To assess the presence of restless legs syndrome, periodic leg movement, and sleep disorders in female adolescents with idiopathic musculoskeletal pain through a sleep scale and polysomnography, and to compare these data in adolescents without pain history.
MethodTwenty-six adolescents diagnosed with idiopathic musculoskeletal pain followed in a pain outpatient clinic and 25 healthy controls matched by age and education were recruited. The restless legs syndrome criteria were evaluated according to the International Restless Legs Syndrome Study Group, the Sleep Disturbance Scale for Children was completed, nocturnal polysomnography was performed, and anxiety symptoms were recorded.
ResultsThe mean age of idiopathic musculoskeletal pain adolescents was 13.9±1.6 years; in controls, it was 14.4±1.4 years. One adolescent in the control group (4 %) and nine patients with idiopathic musculoskeletal pain (34.6 %) fulfilled the restless legs syndrome criteria (p=0.011). The authors did not observe significant differences in Sleep Disturbance Scale for Children scores between the groups in all components: disorders of initiating and maintaining sleep (p=0.290), sleep breathing disorders (p=0.576), disorders of arousal (p=0.162), sleep-wake transition disorders (p=0.258), disorder of excessive daytime somnolence (p=0.594), and sleep hyperhidrosis (p=0.797). The neurophysiological, respiratory, and periodic leg movement parameters were similar in both groups. Having anxiety was not associated with restless legs syndrome (p=0.11). Three patients with idiopathic musculoskeletal pain (11.5 %) presented restless legs syndrome and periodic leg movement simultaneously, which was absent in the control group.
ConclusionFemale adolescents with idiopathic musculoskeletal pain present criteria for RLS more frequently than healthy adolescents. However, this study did not observe relevant changes in objective and subject sleep variables.
Avaliar a presença de síndrome das pernas inquietas, movimento periódico das pernas e distúrbios do sono em adolescentes do sexo feminino com dor musculoesquelética idiopática por meio da escala do sono e da polissonografia e comparar esses dados em adolescentes sem histórico de dor.
MétodoForam recrutados 26 adolescentes diagnosticados com dor musculoesquelética idiopática acompanhados em um ambulatório de dor e 25 controles saudáveis pareados por idade e escolaridade. Avaliamos os critérios da síndrome das pernas inquietas de acordo com o Grupo Internacional de Estudos de Síndrome das Pernas Inquietas, a Escala de Distúrbios do Sono em Crianças, a polissonografia noturna e os sintomas de ansiedade.
ResultadosA idade média dos adolescentes com dor musculoesquelética idiopática foi 13,9±1,6 anos e dos controles foi 14,4±1,4 anos. Um adolescente no grupo de controle (4%) e nove pacientes com dor musculoesquelética idiopática (34,6%) atenderam aos critérios da síndrome das pernas inquietas (p=0,011). Não observamos diferenças significativas nos escores da Escala de Distúrbios do Sono em Crianças entre os grupos em todos os componentes: distúrbios do início e da manutenção do sono (p=0,290), distúrbios respiratórios do sono (p=0,576), distúrbios do despertar (p=0,162), distúrbios da transição sono-vigília (p=0,258), sonolência diurna excessiva (p=0,594) e hiperidrose do sono (p=0,797). Os parâmetros neurofisiológicos, respiratórios e movimento periódico das pernas foram semelhantes nos dois grupos. Ansiedade não foi associada à síndrome das pernas inquietas (p=0,11). Três pacientes com dor musculoesquelética idiopática (11,5%) apresentaram síndrome das pernas inquietas e movimento periódico das pernas simultaneamente, situação ausente no grupo de controle.
ConclusãoAs adolescentes do sexo feminino com dor musculoesquelética idiopática apresentaram critérios para síndrome das pernas inquietas com mais frequência que as adolescentes saudáveis. Contudo, não observamos mudanças relevantes nas variáveis do sono objetivas e subjetivas.
Musculoskeletal pain is a common complaint in children and adolescents. Numerous children develop a chronic or recurrent pain syndrome that results in severe incapacitation. In situations where pain develops in more than one body system and no specific cause can be found, it is called idiopathic musculoskeletal pain (IMP). This term is used because it is descriptive and recognizes the fact that the cause is unknown.1 The clinical diagnosis of IMP is made by excluding other pain syndromes. IMP is defined by the presence of generalized musculoskeletal pain in three or more areas of the body for at least three months, and these symptoms should not be explained by other causes or diseases. IMP may be localized or diffuse, and its etiopathogenesis is not yet fully understood.1–3
According to the international literature, the prevalence of IMP in school children varies between 12 % and 37.4 %.2,4,5 Involvement in more frequent in girls and the onset of symptoms is most common around 12–13 years.3 Some comorbidities, such as sleep problems, are common in adolescents with pain. The most commonly described problems are related to sleep deprivation, sleep inefficiency, and non-restorative sleep.6 However, other syndromes related to sleep, such as restless legs syndrome (RLS), may be observed.
RLS is characterized by prominent urge to move the legs, usually associated with other uncomfortable and unpleasant sensations. RLS has circadian fluctuation, and symptoms usually begin or worsen during periods of inactivity, intensify at night, and are partially or totally relieved by movement. Physical discomfort and the need to move the limbs can cause difficulty in initiating sleep and frequent night waking.7 RLS may be accompanied by periodic limb movements (PLM) during sleep, which in turn can affect sleep quality, especially in the early stages.7 Stereotypical movement of the lower limbs during sleep is the main feature of PLM, characterized by muscle contraction lasting between 0.5 and ten seconds, and repeating at intervals of five to 90s.8,9 PLM can be evaluated through polysomnography (PSG) in all age groups.
The diagnosis of RLS is based on five essential criteria: 1) need or urgency to move the legs due to discomfort; 2) symptoms worsen or are present exclusively at rest or inactivity; 3) symptoms relieve totally or partially with movement; 4) the symptoms perceived during rest and inactivity worsen or occur at night exclusively; and 5) the symptoms described above are not better explained by other diseases or conditions.10 The presence of PLM helps in the management of these patients when the diagnosis of RLS is doubtful.9
Clinical evidence suggests association between growing pain and RLS in children and adolescents, suggesting that there may also be correlation with other painful syndromes.11 The frequency and morbidity of IMP concern clinicians, as these adolescents are at increased risk of chronic pain when they become adults. The authors’ hypothesis is that there is a relationship between RLS, PLM, and sleep disorders in adolescents with IMP. The aim of this study was to evaluate the presence of RLS, PLM, and sleep disorders in adolescents with IMP, comparing them with healthy adolescents matched by age and education.
MethodsThis was a descriptive, cross-sectional, case control study.
ParticipantsAdolescents were randomly selected by convenience among patients treated at a specialized outpatient clinic of a tertiary referral center. From a population of 180 patients from the musculoskeletal pain outpatient clinic of the Pediatric Rheumatology Division of the Universidade Federal de São Paulo, 78 % were girls, aged 12–16 years, and diagnosed with IMP. We recruited 26 female adolescents with the diagnosis of IMP, as they complained of pain in three or more body areas.1 The control group consisted of 25 healthy volunteer adolescents, with no complaints of pain. All adolescents and parents were informed about the procedures of the research protocol, including the requirements necessary to perform the nocturnal PSG. The groups were matched by age and education.
The exclusion criteria were the presence of a medical diagnosis of neurological or psychiatric diseases, including attention deficit hyperactivity disorder (ADHD) or characteristics of the clinical criteria of this disorder. It was decided to consider ADHD as an exclusion criterion due to the association of RSL, PLM, and ADHD.
ProceduresIMP is more frequent in girls and the two boys who were invited to the study refused to participate. Thus, the study included only female adolescents. The invitation to participate was made by telephone call, during which the purpose and stages of the study were stated. Voluntary participation in the study was later reinforced in the personal interview. The evaluations of patients and controls consisted of the following stages.
- 1)
For anamnesis, this study used a semi-structured interview of the participant and parent with the objective of describing and evaluating personal aspects, along with relational, systemic, and educational aspects. The Scale for Assessment of Socioeconomic Status of the Brazilian Association of Research Companies 2012. The clinical evaluation of patients and controls was performed by two pediatric rheumatologists, with over 30 years of combined experience (C.A.L. and M.T.S.L.R.A.T).
- 2)
Clinical sleep evaluations were made based on the essential criteria of the American Academy of Sleep Medicine, 2005, and the International Restless Legs Syndrome Study Group (IRLSSG), 2012.9,10 The criterion of 2003 states that the child must complete all four RSL criteria used for adults: 1) urge to move the limbs, associated with discomfort; 2) relief of this sensation with the movement; 3) worsening or occurrence of symptoms at rest position; 4) worsening of symptoms at night; and, one more aspect. Either the child reports with her own words the unpleasant sensation and/or leg discomfort or two of these three features are present: period leg movements (more five per hour) during sleep, sleep disorder, and parent or siblings with the diagnosis of RLS. In 2012, the IRLSSG excluded the requirement for two of three features and included the fifth criterion, which requires that the symptoms described above are not better explained by other diseases or conditions.12 Support resources and clinical aspects are also considered, which are important in the diagnostic evaluation.9,10,12
- 3)
The current study evaluated the presence of sleep problems using the Sleep Disturbance Scale for Children (SDSC).13 Evaluation of nutritional status was performed through weight and height measurements, calculation of the body mass index (BMI), using World Health Organization (WHO) curves, was performed with Anthro Plus software, v. 3.2.2, 2009.
- 4)
The authors also evaluated anxiety symptoms in the group of adolescents with DMEI and in the Control-through-Child Scale control group, which assesses the form of anxious reaction in children, without constituting an anxiety disorder.14
- 5)
Baseline polysomnography (PSG) consisted of electroencephalogram (F3 / A2, F4 / A1, C3A2, C4 / A1, O2 / A1, O1/ A2), electro-oculogram, chin and leg electromyogram, electrocardiogram, nasal cannula, oronasal thermistor, thoracic and abdominal movement (inductive plethysmography), microphone, oxygen saturation, and body position (EMBLA® S7000 computerized system – Broomfield, CO, United States).15 Staging of sleep and periodic leg movement followed the international consensus, according to the American Academy of Sleep Medicine Manual.16 PSG scoring was performed by a Sleep Medicine-Certified physician (G.A.M.).
The exclusion criteria were also confirmed through completion of the SNAP-IV questionnaire,17 the screening instrument for ADHD that assesses the first criteria (criteria A) of the Diagnostic and Statistical Manual of Mental Disorders (DSM) V.
Data analysisThe sample size calculations were performed using the statistical software 2008 Power Analysis and Sample Size System (PASS – NCSS). The statistical software SPSS v. 20.0 was used for analysis. Categorical variables were expressed as absolute and relative frequencies, while numerical variables were described as means and standard deviation. Given the sample size, the associations between two categorical variables were tested using Fisher’s exact test. Student’s t-test for independent samples or, alternatively, the non-parametric Mann-Whitney U test, were used to compare numerical data between groups. Non-parametric tests were used in situations of groups with small numbers of cases (fewer than ten cases) or violation of the normality assumption, which was tested using the Kolmogorov-Smirnov test. A significance level of 5 % was adopted for all statistical tests.
Ethical approval and consent to participateThis study was approved by the Ethics Committee, opinion no. 304.751. The informed consent and assent terms were obtained from parents and adolescents, respectively.
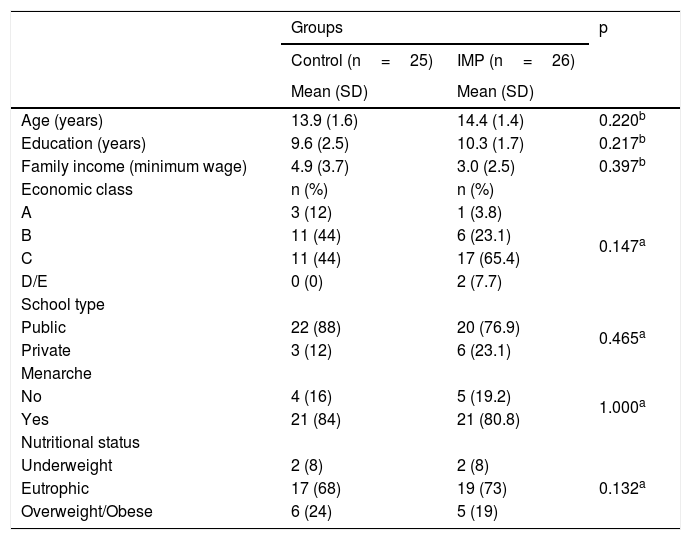
ResultsAll subjects recruited for this study were female. No significant differences were observed between the IMP and control groups regarding age (p=0.220), years of education (p=0.217), family income (p=0.397), socioeconomic status (p=0.147), and school type (p=0.465). The age at menarche was equivalent between the groups (p=0.220) as was the nutritional status distribution (p=0.132), which showed no statistically significant differences (Table 1).
Demographic and socioeconomic data of patients with IMP and controls.
| Groups | p | ||
|---|---|---|---|
| Control (n=25) | IMP (n=26) | ||
| Mean (SD) | Mean (SD) | ||
| Age (years) | 13.9 (1.6) | 14.4 (1.4) | 0.220b |
| Education (years) | 9.6 (2.5) | 10.3 (1.7) | 0.217b |
| Family income (minimum wage) | 4.9 (3.7) | 3.0 (2.5) | 0.397b |
| Economic class | n (%) | n (%) | |
| A | 3 (12) | 1 (3.8) | 0.147a |
| B | 11 (44) | 6 (23.1) | |
| C | 11 (44) | 17 (65.4) | |
| D/E | 0 (0) | 2 (7.7) | |
| School type | |||
| Public | 22 (88) | 20 (76.9) | 0.465a |
| Private | 3 (12) | 6 (23.1) | |
| Menarche | |||
| No | 4 (16) | 5 (19.2) | 1.000a |
| Yes | 21 (84) | 21 (80.8) | |
| Nutritional status | |||
| Underweight | 2 (8) | 2 (8) | 0.132a |
| Eutrophic | 17 (68) | 19 (73) | |
| Overweight/Obese | 6 (24) | 5 (19) | |
IMP, idiopathic musculoskeletal pain; SD, standard deviation.
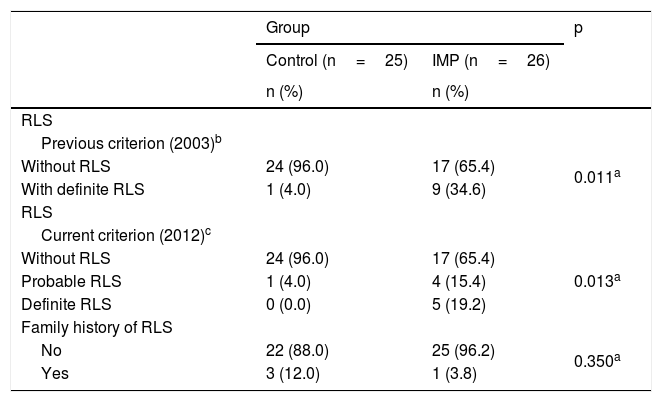
Adolescents with IMP presented the RLS diagnostic criteria more frequently than the control group, regardless of the diagnostic criteria used (Table 2). When the children’s evaluation criteria of 2003 were used,7 it was observed that one individual in the control group had definite RLS, while nine in the IMP group had definite RLS (4.0 % vs. 34.6 %; p=0.011). When the current criteria of 2012 were used,12 it was observed that one individual in the control group had probable RLS, whereas nine in the IMP group presented probable or definite RLS (4.0 % vs. 34.6 %; p=0.013). RLS was identified in the same individuals from the IMP and control groups using the 2003 and 2012 diagnostic criteria. No differences were observed regarding the family history for RLS in the groups studied.
Criteria for the diagnostic evaluation of RLS and family history in patients and controls.
| Group | p | ||
|---|---|---|---|
| Control (n=25) | IMP (n=26) | ||
| n (%) | n (%) | ||
| RLS | |||
| Previous criterion (2003)b | |||
| Without RLS | 24 (96.0) | 17 (65.4) | 0.011a |
| With definite RLS | 1 (4.0) | 9 (34.6) | |
| RLS | |||
| Current criterion (2012)c | |||
| Without RLS | 24 (96.0) | 17 (65.4) | 0.013a |
| Probable RLS | 1 (4.0) | 4 (15.4) | |
| Definite RLS | 0 (0.0) | 5 (19.2) | |
| Family history of RLS | |||
| No | 22 (88.0) | 25 (96.2) | 0.350a |
| Yes | 3 (12.0) | 1 (3.8) | |
RLS, restless legs syndrome.
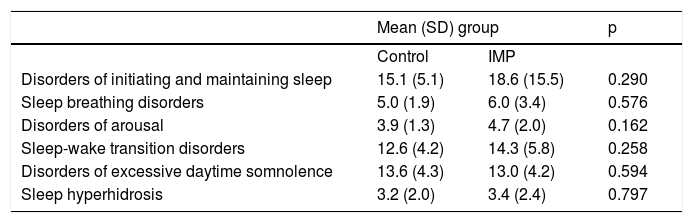
No significant differences were observed between the IMP and control groups in relation to any component of the SDSC, including disorders of initiating and maintaining sleep (p=0.290), sleep breathing disorders (p=0.576), disorders of arousal (p=0.162), sleep-wake transition disorders (p=0.258), disorders of excessive daytime somnolence (p=0.594), and sleep hyperhidrosis (p=0.797). The complete data are shown in Table 3.
Findings of the Sleep Disorder Scale for Children and Adolescents (SDSC).
| Mean (SD) group | p | ||
|---|---|---|---|
| Control | IMP | ||
| Disorders of initiating and maintaining sleep | 15.1 (5.1) | 18.6 (15.5) | 0.290 |
| Sleep breathing disorders | 5.0 (1.9) | 6.0 (3.4) | 0.576 |
| Disorders of arousal | 3.9 (1.3) | 4.7 (2.0) | 0.162 |
| Sleep-wake transition disorders | 12.6 (4.2) | 14.3 (5.8) | 0.258 |
| Disorders of excessive daytime somnolence | 13.6 (4.3) | 13.0 (4.2) | 0.594 |
| Sleep hyperhidrosis | 3.2 (2.0) | 3.4 (2.4) | 0.797 |
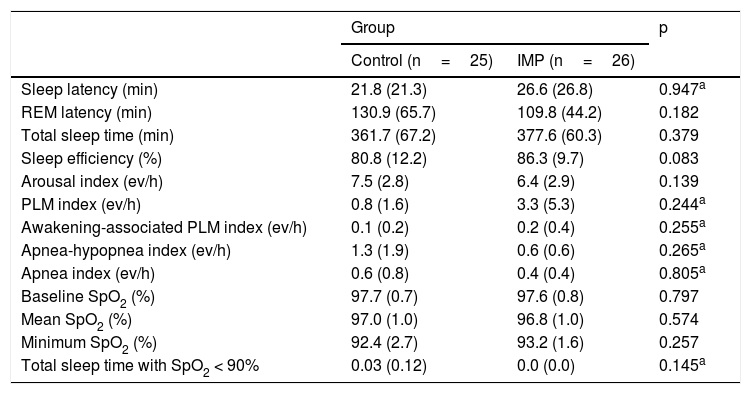
The summary of the PSG findings is presented in Table 4. No significant differences in the neurophysiological, respiratory, and PLM parameters were observed between the control and IMP groups.
Polysomnographic findings of the control and IMP groups.
| Group | p | ||
|---|---|---|---|
| Control (n=25) | IMP (n=26) | ||
| Sleep latency (min) | 21.8 (21.3) | 26.6 (26.8) | 0.947a |
| REM latency (min) | 130.9 (65.7) | 109.8 (44.2) | 0.182 |
| Total sleep time (min) | 361.7 (67.2) | 377.6 (60.3) | 0.379 |
| Sleep efficiency (%) | 80.8 (12.2) | 86.3 (9.7) | 0.083 |
| Arousal index (ev/h) | 7.5 (2.8) | 6.4 (2.9) | 0.139 |
| PLM index (ev/h) | 0.8 (1.6) | 3.3 (5.3) | 0.244a |
| Awakening-associated PLM index (ev/h) | 0.1 (0.2) | 0.2 (0.4) | 0.255a |
| Apnea-hypopnea index (ev/h) | 1.3 (1.9) | 0.6 (0.6) | 0.265a |
| Apnea index (ev/h) | 0.6 (0.8) | 0.4 (0.4) | 0.805a |
| Baseline SpO2 (%) | 97.7 (0.7) | 97.6 (0.8) | 0.797 |
| Mean SpO2 (%) | 97.0 (1.0) | 96.8 (1.0) | 0.574 |
| Minimum SpO2 (%) | 92.4 (2.7) | 93.2 (1.6) | 0.257 |
| Total sleep time with SpO2 < 90% | 0.03 (0.12) | 0.0 (0.0) | 0.145a |
Data are expressed as means and standard deviations; PLM, periodic limb move movements; ev/h, events per hour; SpO2, percutaneous oxyhemoglobin saturation; p, Descriptive level of Student’s t-test or Mann-Whitney test (a).
According the correlation between PLM indexes in the control and IMP groups. The authors observed one individual of the control group with an index of > 5/h and six patients in the IMP group with indexes of > 5/h. These findings were not significantly different between groups (p=0.099).
When evaluating the simultaneous presence of RLS criteria and the presence of PLM, it was observed that three (50 %) of six patients with RLS in the IMP group had PLM. In the control group, there were no adolescents with RLS and PLM.
DiscussionThis is the first study that has evaluated the presence of RLS and sleep quality in a group of female adolescents with IMP. Patients and controls were evaluated using questionnaire for sleep disorders,13 in addition to PSG. This design made a more complete evaluation of sleep possible, commonly implicated in the etiopathogenesis of pain. It was observed that adolescents with IMP more frequently met the current criteria for definitive RLS diagnosis.12 In contrast, no significant differences in sleep disorders were observed between patients and controls.
The association between sleep problems, RLS, and musculoskeletal pain has been reported in studies involving children and adolescents.18 However, in the present study, these variables were evaluated in the same group of pediatric patients followed up in a tertiary reference center for musculoskeletal pain. These results have practical and clinical implications because in the medical consultations of patients with IMP, sleep is of central importance in the therapeutic orientation for patients and their families, either by the clinician or by other professionals of the multidisciplinary team.
As the patients are part of the IMP outpatient clinic, they were already undergoing treatment that includes pain education, mainly explanations to patients and their caregivers about pain coping, as well as relaxation techniques and measures to prevent catastrophizing pain. In addition, this clinic guides the performance of reactive aerobic physical activities. According to individual needs, patients are referred for specialized psychological treatment. In general, drugs such as painkillers, anti-inflammatory drugs, or drugs with action on the nervous system are avoided, since there is no evidence for the use of these therapies for IMP.
Due to the change in essential criteria to be considered in diagnosis in 2012,19 the results were shown according to the previous and current diagnostic RLS criteria.7,12 In this small sample, no differences between the two RLS criteria were found. The literature shows that RLS affects 1.3%–5.9% of adolescents19–21 and there are no studies on the prevalence in children and adolescents with IMP. Definitive RLS was observed in 4 % of healthy adolescents and 34.6 % of patients with IMP. The difference in the RLS frequencies among adolescents with and without pain was significant, which justifies the study of RLS criteria in children and adolescents with IMP.
Sleep disorders such as non-restorative sleep, assessed by questionnaires, have been described in adult women with fibromyalgia.22
A recent study with adolescents evaluated factors influencing excessive daytime somnolence, and the authors observed a higher prevalence of excessive somnolence disorder, sleep hyperhidrosis, and greater sleep restriction during the week using the SDSC.23 Sander et al. found a negative impact on the quality of sleep in children and adolescents regarding the difficulty of returning to sleep in nocturnal awakenings, presence of excessive movements, and daytime sleepiness.21 In turn, in children and adolescents with IMP, sleep disorders such as difficulties in initiating and maintaining sleep and problems in the sleep-wake transition were observed using the SDSC.24 The present study did not observe differences between the groups regarding the sleep disorder components evaluated through the questionnaire or the PSG. The authors believe this may be due to the fact that adolescents with IMP are followed at a specialized clinic, where they receive guidance on the importance of good sleep hygiene. When a sub-analysis was conducted including the ten girls with complaints of difficulty initiating and maintaining sleep (seven IMP and three controls), it was observed that only two girls in the IMP group had the criterion for RSL and a complaint of this sleep disorder (p=0.17).
Although no significant differences were observed between the groups in the frequency of the PLM index, six patients in the RLS group had a PLM index > 5ev/h, whereas in the control group, only one adolescent presented with this finding. Although not significant, the difference may exist, it was not detected given the small sample size (type II error). When evaluating patients with IMP, it was observed that three presented RLS and PLM simultaneously during sleep. Another important point is that PLM is not the main cause of nocturnal sleep disturbance in RLS, nor is sleep disturbance necessarily a consequence of increased PLM during sleep, although RLS is frequently associated with PLM.25 In addition, PLM during sleep occur in a variety of sleep disorders, including narcolepsy, obstructive sleep apnea, and rapid eye movement sleep behavior disorder (REMSBD).26
The presence of RLS and/or PLM during sleep is relevant in adolescents with IMP because the treatment of these sleep disorders has the potential to reduce pain.2 This finding suggests a common pathophysiological mechanism among the three disorders.27 PLM recording, which is an important item for improving diagnostic accuracy, is a practical factor to consider in the detection of PLM in RLS. The present study did not demonstrate greater polysomnographic changes in this sample of individuals, which could be explained by the fact that the results refer only to a baseline PSG conducted in one night. Studies with adults and children suggest that a longer assessment period of at least two days would be more appropriate, as there is a great night-to-night variability of PLM recordings. PLM evident on a particular night can vary significantly in any individual with RLS, which leads to the conclusion that the extent of nocturnal variability of PLM can lead to false-negative results in the characterization of RLS.28,29
The IMP group had 15 adolescents with anxious traits according to the Child Anxiety Trait Scale.14 Of these, four had RLS. In the control group 11 adolescents had anxiety, none with RLS. Having anxiety was not associated with RLS (p=0.11). In a study conducted the present service30 stress, anxiety, and depression were assessed in a group of patients with IMP from the outpatient clinic. Increased stress was noted, but neither anxiety nor depression were observed.
This study has some limitations regarding sample size. It is important to note that patients with IMP were already followed in a specialized clinic and were therefore exposed to guidelines that include sleep hygiene. Additional limitations include the nocturnal variability of PLM and the use of questionnaires in which the respondent needed to recall information about their responses, which leads to a certain inaccuracy or loss of information.
These findings have practical importance, since it was observed that adolescents with IMP have a higher frequency of RLS when compared to healthy pain-free controls. Pediatricians need to be aware of aspects of sleep, especially the difficulty in initiating and maintaining sleep, and the symptoms of RLS. It is necessary to investigate the presence of sleep deprivation, and, in some cases, perform polysomnography. Along these lines, the authors believe that studies on the presence of RLS and PLM involving a larger number of patients with IMP are needed, as well as for a more detailed assessment of pain correction with these clinical findings.
In this study, it was concluded that adolescents with IMP presented a higher frequency of RLS when compared with healthy controls without pain. However, no significant differences were observed in the frequency of sleep disorders, nor objective sleep parameters. Further studies with larger samples and with sleep assessments of naive subjects, as well as long term evaluation, including more data addressing nocturnal PLM variability, may provide useful information on the correlation between sleep disorders and the presence of pain.
FundingThis study was supported financially by CAPES – Coordination of Improvement of Higher Level Personnel and FAPESP – Foundation for Research Support of the State of São Paulo.
Conflicts of interestThe authors declare no conflicts of interest.
The authors thank the principal and coordination team of the Joao Evangelista Elementary School and High School, the volunteers and their parents for the participation in this study, and the employees of the Sleep Institute for conducting the exams. They also thank the friends of the Children’s Rheumatology team of UNIFESP and the ACREDITE – Friends of Children with Rheumatism – for collaborating and making this research viable.
Please cite this article as: Amorim RA, Moreira GA, Santos FH, Terreri MT, Molina J, Keppeke LF, et al. Sleep and restless legs syndrome in female adolescents with idiopathic musculoskeletal pain. J Pediatr (Rio J). 2020;96:763–70.