) | 6 (50.0%) |
| BPD,an (%) | 32 (52.4%) | 9 (75.0%) |
| CHD,an (%) | 7 (11.4%) | 1 (8.3%) |
| Age at episode (days),amean±SD | 236.2±120 | 224.5±115.1 |
BPD, bronchopulmonary dysplasia; CHD, congenital heart disease; CI, confidence interval; LRTI, lower respiratory tract infection; RSV, respiratory syncytial virus; SD, standard deviation.
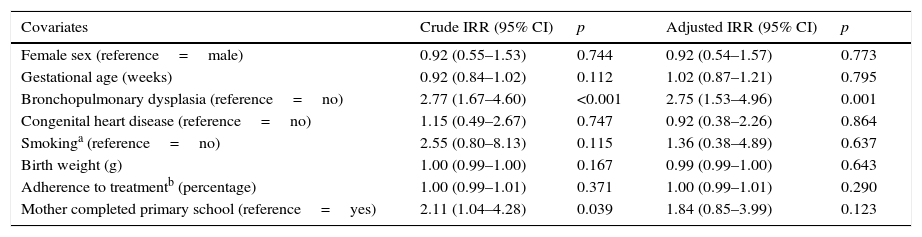
Univariate and multivariate analyses were performed for identification of risk factors for LRTI hospitalization (Table 3). In the univariate analysis, a lower level of maternal education and BPD were shown to increase the risk of LRTI hospitalization in a population receiving palivizumab prophylaxis. In the multivariate model analysis, only BPD was confirmed as an independent risk factor (adjusted IRR, 2.77).
Risk factors for LRTI hospitalizations.
| Covariates | Crude IRR (95% CI) | p | Adjusted IRR (95% CI) | p |
|---|---|---|---|---|
| Female sex (reference=male) | 0.92 (0.55–1.53) | 0.744 | 0.92 (0.54–1.57) | 0.773 |
| Gestational age (weeks) | 0.92 (0.84–1.02) | 0.112 | 1.02 (0.87–1.21) | 0.795 |
| Bronchopulmonary dysplasia (reference=no) | 2.77 (1.67–4.60) | <0.001 | 2.75 (1.53–4.96) | 0.001 |
| Congenital heart disease (reference=no) | 1.15 (0.49–2.67) | 0.747 | 0.92 (0.38–2.26) | 0.864 |
| Smokinga (reference=no) | 2.55 (0.80–8.13) | 0.115 | 1.36 (0.38–4.89) | 0.637 |
| Birth weight (g) | 1.00 (0.99–1.00) | 0.167 | 0.99 (0.99–1.00) | 0.643 |
| Adherence to treatmentb (percentage) | 1.00 (0.99–1.01) | 0.371 | 1.00 (0.99–1.01) | 0.290 |
| Mother completed primary school (reference=yes) | 2.11 (1.04–4.28) | 0.039 | 1.84 (0.85–3.99) | 0.123 |
CI, confidence interval; IRR, incidence rate ratio; LRTI, lower respiratory tract infection.
Bold text is considered significant; p<0.05.
Additional analyzes were performed in the subgroup of 134 patients with BPD; 28 patients with BPD had a total of 32 hospitalization episodes related to LRTI, and nine episodes were confirmed as associated to RSV infection. The LRTI hospitalization rate was 26.4 per 100 patient-years (95% CI, 17.1–35.8), and the RSV hospitalization rate was 7.4 per 100 patient-years (95% CI, 2.7–12.1).
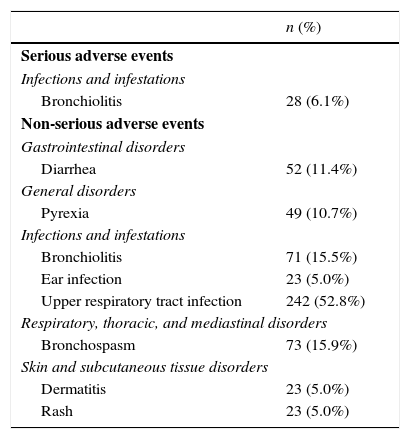
Adverse events during treatment with palivizumabA total of 1165 adverse events were recorded during one year of follow-up. One hundred and two patients (22.3%) had a total of 135 serious adverse events. Table 4 presents the non-serious adverse events (≥5% of patients) reported in the study (n=299). Of the serious adverse events reported, the most common event (≥5% of patients) was bronchiolitis (n=28; 6.1%). Furthermore, 92 serious adverse events (68.1%) resulted in or prolonged a hospital stay, 29 (21.4%) were classified by the investigator as clinically relevant, and 11 (8.1%) were life-threatening. Three deaths (2.2%) were recorded during follow-up and were not considered to be related to treatment with palivizumab or RSV infection. A total of six events of injection-site pain following palivizumab administration were reported in three patients. There were no serious adverse events that were considered to be related to palivizumab.
Most common serious and non-serious adverse events (≥5% of subjects).a
| n (%) | |
|---|---|
| Serious adverse events | |
| Infections and infestations | |
| Bronchiolitis | 28 (6.1%) |
| Non-serious adverse events | |
| Gastrointestinal disorders | |
| Diarrhea | 52 (11.4%) |
| General disorders | |
| Pyrexia | 49 (10.7%) |
| Infections and infestations | |
| Bronchiolitis | 71 (15.5%) |
| Ear infection | 23 (5.0%) |
| Upper respiratory tract infection | 242 (52.8%) |
| Respiratory, thoracic, and mediastinal disorders | |
| Bronchospasm | 73 (15.9%) |
| Skin and subcutaneous tissue disorders | |
| Dermatitis | 23 (5.0%) |
| Rash | 23 (5.0%) |
MedDRA, Medical Dictionary for Regulatory Activities.
Palivizumab is approved for the prophylaxis of severe RSV disease, with proven efficacy in reducing hospitalizations in high-risk infants.8,13 However, consistent prophylaxis may be challenging due to a variety of medical and socio-economic factors of the child.18,19 This study provided a prospective description of the use and effectiveness of palivizumab in Latin American countries.
The rate of RSV-associated hospitalizations observed in this study (2.9 per 100 patient-years) is low and within the range reported for the palivizumab arm in different clinical trials (0.0–5.3%).8,13,20 Furthermore, the present study's results are also consistent with those from other observational studies (1.3–2.9 episodes per 100 patient-years).12,21,22 However, there are few data on RSV hospitalization rates in infants without palivizumab prophylaxis in Latin America. One study from a public hospital in Argentina showed that infants without palivizumab prophylaxis had RSV-LRTI hospitalization rates of 26% and 29% in two historical cohorts.23
It has been shown that the RSV hospitalization rates in patients with palivizumab prophylaxis could be as high as 7.6% in certain circumstances, especially in patients with BPD.13,18,23 In fact, in the present study, a RSV hospitalization rate of 7.4 per 100 patient-years was observed in children with BPD, a finding that is aligned with previously reported results.18 The median time of hospitalization due to illness caused by RSV was seven days, similar to the results noted in studies from other countries22,24 and to what was found in the study by Bauer et al. at a single center in Argentina.23
The incidence and severity of RSV infection in preterm infants has been associated with several factors, such as presence of siblings at home and attendance at daycare, among others.6,25 A prospective cohort study with 152 infants hospitalized for LRTI showed that low birth weight, low educational level of the mother, and poor socioeconomic conditions were risk factors associated with RSV hospitalizations in term and preterm infants without hemodynamic instability, cardiac conditions, or chronic lung disease in Brazil.26 In Argentina, Bauer et al. also observed that children in the household younger than 10 years of age and mothers with incomplete primary school education were associated with an increased risk of RSV hospitalization among preterm infants with and without BPD.27 Moreover, some studies have shown an increased risk of LRTI due to tobacco smoke exposure at home, although further research is necessary to determine its impact on RSV LRTI severity.6 In the present study, only BPD was identified as an independent risk factor for hospitalization due to LRTI, while other factors were not associated with an increased risk.
With regard to safety, no serious adverse events related to palivizumab were reported in the present study. The pattern and type of adverse events were consistent with the known safety profile of palivizumab, which generally reflects the underlying medical conditions of these patients.8,12,18,28 Similar to other studies, upper respiratory infections were the most frequent adverse events, with a proportion comparable to that reported in a Brazilian study (59.4% vs. 52.8% in the present study).29
This study had some limitations, as is the case with observational studies. As the present study did not have a control arm, it does not provide as clear an estimate of the impact of palivizumab prophylaxis as do results obtained through randomized comparative clinical trials.24 In addition, not all hospitalizations had a diagnostic test performed to confirm if the episode was caused by RSV infection. However, because testing for RSV depends on local guidelines and was not always performed, these results should be interpreted with caution. This is mainly due to the variability among the participating sites regarding local guidelines on diagnostic tests, a limitation that was also present in similar real-world studies.21,24,30 Missing information may also be a factor in a few cases where the hospitalization occurred at a hospital that was not involved in the study. Nevertheless, efforts were made to collect data regarding the presence of RSV in these cases. Finally, another limitation was that non-serious adverse events were not classified by the investigators as related or unrelated to palivizumab.
Although the participating sites may not be fully representative of the overall population of patients receiving prophylaxis in Latin American countries, they were selected based on their enrollment capacity, as well as experience with palivizumab and treatment of high-risk children. Conversely, as the children's parents needed to have access to the healthcare system, a selection bias may affect some variables, such as adherence to treatment and educational level, that otherwise could be associated with LRTI hospitalizations. Finally, although the incidence and severity of RSV infection is affected by seasonality,25 this is not expected to compromise the main findings. The present study was carried out during two consecutive years (including more than one RSV season) in different cities and countries, thus providing an overview of the Latin America region while covering most of its RSV seasonality patterns. In fact, to the authors’ knowledge, this is the first multicenter study that included infants receiving prophylaxis from the Latin American region, with the advantage of collecting complete demographic and clinical data for nearly all enrolled children, even though other single-center studies and with shorter follow-up have been conducted in this region.
In conclusion, the hospitalization rate due to RSV infection was low in this population and aligned with those observed in several randomized control trials and observational studies. Based on this study, palivizumab prophylaxis is effective and has a good safety profile in Latin American infants at high risk of severe RSV infection.
FundingThis study was funded by AbbVie, Inc.
Conflicts of interestN Vain has received grant/research support, speaker, and consulting fees from AbbVie. M E Serra is employee of FUNDASAMIN, who was contracted by AbbVie for study data management. AbbVie Inc. was involved in the study design; collection, analysis, and interpretation of data, and the preparation and approval of this manuscript. L. Castillo and G. Bugarin are employees of AbbVie and may hold stock or options.
The authors want to express their gratitude to Dr. Guillermo de Jesus Ruelas Orozco, Dr. Margarita Morales Marquez, Dr. Carolina Salazar, Dr. Adriana Castro, and Mrs. Mariana Jacqueline Martínez Muñoz for their collaboration in this study, and to Mrs. Milene Fernandes (Eurotrials, Scientific Consultants), who assisted in writing this paper.
Please cite this article as: Castillo LM, Bugarin G, Arias JC, Rangel JI, Serra ME, Vain N. One-year observational study of palivizumab prophylaxis on infants at risk for respiratory syncytial virus infection in Latin America. J Pediatr (Rio J). 2017;93:467–74.