To analyze the development and prevalence of gastrointestinal signs and symptoms associated with the development of the digestive tract, and to assess the measures aimed to reduce their negative impacts.
Source of dataConsidering the scope and comprehensiveness of the subject, a systematic review of the literature was not carried out. The Medline database was used to identify references that would allow the analysis of the study topics.
Synthesis of resultsInfants frequently show several gastrointestinal signs and symptoms. These clinical manifestations can be part of gastrointestinal functional disorders such as infantile colic, infant regurgitation, and functional constipation. Allergy to cow's milk protein and gastroesophageal reflux disease are also causes of these clinical manifestations and represent an important and difficult differential diagnosis. The diseases that course with gastrointestinal signs and symptoms can have an impact on family dynamics and maternal emotional status, and may be associated with future problems in the child's life. Comprehensive pediatric care is essential for diagnosis and treatment. Maternal breastfeeding should always be maintained. Some special formulas can contribute to the control of clinical manifestations depending on the established diagnosis.
ConclusionDuring the normal development of the digestive tract, several gastrointestinal signs and symptoms may occur, usually resulting from functional gastrointestinal disorders, gastroesophageal reflux disease, and allergy to cow's milk protein. Breastfeeding should always be maintained.
Analisar o desenvolvimento e a prevalência de sinais e sintomas gastrintestinais associados com o desenvolvimento do tubo digestivo e as medidas que visam diminuir suas repercussões negativas.
Fontes dos dadosConsiderando a abrangência e amplitude do tema, não foi realizada revisão sistemática da literatura. Utilizou-se a base de dados do Medline para a identificação de referências bibliográficas que permitissem contemplar os temas de estudo.
Síntese dos resultadosO lactente apresenta com elevada frequência vários sinais e sintomas gastrintestinais. Estas manifestações clínicas podem fazer parte de distúrbios funcionais gastrintestinais como a cólica do lactente, regurgitação do lactente e constipação intestinal funcional. A alergia à proteína do leite de vaca e doença do refluxo gastroesofágico também são causas destas manifestações clínicas e representam um importante e difícil diagnóstico diferencial. As doenças que cursam com sintomas e sinais gastrintestinais podem ter consequências na dinâmica familiar e no estado emocional das mães. Podem se associar com problemas na vida futura da criança. A atenção pediátrica completa é fundamental para o diagnóstico e o tratamento. O aleitamento natural deve sempre ser mantido. Algumas fórmulas especiais podem contribuir para o controle das manifestações clínicas na dependência do diagnóstico estabelecido.
ConclusãoDurante o desenvolvimento normal do tubo digestivo podem ocorrer vários sinais e sintomas gastrintestinais em geral decorrentes dos distúrbios gastrintestinais funcionais, da doença do refluxo gastroesofágico e da alergia à proteína do leite de vaca. Aleitamento natural deve sempre ser mantido.
Since the conception, characterized by the moment when the sperm enters the egg, the biological potential for the formation of a new human being develops into a fascinating process of cell multiplication and differentiation. The maturation of the individual occurs during the course of different stages of life. Currently, the first 1000 days, starting at conception, are considered to be a critical period to define the health status of the individual and can have consequences throughout life.1,2
The first two years of life are an important part of this period, which is characterized by accelerated growth rate and development of several organs and systems. Therefore, gastrointestinal signs and symptoms can occur in infants,3,4 which may be linked to several anatomical and functional changes observed at this stage of life. In addition to these clinical manifestations, there may be repercussions in the individual's future,5 as well as consequences that will interfere in family dynamics and the parents’ emotional status.
It can be said that the development of the digestive system comprises not only the anatomical and functional aspects of its organs but also local and systemic interactions with the intestinal immune system and the establishment of the gastrointestinal microbiota. The interaction among these three processes has been the object of many basic science and clinical studies searching for alternatives that can provide favorable results during the first 1000 days and several future cycles of life.
This article aimed to analyze the emergence and prevalence of gastrointestinal signs and symptoms associated with the development of the gastrointestinal tract and measures aimed to reduce their negative impacts.
MethodsConsidering the scope and comprehensiveness of the subject, a systematic review of the literature was not carried out. The Medline database was used to identify literature references that included the subject of the study. Among others, were used the following terms in the search for information, “digestive,” “tract,” “ontogeny,” “microbiota,” “development,” “premature,” “oral,” and “tolerance”. When necessary, the research was restricted to humans. The author also used his personal experience and trends discussed in national and international scientific events.
Anatomical and functional development of the gastrointestinal tractThe intestine is one of the most complex organs in the human body, both from the anatomical and functional point of view. The intestinal cells and tissues are formed from all three germ layers. Intestinal stem cells derive from the mesoderm, the muscle tissue of the endoderm, and the enteric nervous system of the ectoderm.6 In addition to the digestive and absorptive functions, there are also the intestinal endocrine system (involved in the regulation of systemic levels of nutrients and feeding behavior) and the so-called intestinal barrier, which prevents the passage of the intestinal contents, including microorganisms, into the body.
The epithelial function is carried out by four types of cells: enterocytes, which are responsible for absorption, and three types of secretory cells (mucus-producing goblet cells, antibacterial substance-producing Paneth cells, and several enteroendocrine cells that regulate satiety, intestinal absorption, proliferation of pancreas β-cells, and hormone secretion, among others).6 It is also noteworthy the intense epithelial proliferation of stem cells, which renew the luminal surface of the intestine in a few days, and the complex interaction among the epithelium, smooth muscle, and enteric nervous system, to ensure a unidirectional flow provided by intestinal peristalsis.6
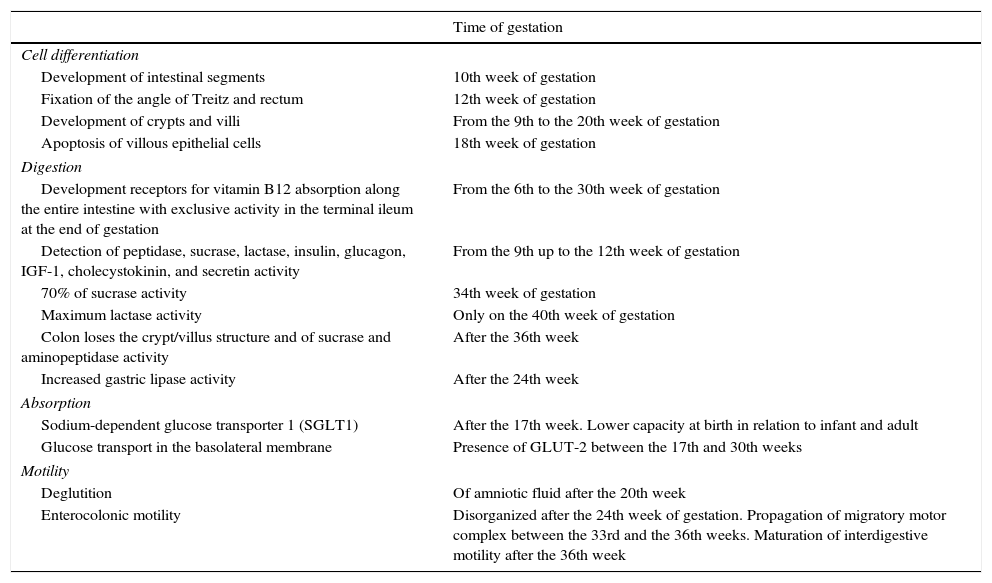
The development of the intestine during gestation can be divided into four basic processes: 1. cell differentiation; 2. digestion; 3. absorption; and 4. motility.7Table 1 shows the major milestones of intestinal development.7
Main milestones of intestinal development7
| Time of gestation | |
|---|---|
| Cell differentiation | |
| Development of intestinal segments | 10th week of gestation |
| Fixation of the angle of Treitz and rectum | 12th week of gestation |
| Development of crypts and villi | From the 9th to the 20th week of gestation |
| Apoptosis of villous epithelial cells | 18th week of gestation |
| Digestion | |
| Development receptors for vitamin B12 absorption along the entire intestine with exclusive activity in the terminal ileum at the end of gestation | From the 6th to the 30th week of gestation |
| Detection of peptidase, sucrase, lactase, insulin, glucagon, IGF-1, cholecystokinin, and secretin activity | From the 9th up to the 12th week of gestation |
| 70% of sucrase activity | 34th week of gestation |
| Maximum lactase activity | Only on the 40th week of gestation |
| Colon loses the crypt/villus structure and of sucrase and aminopeptidase activity | After the 36th week |
| Increased gastric lipase activity | After the 24th week |
| Absorption | |
| Sodium-dependent glucose transporter 1 (SGLT1) | After the 17th week. Lower capacity at birth in relation to infant and adult |
| Glucose transport in the basolateral membrane | Presence of GLUT-2 between the 17th and 30th weeks |
| Motility | |
| Deglutition | Of amniotic fluid after the 20th week |
| Enterocolonic motility | Disorganized after the 24th week of gestation. Propagation of migratory motor complex between the 33rd and the 36th weeks. Maturation of interdigestive motility after the 36th week |
These physiological properties are very important to understand the feeding process of the newborn, especially those preterm. The transition from parenteral feeding via the umbilical cord and small amounts of swallowed amniotic fluid to obtaining complete nutrients from the colostrum and breast milk occurs at birth.7 At that time, the intestine undergoes a rapid morphological growth process in terms of length and absorptive surface. It is noteworthy that the intestinal length is estimated at 50cm in the middle of pregnancy, at approximately 100cm in the weeks before birth, and 200cm in the first weeks of life.7 The colostrum stimulus is important in this process.7
Preterm infant feeding is a major challenge due to the immaturity of the digestive system (especially of the swallowing reflex), the lower lactase activity, and the immature pattern of the gastrointestinal tract motility. These facts explain, at least in part, the high prevalence of food intolerance, gastroesophageal reflux, and constipation in preterm infants.7
Colonization of the digestive tractThe development of molecular biology techniques, which do not depend on bacterial isolation through conventional culture methods, has led to extraordinary increase in the knowledge of microorganisms that live in the intestine.8 More than 1013 microorganisms can be found per gram of intestinal contents in the colon. Knowledge about bacterial diversity has also increased since the identification of new species that comprise the digestive tract microbiota. It is believed that the number of genes in the human microbiota is 100 times higher than in the human body. It is noteworthy that there is a mutual and beneficial interaction between the microbiota and the host. Thus, there are not only nutritional interactions (colonic fermentation with the production of short-chain fatty acids that are absorbed in the colon, and lipid and protein hydrolysis) but also interaction with the intestinal immune system.1,8
It should be remembered that the intestinal microbiota is not constituted only of bacteria. Fungi and viruses are also found in the lumen of the gastrointestinal tract. The potential therapeutic use of viruses interfering with the intestinal microbiota composition represents a new challenge in research on the microbiome of humans.9
The belief that the fetus lives in a sterile environment has been modified in recent years based on evidence of the existence of microorganisms in the amniotic fluid, umbilical cord blood, fetal membranes, and the placenta.10 These findings should be interpreted with caution due to the possibility of contamination of the samples at the time of collection.10 The changes in the mother's vaginal and colonic microbiota during pregnancy are noteworthy, considering that the maternal microbiota influences the formation of the fetus’ and the newborn's microbiota.10
On the first day of life there is fast newborn intestinal colonization by microorganisms from the maternal and environmental microbiota.8 The first colonizing agents belong to the genera Escherichia and Enterococcus. Subsequently, anaerobic bacteria of the Bifidobacterium and Bacteroidetes genera appear. That is, the first bacteria are facultative anaerobic (Staphylococcus, Streptococcus, Enterococcus, Enterobacter) that contribute to the development of an anaerobic environment in the intestine, which thus allows the for colonization by obligate anaerobes (Bifidobacterium, Bacteriodetes, Clostridium, Eubacterium).1
With the introduction of complementary foods to breast milk in the infant's diet an important impact is observed on the intestinal microflora, characterized by decreased participation of bifidobacteria (which, however, remains predominant) and increased diversity with greater participation of bacteria from the genera Bacteroides and Clostridium.1 A study performed with Brazilian infants receiving exclusive breastfeeding identified six phyla in the fecal microbiota: Bacteroidetes, Firmicutes, Fusobacterium, Proteobacteria, Actinobacteria, and Verrucomicrobia.11 At three months of life, there was a predominance of Streptococcus and Escherichia; at six months, the predominance of Escherichia was observed.11
The colonization of the digestive tract of the newborn and infant depends on several factors, especially the type of delivery and type of feeding. Over the first two years of life, it is observed that the caesarean delivery is associated with greater abundance of Firmicutes and lower of Bacteroidetes. During the first six months of life it is observed that the colonization by Bacteroidetes occurs at a later phase. At the end of two years of life, the relative abundance of microbial constituents is similar in both infants born by vaginal delivery and caesarean section. It was also observed that the circulating levels of cytokines produced by helper T lymphocytes are lower among those born through caesarean section. It should be noted that the fecal microbiota of mothers of children born through caesarean section and vaginal delivery were not different.12
Regarding the type of feeding, it is well known that the intestinal microbiota of infants who are exclusively breastfed is different from those fed with artificial formula.13–15 Breast milk is rich in oligosaccharides that influence the composition of the intestinal microbiota.16,17 It is also known that the oligosaccharide profile in breast milk is not the same in all mothers.18,19 The specific effects of these different oligosaccharide profiles on the intestinal microbiota are not yet fully known.18,19 However, breast milk was considered practically sterile until some years ago. Recently a hypothesis was raised that suggests a new way of communication between the mother's and the infant's microbiota.20,21 In this context, it is suggested that bacteria from the mother's intestinal microbiota would reach the breast milk by translocation from the intestinal lumen and through bloodstream transportation, featuring an internal enteromammary pathway.20,21 This hypothesis was created based on animal studies. These bacteria could influence the infant's colonization process and maturation of the immune system. Another possible pathway is the entry of bacteria from the skin tissue of the mother in the mammary gland through the nipple.20,21 Thus, colostrum and human milk are not only sources of oligosaccharides that stimulate the formation of intestinal microflora but also probably a source of bacteria for the infant. It is estimated that 800mL of breast milk can contain up to 105 to 107 colony-forming units. The bacteria that have been identified in breast milk samples belong mainly to the genera Lactobacillus, Staphylococcus, Enterococcus, and Bifidobacterium.20,21
The intestinal microbiota, through molecular structures that constitute the microbe-associated molecular patterns (MAMPs) interacts with the intestinal immunological system and the intestinal barrier and also interferes with the production of mucus. They stimulate cell proliferation in the crypts and Paneth cells responsible for producing the antimicrobial peptides called defensins. This interaction (“crosstalk”) occurs with specific receptors, such as the toll-like receptors. Pro-inflammatory responses can be neutralized by the specialized regulatory T (Treg) cells through interleukin 10 production.
This mechanism is very important in the development of oral tolerance. Probiotic bacteria such as Lactobacillus GG and Bifidobacterium breve can stimulate the process of immunological tolerance through interleukin-production stimulation.1,22,23
The M cells located next to Peyer's patches are responsible for the presentation of intestinal lumen contents and thus, stimulate the immunological system of the mucosa.1
Abnormalities in oral tolerance development in the first months of life can cause non-IgE-mediated allergy to cow's milk protein.24
Gastrointestinal signs and symptoms in infantsThe occurrence of digestive symptoms is common in the first months of life, such as regurgitation, vomiting, colic, and constipation. Some clinical manifestations, although they do not constitute a defined disease, may be of concern to parents, such as flatulence, which, incidentally, is hard to quantify to be characterized as excessive or not. Many of these symptoms may be transient and are attributed to immaturity and/or are considered as part of the development of the gastrointestinal tract. Many of these clinical manifestations are included in functional gastrointestinal disorders and their diagnosis can be established by the Rome III criteria.25 The gastroesophageal reflux disease (GERD) and allergy to cow's milk protein are not functional gastrointestinal disorders; however, they can lead to several gastrointestinal signs and symptoms, especially in the first year of life.26,27 In pediatric practice, allergy to cow's milk protein and GERD are often part of the differential diagnosis of gastrointestinal functional disorders.28,29
Therefore, the infant can have different gastrointestinal signs and symptoms, especially in the first six months of life. The physician should recognize the clinical manifestation as isolated and/or transient or as part of a functional gastrointestinal disorder, allergy to cow's milk, or even GERD. Other diseases that are not restricted to the digestive tract are included in the differential diagnosis. Only after careful evaluation and definition of the diagnostic hypothesis it is possible to define the approach to be used. Table 2 presents information that can contribute to the differential diagnosis of gastrointestinal signs and symptoms in infants.25–32 Cyclic vomiting syndrome and rumination were not included, because they are not as prevalent as other functional gastrointestinal disorders.
Diagnostic criteria for infant diseases presenting with predominantly gastrointestinal symptoms.
| 1. Infantile colic | Rome III Criteria (2006):25 all of the following characteristics from birth to 4 months of age: 1. episodes of paroxysms of irritability, fussing, or crying that starts and stops without obvious causes; 2. episodes lasting three or more hours/day for three days/week for at least one week; no failure to thrive. |
| 2. Infant regurgitation (“physiological reflux”) | Rome III Criteria (2006):25 All of the following characteristics in otherwise healthy infants: 1. two or more regurgitations per day for three or more weeks; no retching, hematemesis, aspiration, apnea, failure to thrive, feeding or swallowing difficulties, or abnormal posture. |
| 3. Functional intestinal constipation | Rome III Criteria (2006)25 and ESPGHAN/NASPGHAN guideline (2014):30 duration of at least on month of two or more of the following characteristics: 1. fewer than three bowel movements a week; 2. at least one weekly episode of fecal incontinence after sphincter control; 3. history of excessive retention of feces; 4. history of pain and/or difficulty in bowel movements; 5. presence of large fecal mass in the rectum; 6. history of elimination of large-diameter feces that can clog the toilet. Additional symptoms: irritability, decreased appetite, and early satiety. These symptoms disappear after elimination of large amounts of feces. |
| 4. Functional diarrhea (“irritable bowel syndrome”) | Rome III Criteria (2006):25 all of the following characteristics: 1. daily painless, recurrent passage of three or more large, unformed stools; 2. duration longer than 4 weeks; 3. onset between 6 and 36 months of age; 4. passage of stools during waking hours; 5. No failure to thrive if caloric intake is adequate. |
| 5. Infantile dyschezia | Roma III Criteria (2006):25 It should include the two following characteristics in infants younger than 6 months: 1. at least 10min of straining and crying before successful passage of soft stools; 2. no other health problems. |
| 6. Gastroesophageal reflux disease (GERD) | NASPGHAN/ESPGHAN Guideline (2009):26 It is present when gastroesophageal reflux causes symptoms that are uncomfortable and/or complications. Clinical manifestations suggestive of GERD before 18 months: recurrent regurgitations and/or vomiting accompanied by failure to thrive; stressed behavior or crying without explanation. |
| 7. Allergy to cow's milk protein | Brazilian Consensus (2007)31 and ESPGHAN Guideline (2012):27 adverse reaction, reproducible, caused by an immune reaction triggered by antigen(s) of certain food(s). In infants, it is often a delayed reaction (non-IgE-mediated). In a group of 159 infants with suspected allergy to cow's milk protein, the following gastrointestinal signs and symptoms were found (each infant could have more than one clinical manifestation): regurgitation and vomiting in 53.5%; colic in 34.0%; diarrhea in 25.2%, of which approximately 30% with blood; blood in the stool in 14.5%; and constipation in 15.7%. Weight and length deficit were commonly observed.32 In most cases, the diagnosis must be confirmed by challenge test with the suspected food, to be performed four to 12 weeks after the start of the elimination diet when the symptoms have already been controlled.27,31 |
Infantile colic, infantile regurgitation, constipation, functional diarrhea, and infantile dyschezia are included among the gastrointestinal functional disorders and the Rome III criteria may be used for the diagnosis (see comments in the text). Gastroesophageal reflux disease and allergy to cow's milk protein are included in the table because they are prevalent causes of gastrointestinal signs and symptoms in infants and have events that are similar to gastrointestinal functional disorders.
In 2015, a group of specialists from several countries evaluated the available information in the literature on the prevalence of gastrointestinal functional disorders in the first year of life.5 The median prevalence of infantile colic in 30 articles was 18%; however, great variability was observed due, at least in part, to the diversity of diagnostic criteria. It is interesting to mention that, in a study conducted in Brazil that characterized infantile colic according to the Wessel criterion, 16% of the 1086 infants assessed had colic; however, according to their mothers, 80% had colic.33 Therefore, there is no full consensus among the different diagnostic criteria used in the studies of prevalence, as well as the opinions and expectations of parents. Regarding the prevalence of infant regurgitation, 13 articles were retrieved, with results ranging from 3% to 87%. The Rome III criteria was used in only two of these 13 articles (two or more regurgitations a day for more than three weeks), with a prevalence of 17% and 26%. It should be noted that, in general, infant regurgitation disappears spontaneously up to 12 months of life.5,34,35 In turn, the prevalence of GERD in infants is unknown, but it is accepted that it is much lower than that of infant regurgitation.35 The prevalence of functional constipation in the first year of life showed great variation, from less than 1% to 39% in the eight studies retrieved.5 Two cross-sectional studies that used the Rome III criteria found a prevalence of constipation of 0.05–5%.5 In the author's opinion, the Rome III criteria underestimate the diagnosis of intestinal constipation, especially in the first year of life.36,37 A recent study compared the prevalence of intestinal constipation in 831 Brazilian infants using the Rome III criteria and another broader criterion (elimination of hard stools with pain/difficulty and/or less frequently than three times a week; or elimination of cylindrical hard stools with cracks or scybalous stools, even if eliminated without pain or difficulty).38 According to the Rome III criteria, the prevalence of intestinal constipation was 1.1%, whereas the broader criterion identified constipation in 19.6% of the study population.38
It is estimated that the prevalence of allergy to cow's milk protein ranges from 2% to 3% in the first year of life.27 It must be remembered, however, that clinical manifestations suggestive of allergy to cow's milk are present in a higher percentage of infants, approximately 9% according to a prospective study carried out in the Netherlands.39 It can be estimated that only a portion of infants (approximately 30–50%) that undergo an elimination diet will show a positive challenge test, confirming the diagnosis.39,40
Thus, it can be observed that many infants have gastrointestinal signs and symptoms. Some of these infants have more than one symptom.4,41 These gastrointestinal manifestations are often reasons for changes in the infants’ diet, mainly changes in the type of infant formula.42,43
Pediatric care for infants with gastrointestinal clinical manifestationsThe first point to be considered is whether the infant's gastrointestinal sign or symptom is actually part of a disease or simply part of the normal digestive physiology or of the development of the gastrointestinal tract in the first year of life.
During pediatric visits, parents often mention that their children pass an excessive volume of gas. An article with suggestions of conduct for frequent gastrointestinal signs in infants discusses this question, associating it with crying and fussiness.3 It is noteworthy that the presence of gas in the digestive tract is normal. Excess gas, however, could result from an inappropriate breastfeeding technique, with the occurrence of aerophagia.3
Excessive crying and fussiness are often considered as gastrointestinal manifestations.44,45 In 1954, Wessel defined infantile colic as episodes of irritability, agitation, or intense crying for at least three hours, three days a week and lasting more than three weeks (“rule of three”).46 In general, it disappears at approximately four months of life.44,45 In 2006, infantile colic was included in the Rome III criteria (Table 2), which aims to standardize the diagnosis of functional gastrointestinal disorders.25 In practice, both for health professionals and parents, these criteria do not always have to be fully met for infantile colic to be considered a concern and a serious problem.
A study conducted in the pediatric emergency department of the Netherlands has shown that, in the care of infants with crying as the main cause of consultation, approximately 6% of parents acknowledged the practice of actions that could pose a serious health hazard to infants, such as shaking (which can be a cause of the “shaken baby syndrome”) and attempts at suffocation.47 Alarming results were found in another study carried out in Japan.48
Mothers of infants with colic may have decreased quality of life, especially in the areas of physical and social performance, and increased risk of depression,49,50 which may decrease with the control of colic.51 Thus, infantile colic, expressed as intense and inconsolable crying, constitutes an important health problem in the first four months of life. In addition to the infant's suffering, it can be the cause of a great deal of anxiety in parents and reduce the family's quality of life.49 It should be emphasized that pediatric assessment of crying infants should be detailed and careful, aiming to identify causes that require specific therapy. A retrospective study conducted in Toronto highlights the importance of clinical history, physical examination, and urinalysis in the evaluation of febrile infants with crying, irritability, and fussiness.52 It is important to remember that infantile colic occurs equally in the presence of natural and artificial feeding.44,45
In general, regurgitation as an isolated manifestation in infants reflects the occurrence of physiological reflux (infant regurgitation according to the Rome III criteria, Table 2). In GERD other signs are observed associated with regurgitation and vomiting, including weight gain deficit and clinical manifestations attributed to probable reflux esophagitis, such as irritability, excessive crying, and difficulty feeding.26,34,35 The diagnosis of GERD is essentially clinical. Infants with mild symptoms and no warning signs are called “happy vomiters” and there is no need for medication.34
There are questionnaires developed to help differentiate between infantile regurgitation and GERD. In the analysis of a questionnaire for this purpose, the clinical evaluation by an experienced specialist was used as reference, supplemented or not with other exams.53 Ultrasonography has no value in differentiating infantile regurgitation from GERD26,34,35 and thus, it should not be indicated for assessment of the infant with suspected GERD. It is very useful when there is suspicion of hypertrophic pyloric stenosis. Contrast radiography of the esophagus, stomach, and duodenum may be indicated when anatomical abnormalities of the upper gastrointestinal tract are suspected.26,34,35
Two articles have been recently published on the practice of Brazilian pediatricians regarding the evaluation of infants with excessive crying or suspected GERD.54,55 In the first study, 132 pediatricians were asked to analyze a case of an exclusively breastfed infant with excessive crying, regurgitation, and fully satisfactory weight gain. The diagnostic hypothesis of GERD was suggested by 63% of respondents, while 24% considered the possibility of infantile colic.54 This result suggests that there is an overestimation of the possibility of GERD in the clinical context. The second study involved 140 professionals who analyzed two typical clinical cases, one of infantile regurgitation (physiological reflux) and another with GERD.53 Based on the answers about the approach to be adopted, the authors concluded that most pediatricians correctly differentiated physiological reflux from GERD.55
Regarding the intestinal habits, it should be noted that, in general, the passage of first meconium occurs within the first 24h of life. Preterm newborns with less than 1500g of birth weight may have the passage of first meconium after 48h.56
There are few studies in the literature that evaluated intestinal habits of infants.56–62 Considering the information in these articles, it can be said that, in the first month of life, most infants have four to six bowel movements a day. This number decreases after the second month of life. The number of stools and stool consistency is associated with the type of feeding. It is well established that breastfed infants have more bowel movements when compared with formula-fed infants.58,60 The stool of breastfed infants is softer than those who receive infant formula.56,58 The type of infant formula can also influence the consistency of stools and stool frequency. Infants fed conventional or soy infant formula had half the frequency of bowel movements and harder stool consistency compared to breastfed infants.63 In comparison, those receiving extensively hydrolyzed protein formula have an increased frequency of bowel movements and softer stool consistency.63
The main changes in the infant's intestinal habits are diarrhea and constipation. Generally, diarrhea is defined as the occurrence of three or more loose stools in the preceding 24h.64,65 It is a broad definition that includes bacterial and viral infections of the digestive tract (acute and persistent diarrhea), which can cause dehydration and malnutrition, and are not included in the scope of this article. Nonetheless, it is worth mentioning that some parents mistakenly consider the frequency and consistency of stools in the first month of life as “diarrhea”, which actually constitutes normal bowel habits. Conversely, intestinal constipation usually starts with the elimination of hard, scybalous stools, with pain or difficulty.66 However, the Rome III criteria25 does not take into account the shape and consistency of stools to characterize intestinal constipation in infants, which can delay the diagnosis.
Principles and perspectives for the control of gastrointestinal signs and symptoms in infantsFor infants with colic and regurgitation, the most important procedure is elucidating parents about the benign and transitory characteristic of these manifestations.3,4,26,34,44,45 In this context, parents should be sure that additional exams are not required and that their children are not at immediate or future risks. It is advisable to check the sources that parents are possibly accessing to obtain information about the health of their children and, when necessary, appropriate and corrective pediatric guidelines should be offered.
It is important to avoid prescribing unnecessary medications. A common approach is to conduct unjustified and often prolonged empiric treatment for probable esophagitis due to gastroesophageal reflux in infants with excessive crying and irritability.
In this context, it is important to mention the abusive use of proton-pump inhibitors in infants.67 According to clinical trials68,69 and a meta-analysis,70 proton-pump inhibitors provide no reduction in the daily duration of infantile crying and can also cause adverse affects.67 A study carried out in Brazil showed that prokinetics, such as domperidone and bromopride, are considered in the treatment of infants with infantile regurgitation and GERD,55 which goes against the most recent recommendations.26,34,35
A study carried out in 11 European countries71 with 567 pediatricians showed that few follow the NASPGHAN/ESPGHAN (North American Society for Pediatric Gastroenterology, Hepatology and Nutrition/European Society for Pediatric Gastroenterology, Hepatology, and Nutrition) guideline, published in 2009.26 The reasons for the lack of adherence to the care protocols should be the subject of future studies. It should be noted that there is no evidence that dimethicone is effective in the treatment of infantile colic.44,45
Regarding the feeding regimen, exclusive breastfeeding should always be maintained. No gastrointestinal signs or symptoms should be understood as reason for breastfeeding interruption, as shown in the reviews34,44,45 and guidelines26,27,30,31,35,36 for the treatment of gastrointestinal functional disorders.
Regarding infants with suspected allergy to cow's milk while receiving exclusive breastfeeding, the mother should eliminate cow's milk proteins from her diet.27,31 Infants with food allergies during breastfeeding and with severe clinical manifestations, with stunting and/or iron deficiency anemia, for instance, should be individually assessed by specialists. Specialized assessment should also be carried out in infants with suspected disease such as galactosemia and congenital glucose–galactose malabsorption, among others.
For infants who no longer receive breast milk and when relactation cannot be achieved, there are several formulas that have been developed to contribute for the control of gastrointestinal signs and symptoms in infants, including: 1. thickened formulas, 2. soy formulas, 3. formulas with partially hydrolyzed proteins and lower lactose content, 4. formulas with prebiotics, 5. formulas with extensively hydrolyzed proteins, with and without lactose, and 6. amino acid formulas.
Thickened formulas provide a decrease in the frequency and volume of regurgitation. Thus, they can decrease the parents’ anxiety. It should be noted that they are formulas that reduce regurgitation, but not the total time of exposure of the esophagus to gastric acid.26,34,35
Another situation that constitutes a major healthcare problem is that of infants with regurgitation, vomiting, irritability, and excessive crying, associated or not with other clinical manifestations. Diagnostic hypotheses of GERD and allergy to cow's milk protein are often considered. The ESPGHAN/NASPGHAN guideline for infants with frequent regurgitation and irritability, associated or not with decreased weight gain, recommends the exclusion of cow's milk protein from the diet for two to four weeks.26 The guideline suggests formulas with extensively hydrolyzed proteins or amino acid formulas as substitutes.26 Thus, in case of patient improvement after this period of elimination diet, a challenge test should be planned to confirm the diagnosis.27 However, many physicians that recommend cow's milk elimination from the diet do not perform the challenge test to ascertain the diagnosis of allergy to cow's milk.72,73 This procedure maintains a substantial proportion of infants39,40 that are not allergic to cow's milk (but who casually had a reduction in clinical manifestations) on a replacement diet, which is more expensive, for a period longer than necessary. Although not indicated for infants younger than six months, about one-third of pediatricians would prescribe soy formula to infants with suspected gastroesophageal reflux secondary to allergy to cow's milk protein.55 It should be noted that soy formulas are not indicated for the treatment of infantile colic,3,4,44,45 allergy to cow's milk protein in the first six months of life,27,31 infant regurgitation, or GERD.26,34,35
Formulas with partially hydrolyzed proteins and low lactose content have been used in infants with gastrointestinal manifestations such as colic, regurgitation, flatulence, and hardened stools, based on open clinical trials.74–77 It can be hypothesized that their effectiveness in these circumstances is related to the following mechanisms: 1. increased rate of gastric emptying78,79 and 2. decrease in the effects of excessive fermentation of carbohydrates in the intestinal lumen, considering that in patients with infantile colic, a greater production of hydrogen in the expired air has been demonstrated in the hydrogen breath test.80–82 It should be noted that no formula with partially hydrolyzed proteins, with normal or low content of lactose, should be used in the treatment of infants with suspected allergy to cow's milk protein.77
Breastfed infants have low risk for constipation. This may be a consequence of the oligosaccharide content in human milk, which increases the frequency of bowel movements and decreases stool consistency.83 Several mechanisms may account for this effect:81 1. increased microbial mass (including probiotic bacteria) due to the availability of oligosaccharides to be fermented; 2. selective growth of Bifidobacterium and Lactobacillus bacteria that ferment the oligosaccharides and produce short-chain fatty acids that increase the water content in the stool.83 In turn, the short-chain fatty acids can stimulate intestinal motility, as they are used as an energy source by colonocytes and induce tonic and phasic contractions in the circular muscles of the colon; and 3. oligosaccharides play the role of soluble fibers and, thus, increase the water content of the stool.83
The same effect on intestinal habit and stool consistency can be obtained by adding prebiotics to infant formula.84–87 Thus, more frequent bowel movements with softer consistency can be obtained with the use of infant formula with added prebiotic, which can potentially reduce the likelihood of constipation onset. The NASPGHAN/ESPGHAN guideline offers a specific algorithm for constipation before six months of age.30
Probiotics are live microorganisms that, when consumed in adequate amounts, provide a beneficial effect to the host's health.88,89 Infants with colic90 or allergy to cow's milk protein91 may show alterations in the intestinal microbiota, as observed in functional gastrointestinal disorders and other allergic diseases.92 Thus, the role of probiotics in controlling these diseases in infants has been the subject of several studies.
Clinical trials51,93–95 and a meta-analysis96 showed that Lactobacillus reuteri DSM 17938 provides a reduction in daily crying time in patients with infantile colic. In turn, Lactobacillus GG can accelerate the acquisition of tolerance by infants with allergy to cow's milk protein.22,23 From a clinical point of view, it is important to emphasize that the effect of a particular probiotic is specific to a particular strain. A clinical trial showed that administration of Lactobacillus reuteri DSM 17938 can reduce the frequency of gastrointestinal symptoms in the first months of life.97 These data show how the development of research in the field of prebiotics and probiotics for the prevention and control of gastrointestinal signs and symptoms in infants is promising.
In conclusion, the prevalence of gastrointestinal signs and symptoms in infants is very high. The diagnosis is usually based on information from parents and the clinical examination of the child, including weight assessment. The lack of specific and precise tests may hinder the differential diagnosis. Offering recommendations and support to parents is crucial, regardless of the diagnosis. Exclusive breastfeeding should always be stimulated and maintained.
Conflicts of interestThe author has presented at conferences and/or performed scientific consulting for Danone Nutrição Especializada, Danone Early Nutrition, Mead Johnson, Laboratório Bago, and Laboratório Aché.
Please cite this article as: Morais MB. Signs and symptoms associated with digestive tract development. J Pediatr (Rio J). 2016;92(3 Suppl 1):S46–56.