This study aimed to evaluate annual trends of early neonatal sepsis and antimicrobial use in very low birth weight infants for 12 years, as well as to identify microbiological agents, antimicrobial sensitivity profiles, and association with early neonatal death.
MethodThis was a retrospective cohort study including 1254 very low birth weight infants admitted from 2006 to 2017. Four groups were evaluated: culture-confirmed sepsis; presumed neonatal sepsis; ruled out neonatal sepsis group; and infants not exposed to antibiotics.
ResultsThe medians of gestational age and birth weight were 29 weeks (27–31) and 1090g (850–1310), respectively. The rates of culture-confirmed sepsis, presumed neonatal sepsis, ruled out neonatal sepsis, and not exposed to antibiotics were 1.3, 9.0, 15.4, and 74.3%, respectively. From the initial group of newborns whose antimicrobial treatment was administered for sepsis’ suspicion, it was possible to discontinue antibiotic in 44%. The culture-confirmed sepsis rates remained stable (p=0.906). Significant tendencies of decreasing presumed sepsis rates (p<0.001) and increased ruled out neonatal sepsis/not exposed to antibiotics rates (p<0.001) were observed. Streptococcus agalactiae and enteric Gram-negative rods were the predominant agents and most of them were sensitive to crystalline penicillin/ampicillin (88.2%) and to ampicillin and/or amikacin. Early death occurred in 10.8%, specifically in the culture-confirmed sepsis and presumed neonatal sepsis groups.
ConclusionThe confirmed sepsis rate was low and remained stable. There was a significant downward trend in the presumed neonatal sepsis rate and a significant upward trend in the ruled out neonatal sepsis group. The rate of not exposed to antibiotics infants was high, also presenting a significant downward trend. The identified bacteria were those commonly found and showed usual antimicrobial susceptibility patterns. Death predominantly occurred in groups that received antibiotic treatment.
Neonatal mortality represents about 70% of the infant mortality rate in Brazil, and sepsis is the national infant leading cause of death, especially in preterm and very low birth weight (VLBW) infants.1 Recently, American data from the National Institute of Health showed a rate of early-onset neonatal sepsis (EONS) of 13.9/1000 VLBW live birth.2 The associated clinical signs are nonspecific, which hinders the diagnosis and often leads to the overuse administration of antibiotics.3 Antimicrobial agents are the drugs most prescribed in neonatal intensive care units. Their inappropriate or excessive use is associated with serious adverse effects related to an alteration of the normal microbiota, such as the selection of multi-resistant microorganisms4 and increased risk of invasive candidiasis, necrotizing enterocolitis, late-onset sepsis, and death.5 Additionally, other foreseeable consequences of exposure to this class of medication include acute toxicity, need for intravenous access, and increased costs for hospitals.6
It has been increasingly reported in the literature that, in unconfirmed cases of neonatal sepsis, antibiotics can safely be discontinued in clinically stable patients with negative blood cultures after 36–48h of incubation.4,7 Furthermore, it is described that the antimicrobial control program, based on the reevaluation of the medication need on the third day of use, contributes to the prevention of emergencies or the induction of multi-resistant bacteria,8 which continues to be an underestimated but increasingly reported threat in neonatal units worldwide.4
This neonatal unit has been performing an institutional antimicrobial use control program for 25 years, with well-established clinical protocols for managing infants with a risk of infection, focusing on the rational use of antibiotics. This approach includes a correct diagnosis of neonatal sepsis, based on prompt infectious screening and treatment of infants who show clinical signs compatible with sepsis with or without evident risk factors. Treatment is always preceded by sampling of blood cultures, followed by rigorous clinical and laboratory reevaluation within 48h of treatment. Then, two options are applicable: maintaining antibiotics in the proven or presumed cases of sepsis or discontinuing antimicrobials when the infection is ruled out or unlikely even in preterm and extremely low birth weight infants.
This management by risk assessment is unusual in Brazil, notably for VLBW infant sepsis evaluation,9,10 and the authors hypothesize that this evaluation tends to have a significant impact on reducing the use of antibiotics in very low birth weight infants. Thus, this study aimed to evaluate the rates of culture-confirmed sepsis (CCS) and presumed EONS, as well as rates of ruled out neonatal sepsis (ROS) cases and rate of newborns not exposed to antibiotics in the first 48h of life (NEATB), in the period from 2006 to 2017. As secondary outcomes, the microbiological agents associated with EONS and their antimicrobial sensitivity profiles were evaluated, as well as the association of sepsis and early neonatal death in this population.
Material and methodsThis was a retrospective single-center cohort study using a computer-based database analysis, part of the Brazilian Network on Neonatal Research registry (RBPN in Portuguese). It was conducted in the Womens’ Hospital at the University of Campinas – Unicamp, Brazil, in a tertiary neonatal referral unit, consisting of 15 intensive care and 15 intermediate care beds. A convenience sample was used and included all VLBW infants admitted to the neonatal intensive care unit from January 1, 2006, to December 31, 2017.
EONS was defined as the presence of clinical signs compatible with infection initiated within the first 48h of life. The disease was categorized as CCS if microorganisms grew in blood culture and/or culture of cerebrospinal fluid; and presumed early-onset neonatal sepsis (PNS), when there were clinical signs compatible with the disease associated with abnormal leucogram and/or increase of serum C-reactive protein (CRP), but negative cultures.
Two other groups were evaluated: those with ROS – newborns with the presence of clinical signs suggesting sepsis, with or without evident prenatal risk of infection, whose antimicrobial treatment was discontinued after 48–72h due to clinical evolution not compatible with sepsis, negative blood cultures, associated with a normal white blood cell count and two normal CRP serial serum dosages (<1.0mg/dL). Finally, a group of NEATB newborns with no apparent risk of EONS and who were not submitted to infection screening, such as those born to pregnant women without labor, with an intrapartum rupture of membranes, without fever or other signs of infection and with pregnancy completed by the cesarean section.
Clinical signs considered as sepsis manifestations were: respiratory distress, apnea, cyanosis, pallor, lethargy, irritability, abnormal movements, temperature instability, gastric residue, abdominal distention, vomiting, bradycardia, tachycardia, poor perfusion, reduced capillary refill, hypotension, and bleeding.
The empirical treatment adopted was ampicillin or penicillin G (after 2007) and amikacin. Adjustments were made individually according to the antimicrobial sensitivity profile and other clinical aspects (renal failure, for example).
For blood culture analysis, a minimum of 1mL blood was added into a specific vial (BD BACTEC®; Becton Dickinson – Sparks, MD, United States). Samples were proceeded and incubated immediately in the laboratory in an automated continuous monitoring blood culture system.
Early neonatal death was defined as that occurring between 0 and 6 days of life, and it was associated with sepsis when this cause was indicated as the direct cause of death.
Maternal and neonatal variables were examined for the description and comparison of the population. Small for gestational age (SGA) was defined as birth weight <10th percentile for gestational age.11
Categorical variables were expressed in absolute and relative frequencies and compared by chi-squared or Fisher's test. Continuous variables had non-parametric distribution. They were expressed as median and interquartile range (IQR), and evaluated by the Mann–Whitney test. The Kruskal–Wallis test was used to compare the three groups of infants whom antibiotics have been administrated, followed by the Dunn post-hoc test. The annual trend evaluation was performed by the Cochran–Armitage test. The accepted significance value was p=0.05.
This study was approved by the local Research Ethics Committee (ethical approval number 88964618.7.0000.5404).
ResultsIn the 12 years of the study, 1254 VLBW infants were admitted (94.6% inborn). The median gestational age was 29 weeks (IQR, 27–31) and the median birth weight was 1090g (IQR, 850–1310), respectively. General characteristics of the population are summarized in the first columns of Tables 1 and 2.
Distribution of maternal variables according to the group of very low birth weight infants – confirmed sepsis, presumed sepsis, and ruled-out sepsis.
| Total (1254) | CCS (17) | PNS (113) | ROS (192) | pa | |
|---|---|---|---|---|---|
| Age (years)b | 27 (22–33) | 24 (23–26) | 26 (21–32) | 26 (20–32) | 0.562 |
| White ethnicity | 693 (56.2) | 12 (70.6) | 59 (52.2) | 112 (58.3) | 0.291 |
| Arterial hypertension | 494 (39.5) | 3 (17.6) | 27 (24.1) | 41(27.4) | 0.779 |
| Diabetes mellitus | 104 (8.3) | 3 (17.6) | 3 (2.7) | 19 (9.9) | 0.023 |
| Clinical chorioamnionitis3 | 67 (5.4) | 7 (41.2) | 18 (16.4) | 20 (10.4) | 0.002 |
| Antenatal corticosteroid | 944 (75.3) | 11(64.7) | 74 (65.5) | 127 (66.1) | 0.988 |
| Peripartum hemorrhage | 88 (7.0) | 0 (0) | 2 (1.8) | 15 (7.8) | 0.045 |
| Single gestation | 959 (76.5) | 12 (70.6) | 81 (71.7) | 149 (77.6) | 0.464 |
| Rup. of membranes>18h | 173 (14.0) | 5 (29.4) | 30 (28.0) | 45 (25.9) | 0.898 |
| Vaginal delivery | 330 (26.3) | 11 (64.7) | 46 (40.7) | 91 (47.4) | 0.898 |
CCS, culture confirmed early-onset neonatal sepsis; PNS, presumed sepsis; ROS, ruled-out sepsis; rup., rupture.
Categorical variables expressed in absolute frequency (percentage).
Distribution of neonatal variables according to the group of very low birth weight newborns – confirmed sepsis, presumed sepsis, and ruled-out sepsis.
| Total (1254) | CCS (17) | PNS (113) | ROS (192) | p | |
|---|---|---|---|---|---|
| Male sex | 629 (50.2) | 7 (41.2) | 64 (56.6) | 113 (58.8) | 0.366 |
| SGA11 | 495 (39.5) | 3 (17.6) | 32 (28.3) | 44 (22.9) | 0.454 |
| Resuscitation | 693 (55.7) | 14 (82.3) | 86 (77.5) | 133 (70.7) | 0.313 |
| 1st min Apgar<7 | 576 (46.4) | 11 (68.7) | 72 (65.4) | 119 (63.6) | 0.891 |
| 5st min Apgar<7 | 169 (13.6) | 6 (37.5) | 22 (20.0) | 35 (18.6) | 0.194 |
| Birth weighta | 1090 (850–1310) | 950 (645–1350) | 935 (750–1115) | 945 (767–1222) | 0.590 |
| Gestational agea | 29 (27–31) | 28 (26–30) | 28 (26–29) | 27 (26–29) | 0.896 |
| SNAPPEIIb | 15 (5–32) | 30 (15–67) | 30 (15–49) | 22 (8–38) | 0.003 |
CCS, culture confirmed early-onset neonatal sepsis; PNS, presumed sepsis; ROS, ruled out sepsis; GA, gestational age; SGA, small for gestational age (birth weight <10th percentile); min, minute.
Categorical variables expressed in absolute frequency (percentage). Continuous variable expressed as median and interquartile range.
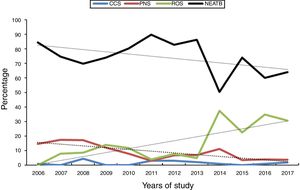
The total number of EONS episodes was 130: 17 (1.3%) cases of CCS and 113 cases of PNS (9.0%). The CCS rate corresponded to a prevalence of 15.5/1000 live VLBW infants, with the number of cases per year ranging from zero to four. In this cohort, from the initial group of newborns whose antimicrobial treatment was administered for sepsis’ suspicion, it was possible to discontinue antibiotic in 44% (192 cases of ROS group, corresponding to 15.4% of the entire cohort) and 932 infants (74.3%) did not require antibiotics up to 48h of life.
In the annual trend assessment of the four groups, the CCS rate remained stable (p=0.906), a significant decrease was observed in the PNS rate (p<0.001) and a significant increase was observed in ROS cases (p<0.001). The rate of NEATB infants was high (minimum of 50.5 and maximum of 84.4%) and with a significant downward trend (p<0.001), as shown in Fig. 1.
Considering the subgroup of extremely low birth weight infants (<1000g), sepsis’ rates showed a pattern similar to that of the entire cohort, with the following prevalence frequencies: CCS, 2.0%; PNS, 12.8%; ROS, 21.3%; and NEATB, 63.1%. The annual distribution was similar to that of the entire group, with a stable rate for CCS (p=0.167), significant decrease in PNS (p=0.001), significant increase in ROS (p=0.001), and stable trend of NEATB over the 12 years (p=0.077).
A decrease was observed in the rate of NEATB newborns and a concomitant increase in the ROS group after 2014. In the comparison of the two periods, 2006–2013 and 2014–2017, significant difference was observed for prevalence of chorioamnionitis (p=0.839), prolonged rupture of membranes (p=0.859), vaginal delivery (p=0.493), and birth weight <1000g (p=0.121), but there was a higher frequency of more immature infants in the early period (23–25 weeks vs. 31 to ≥35 weeks, p=0.046).
The identified microbiological agents in the 17 culture-confirmed cases were: Streptococcus agalactiae (4), Escherichia coli (5), Haemophilus influenzae (2), Streptococcus oralis (1), Staphylococcus aureus (1), Staphylococcus epidermidis (1), Klebsiella pneumoniae (1), and Candida albicans (1). Information on time to positivity was available in 9/17 episodes, with a median time of 9h (ranging from 6 to 20h). Positive cerebrospinal fluid culture was observed in one case (E. coli) and the same agent was identified in the blood culture. Antibiotic susceptibility results showed that 88.2% of Gram-positive bacteria were sensitive to penicillin G and ampicillin, and all Gram-negative bacilli were susceptible to ampicillin and/or amikacin.
NEATB group comprised heavier and more mature infants than the other three groups: birth weight – median 1147g (IQR, 890–1335g), p<0.001 and gestational age median 30 weeks (IQR, 28–32 weeks), p<0.001. Furthermore, a higher frequency of maternal hypertension (45.5%, p<0.0001), antenatal corticosteroid use (78.5%, p<0.0001), and SGA infants (44.7%, p=0.001) were found in this group. They also presented the lowest prevalence of the need for resuscitation at birth (49.5%, p<0.0001) and the lowest median of SNAPPE II score (9 IQR 8–10, p 0.001).
According to Table 1, when comparing maternal variables among the CCS, PNS, and ROS groups, a higher occurrence of diabetes mellitus (17.6%, p=0.023) and chorioamnionitis (41.1%, p=0.002) was observed in the CCS group, while peripartum hemorrhage was more common in the ROS group (7.8%, p=0.001). This latter group also presented a lower SNAPPE II median (22 IQR 8–38, p=0.033). Birth weight (0.590) and gestational age (p=0.896) were similar in the three groups (Table 2).
There were 136 early neonatal deaths in the period, corresponding to 10.8% of the cohort. There was a predominance of this outcome in the CCS (9/17=52.9%) and the PNS groups (22/113; 19.5%). For ROS group, the mortality rate was 5.7% (11/192) and in the NEATB group it was 10.1% (94/932).
Analyzing the distribution of the direct cause of death, all the nine events in the CCS group were attributed to sepsis. In the PNS group, 45.4% (ten cases) were directly caused by EONS, and 36.3% (eight cases) were related to respiratory failure secondary to pulmonary hemorrhage and pneumothorax. In the ROS group, death was mainly due to late sepsis (4; 36.3%) and asphyxia (3; 13.6%). In the NEATB group, the most prevalent direct causes were asphyxia (16%), major congenital malformation (11.7%), late sepsis (10.6%), and pneumothorax (9.5%).
DiscussionThis study presented relevant results regarding to the discontinuation of antibiotic therapy after 48/72h of use. It occurred in more than 40% of the groups whose antibiotics were prescribed due to initial sepsis suspicion (CCS, PNS, and CCS groups). Moreover, nearly three-quarters of the VLBW infants did not require antibiotics, demonstrating the impact of a strategy for the rational use of antibiotics that began over two decades ago and has continued over the years.
From 2013 onwards, the time to discontinue antimicrobials was reduced to 48h, since it was demonstrated in extensive evaluations studies that the time to positivity of blood cultures occurred up to 97% by 48h of incubation.7,12,13 Accordingly, the American Academy of Pediatrics, in its 2018 guideline on the management of newborns born at 346/7 weeks’ of gestation with proven or suspected bacterial sepsis, recommends that antibiotic therapy should be discontinued between 36 and 48h of incubation when blood cultures are negative, unless there is localized disease.14
This one-day shortening is particularly important to newborns and the health care system. According to Ellsbury,15 the unnecessary exposition of antibiotics and other drugs in the neonatal unit means an increase in the risk of adverse drug events, consumes additional nursing and pharmacy resources, and it increases hospital costs.
An interesting aspect of the annual trends of the ROS and the NEATB groups was observed in the more recent years (2014–2017). Compared to previous years, the frequency of more immature infants (24–26 weeks) was significantly more common in the 2014–17 period, without differences regarding another sepsis’ risk factors. The prevalence of EONS is inversely related to gestational age: in a large American cohort, sepsis rate in preterm infants born at 22–28 weeks was threefold higher than that found among moderate preterm infants and more than 18-fold higher than among 34–36 weeks preterm infants.2 Additionally, extreme preterm infants tend to develop a respiratory and/or hemodynamic clinical course more severe than more mature newborns, and this fact contributes to “rule-out sepsis workup” and to starting antimicrobial use.7
These high rates reported above of non-use or discontinuation of antimicrobials at 48–72h of life are uncommon in the literature and clinical practice, both in Brazil and worldwide.10,16 Rugolo et al. conducted a recent survey in Brazil on neonatologists’ perception of risk and management in EONS. There was a trend to overestimate the risk, and even in medium and low-risk scenarios, a considerable number of responders pointed out that they would indicate sepsis tests and start empirical antibiotics.
Differently from the present findings, recent data from a multicenter study involving more than 14,000 VLBW from the Canadian Neonatal Network showed a rate of 21% of non-use and 38% of discontinuation of antibiotics by the third day of life.17 In a contemporary multicenter study, Ellsbury et al.15 evaluated a similar cohort of 58,555 VLBW infants from 2007 to 2013 (median gestational age of 29 weeks and weight of 1100g). Although they had observed a downward trend to ampicillin use rate in the first three days of life, it remained high and ranged from 74.1% to 80%. As for the use for more than three days, despite negative blood cultures, rates varied from 32.4% to 28.4%
More alarming was the reporting of antimicrobial use with no identifiable risk of infection. In the study of Mukhopadhyay and Puopolo,7 2748 VLBW infants were evaluated in the United States, and 605 (22%) of these were newborns of a mother with preeclampsia, born by cesarean section, and without labor, premature rupture of membranes or chorioamnionitis. Almost all of them (99.8%) underwent blood culture at birth, and a significant majority (85%) received empirical antibiotic therapy. Furthermore, another American multicenter study evolving 30 university hospitals in 24 states showed that 94% of 790 extremely low birth weight infants underwent blood culture sampling and received antimicrobials at birth. In approximately 35% of them, antibiotics were discontinued for up to three days, and in 30% they were continued for at least seven days, despite the negative blood cultures and no identifiable antepartum risk factors or clinical signs explaining the prolonged administration of antimicrobial therapy.18
In the assessment of maternal and neonatal characteristics, the NEATB group was associated with SGA infants, those who have greater weight, advanced gestational age, and a higher rate of antenatal corticosteroid use. They were evaluated by the medical team as infants without or low risk of developing EONS. Conversed, infants with prolonged rupture of membranes, chorioamnionitis, vaginal delivery, and need for resuscitation at birth were associated with CCS and PNS, reflecting the clinical reasoning of the neonatal team in terms of investigating and adequately treating such groups of newborns.
According to Ting,17 the concern that inapparent amniotic infection leads to premature labor, premature membrane rupture, and chorioamnionitis, is the most used argument to justify the initiation of antibiotic therapy in the early days of life. However, without an appropriate clinical test, the number of infants treated may be higher than those who are genuinely ill. Clinical aspects of the mother and newborn can be used to identify those at low risk of developing sepsis, and those without significant risk factors should have antibiotics suspended if blood culture is negative and clinical evolution is not compatible with sepsis.
One concern regarding this study was the possibility of recurrence of sepsis in the ROS group. A study by Cantey et al.19 demonstrated that there was no difference in safety outcomes after implementation of automatic discontinuation of antibiotics after five days of treatment in presumed sepsis, compared to previous practices when longer a course of antibiotics was administered, even when blood cultures were negatives. The evaluation of a total of 2502 infants showed that only in seven (0.28%) antimicrobial drugs were reintroduced within 14 days of completing a previous treatment course.
As secondary outcomes, the CCS rate in the present study was low – 1.3%, equivalent to 15.5/1000 VLBW infants, according to worldwide data.2,7,20 Recent data from the Brazilian Neonatal Research Network, from an evaluation of 10,045 VLBW newborns under 33 weeks of gestational age, showed a blood culture positivity rate in EONS of 2.6%, with inter-center variation from 0.4 to 6.4% [personal information].
The microbiological agents identified were those commonly described in the literature, with a predominance of S. agalactiae and Gram-negative enteric bacilli.2,7,21 Antimicrobial susceptibility profile reinforced the empirical treatment in EONS – penicillin G or ampicillin associated with an aminoglycoside. In 1996, this neonatal unit stopped using third-generation cephalosporins for empirical treatment of early and late neonatal sepsis due to the emergence of multidrug-resistant enteric Gram-negative bacilli. A local study showed a significant decrease in multidrug-resistant E. cloacae colonization and infection rates when the empirical use of ceftriaxone was no longer recommended.22 Since then, amikacin has been used as the first-line empirical aminoglycoside for early and late neonatal sepsis.
Two of the proven cases of sepsis were caused by bacteria commonly associated with late sepsis or culture contamination, one by S. epidermidis and the other by S. aureus. As there was a compatible clinical picture in the first 48h of life, associated with early growth in two blood culture samples, altered white blood cell count and abnormal CRP level, they were considered the causal agents. In Schrag's study, S. aureus was responsible for 3.5% of sepsis cases in the first two days of life.23 Rates of EONS caused by coagulase-negative Staphylococcus are usually low, but there are reports of values as high as 10.7% and 22.5%.24,25
A limitation of this study is that the evaluation was performed on a computerized database, without the inclusion of variables such as previous use of antimicrobials by the mother, colonization rate by S. agalactiae, and labor conditions that may impinge on the possibility of developing EONS. Although the evaluation of these variables could not be performed by the study, such clinical aspects were considered by the team responsible for the management of these infants, and it reflects a well-established clinical experience of this unit even after more than 20 years.
In conclusion, the confirmed sepsis rate was low and remained stable. There were a significant downward trend in the PNS rate and a significant upward trend in the ROS group. The rate of NEATB infants was high, with also a significant downward trend. Etiological agents are those commonly associated with EONS and usual antimicrobial sensibility. Early neonatal death predominantly occurred in groups that received antibiotic.
Conflicts of interestThe authors declare no conflicts of interest.