To compare the clinical evolution in patients with refractory functional constipation undergoing different therapeutic regimens: oral laxatives and antegrade enemas via appendicostomy or clinical treatment with oral laxatives and rectal enemas.
MethodsAnalysis of a series of 28 patients with a mean age of 7.9 years (2.4–11), followed-up in a tertiary outpatient clinic. Refractory functional constipation was defined as continuous retentive fecal incontinence after at least a 12-month period of consensus therapy. After the diagnosis of refractory condition, appendicostomy was proposed and performed in 17 patients. Outcomes: (1) persistence of retentive fecal incontinence despite the use of enemas, (2) control of retentive fecal incontinence with enemas, and (3) control of retentive fecal incontinence, spontaneous evacuations, with no need for enemas.
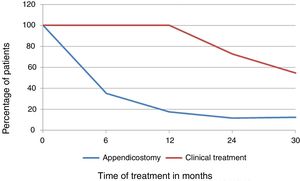
ResultsSix and 12 months after the therapeutic option, control of retentive fecal incontinence was observed only in patients who underwent surgery, 11/17 and 14/17, p=0.001 and p=0.001, respectively. At 24 months, control of retentive fecal incontinence was also more frequent in operated patients: 13/17 versus 3/11 with clinical treatment, p=0.005. In the final evaluation, the median follow-up times were 2.6 and 3 years (operated vs. clinical treatment, p=0.40); one patient in each group was lost to follow-up and 9/16 operated patients had spontaneous bowel movements vs. 3/10 in the clinical treatment group, p=0.043. Surgical complications, totaling 42 episodes, were observed 14/17 patients.
ConclusionAppendicostomy, although associated with a high frequency of complications, controlled retentive fecal incontinence earlier and more frequently than clinical treatment. The choice of one of the methods should be made by the family, after adequate information about the risks and benefits of each alternative.
Comparar a evolução clínica em crianças com constipação intestinal funcional refratária sob diferentes regimes terapêuticos: laxativos orais e enemas anterógrados via apendicostomia ou tratamento clínico com laxativos orais e enemas via retal.
MétodosAnálise de uma série de 28 pacientes, 7,9 anos (2,4-11), acompanhados em ambulatório terciário. Constipação intestinal funcional refratária foi definida como manutenção da incontinência fecal retentiva, em terapia consensual, por pelo menos 12 meses. Após diagnóstico de refratariedade, era proposta apendicostomia. Dezessete pacientes realizaram o procedimento cirúrgico. Desfechos: 1. Manutenção de incontinência fecal retentiva em uso de enemas; 2. Controle da incontinência fecal retentiva em uso de enemas; e 3. Controle da incontinência fecal retentiva, evacuações espontâneas, sem necessidade de enemas.
ResultadosSeis e 12 meses após opção terapêutica, controle da incontinência fecal retentiva foi observado apenas nos pacientes operados, 11/17 e 14/17, p=0,001 e p=0,001. Aos 24 meses, controle da incontinência fecal retentiva também mais frequente nos operados 13/17 versus 3/11 tratamento clínico, p=0,005. Na avaliação final, medianas de tempo de seguimento: 2,6 e 3 anos (operados versus tratamento clínico, p=0,40), um paciente em cada grupo abandonou o seguimento e 9/16 operados apresentavam evacuações espontâneas versus 3/10 no tratamento clínico, p=0,043. Complicações cirúrgicas, 42 episódios, acometeram 14/17 pacientes.
ConclusãoA apendicostomia, embora associada a elevada frequência de complicações, controlou a incontinência fecal retentiva de maneira mais precoce e frequente que o tratamento clínico. A escolha de um dos métodos deverá caber à família, após adequada informação sobre riscos e benefícios de cada alternativa.
The prevalence of functional constipation (FC) in children is high in Western societies.1 In Brazil, the importance of this condition in the demographic profile of pediatric diseases is demonstrated by its prevalence, between 17% and 36%, observed in schoolchildren and in specialized outpatient clinics, respectively.1,2
Half of children followed-up for 6–12 months recover from FC, achieving regular bowel movements without the need for laxatives3; however, in approximately 25% of patients treated in tertiary outpatient clinics, complaints of infrequent bowel movements and retentive fecal incontinence (RFI) persist for long periods, and even up to adulthood.3–5 When the therapeutic response is unsatisfactory at maximum doses of laxatives associated with the use of enemas and there are no alarm signals to investigate an organic cause justifying the symptoms, the condition is termed refractory or intractable intestinal constipation.4
This condition includes a complex and heterogeneous group of patients, in which the symptom-triggering variables are not fully established. RFI negatively interferes in social interaction, and the quality of life of these patients was considered to be lower than that of individuals with inflammatory bowel disease and gastroesophageal reflux disease.6 The need to overcome the symptoms culminates, sometimes, in the consideration of a surgical procedure to eliminate RFI and keep the left colon and rectum empty.7 The use of antegrade enemas was introduced by Malone in 1990 with the aim of emptying the left colon and achieving control of fecal incontinence.8 The greatest benefit of this procedure in the long term is the possibility of colonic motility recovery.9
The literature regarding the rates of patients with refractory functional constipation (RFC) treated with appendicostomy who became independent of the ostomy is scarce.10 The aim of this study was to compare the long-term clinical evolution of the use of appendicostomy for antegrade enemas associated with laxatives with the clinical treatment, comprising oral laxatives and rectal enemas, in patients with RFC.
MethodsThis series of 28 cases included patients with FC followed-up at a tertiary service outpatient pediatric gastroenterology clinic and in whom the symptoms met the diagnostic criteria of Roma III11,12 and showed no alarm signals justifying the investigation of organic causes. All patients underwent anorectal manometry with recording of the rectal sphincter reflex presence, to ensure the functional integrity of the myenteric plexus.
Patients were included in a study for scintigraphic assessment of colonic transit and continued to be systematically followed-up on the same basis.13 The evaluation was systematized, the attendance to follow-up consultations was monitored, and families were asked about their access to the medication, either with their own resources or through donations. They were also asked about possible side effects of the medication and advised against modifying medication dose without medical consultation. The presence of fecal impaction was confirmed by the report of retentive fecal incontinence in the anamnesis, presence of fecal mass at the abdominal palpation, and digital rectal examination. If it was not possible to perform a rectal examination, due to the patient's non-consent, a plain abdominal X-ray in the anteroposterior view was requested. Once the fecal impaction was confirmed, the patient was admitted to the university hospital to undergo rectal enemas with glycerin solution at 12%, 20mL/kg, three times a day for three to five days and, depending on the clinical evaluation of the enema output, a radiological assessment was performed prior to hospital discharge.
The diagnosis of RFC was established when RFI persisted after at least 12 months of follow-up with the medical specialty, during treatment with maximum tolerable doses of osmotic laxatives (lactulose 2mL/kg/day or polyethylene glycol 3350, 1.5-2g/kg/day), associated with rectal enemas (12% glycerin solution or saline solution, 20mL/kg/day, up to 500mL); the enemas were recommended if spontaneous evacuation did not occur after two days, in accordance with international guidelines.10–12
When the diagnosis of RFC was made, a study of the colonic transit time was carried out by scintigraphy, and the appendicostomy procedure was offered to all patients, being accepted by 17 of them. After the colonic transit study, and after the decision for any of the therapeutic measures, the evolution of these patients continued to be systematically recorded in their medical records, on the same basis described above.
To analyze the evolution of intestinal constipation symptoms during follow-up, three outcomes were considered: (1) maintenance of the RFI with enemas, (2) control of the RFI with enemas and (3) control of the RFI and spontaneous evacuations, without enemas.
Colon transit study – the nuclear study of colonic transit was carried out according to the service protocol.13 Two types of motor patterns were observed: slow colonic transit (SCT), when 48-h images showed a large part of the marker in the proximal and transverse colon, and distal retention (DR), if the radioisotope had passed the transverse colon within 30h after the test meal but remained retained in the rectosigmoid region for at least 48h of observation.13,14
Operative technique – the appendicostomy was performed by laparotomy or laparoscopy, and the patient was hospitalized for approximately two days. After two weeks, intermittent catheterization was started.15
Solutions for enemas through appendicostomy/rectal administration – enemas were performed when spontaneous evacuations did not occur after 48h with 0.9% saline solution or 12% glycerin solution, 10–20mL/kg. In the operated patients, this solution was infused with the patient sitting in the toilet. When they had spontaneous bowel movements and did not use the stoma, patients were instructed to catheterize the orifice at least once a day, in order to avoid stenosis.
Statistical analysisThe data were analyzed using IBM Statistic SPSS (IBM SPSS Statistics for Windows, version 20.0. NY, EUA). The Mann–Whitney and Fisher exact tests were applied to continuous and categorical variables, respectively. p-values<0.05 were considered significant.
The study was approved and authorized by the institution's Research Ethics Committee on July 28, 2015, #: 163,392. CAAE 47138715.9.0000.5404.
ResultsDuring a five-year period, 28 patients were diagnosed with RFC. Clinical data regarding duration and treatment of constipation are shown in Table 1. The median age at constipation onset was 1 year, ranging from 3 months to 7 years of age in the group that chose appendicostomy, and 1 year, ranging from 3 months to 3 years of age in the group that chose clinical treatment (p=0.68, Mann–Whitney's test). The median age of the patients at the beginning of the follow-up in the tertiary service was 8.3 years (2.5–10.8) and 6.7 years (2.4–11), respectively, for the appendicostomy and clinical treatment groups (p=0.70, Mann–Whitney's test).
Duration and type of treatment for constipation in 28 patients with refractory functional constipation, classified according to the type of treatment: appendicostomy or clinical treatment.
| Median (min–max) | Total | Type of treatment | pa | |
|---|---|---|---|---|
| n=28 | Appendicostomyn=1713 boys | Clinicaln=119 boys | ||
| Duration of constipation until referral to the tertiary service (years) | 5 (1.3–10.6) | 5.4 (1.3–9.7) | 5 (2–10.6) | 0.62 |
| Total time of FC (previous history+tertiary service) (years) | 11.4 (4.4–18.3) | 12 (6.4–18.3) | 10.8 (4.4–16) | 0.20 |
| Follow-up time in the specialty until final evaluation (years) | 5.7 (1.7–12) | 6.4 (2.4–12) | 4 (1.7–7) | 0.008 |
| Time of follow-up after the therapeutic option (years) | 3.3 (0.7–6) | 2.6 (1.4–5) | 3 (0.7–6) | 0.40 |
FC, functional constipation.
The comparative analysis of the outcomes between the appendicostomy and clinical treatment groups is shown in Fig. 1 and Table 2. In the final evaluation, one patient in each group was lost to follow-up. Among the patients who underwent the appendicostomy, two out of 16 discontinued stomal irrigation for stenosis after four months and 2.4 years, respectively, and restarted using rectal enemas, persisting with RFI; for the purpose of outcome analysis, these patients were maintained in the surgical group.
Clinical symptoms assessed at follow-up of 28 patients with refractory functional constipation, distributed according to the type of treatment: appendicostomy or clinical treatment.
| Assessments | Type of treatment | pd | |
|---|---|---|---|
| Appendicostomy (n=17) | Clinical (n=11) | ||
| 6 months | |||
| RFI with the use of enemas | 6 (35.3%) | 11 (100%) | 0.001 |
| Control of RFI with the use of enemas | 11 (64.7) | 0 (0.0%) | |
| Spontaneous regular bowel movements | 0 (0.0%) | 0 (0.0%) | |
| 12 months | |||
| RFI with the use of enemas | 3 (17.6%) | 11 (100%) | 0.001 |
| RFI control with the use of enemas | 14 (82.4%) | 0 (0.0%) | |
| Spontaneous regular bowel movements | 0 (0.0%) | 0 (0.0%) | |
| 24 months | |||
| RFI with the use of enemas | 2 (11.8%) | 8 (72.7%) | 0.005 |
| RFI control with the use of enemas | 13 (76.4%) | 3 (27.3%) | |
| Spontaneous regular bowel movements | 2 (11.8%) | 0 (0.0%) | |
| Final evaluationc | (n=16)a | (n=10)b | |
| RFI with the use of enemas | 2 (12.5%) | 6 (54.6%) | 0.043 |
| RFI control with the use of enemas | 5 (32.3%) | 1 (9.0%) | |
| Spontaneous regular bowel movements | 9 (56.2%) | 3 (27.4%) | |
A total of 42 complications were observed in 17 patients, with some patients having more than one complication; these complications resulted in the need for 22 surgical reinterventions and stoma use discontinuation by three patients. The use of appendicostomy was discontinued in three situations: one patient with morbid obesity, who had contraindication for surgical reintervention, one who decided against the reintervention after stoma stenosis, and another who abandoned treatment after seven complications. Two patients required reconstruction of the left stoma. In the presence of stoma leakage, an anti-reflux valve was created, and the stoma was re-constructed. Therefore, 14/17 patients (88.2%) maintained adherence to antegrade irrigations even after complications were observed.
The colonic transit patterns and association with the outcome at the end of the study are shown in Table 3.
Outcomes according to colonic transit pattern in patients with refractory functional constipation and treated with appendicostomy or clinical treatment.
| Type of treatment | Total | Colonic transit | pc | |
|---|---|---|---|---|
| DR | SCT | |||
| Appendicostomy | n=16a | n=7 | n=9 | |
| RFI with the use of enemas | 3 (17.7%) | 1(14.3%) | 2 (20.0%) | 1.00 |
| RFI control with the use of enemas | 5 (29.4%) | 2 (28.5%) | 3 (30.0%) | |
| Regular bowel movement | 8 (47.0%) | 4 (57.2%) | 4 (40.0%) | |
| Clinical treatment | n=10b | n=6 | n=4 | |
| RFI with the use of enemas | 6 (54.5%) | 4 (57.1%) | 2 (50.0%) | 0.87 |
| RFI control with the use of enemas | 1 (9.0%) | 1 (14.3%) | 0 (0.0%) | |
| Regular bowel movement | 3 (27.2%) | 1 (14.3%) | 2 (50.0%) | |
DR, distal retention; SCT, slow colonic transit; RFI, retentive fecal incontinence.
This study assessed RFI control at 6 and 12 months after appendicostomy or clinical treatment; at 24 months, the frequency of RFI control remained higher in those submitted to the operation, however, these patients had many surgical complications.
The conditions associated with the therapeutic refractoriness of FC are not clarified, and it has not been defined whether this condition would result from long-term fecal impaction or if it is a primary alteration in colonic motility.14 Treatment delay, defined as the time between the age of onset of intestinal constipation and admission to a specialized service, is inversely related to the time of recovery of the clinical condition.5
In this study, a minimum period of 12 months with monthly return visits, under protocol therapeutics, systematized anamnesis, and schedule control, was used to demonstrate adherence to therapy, as best as possible, and to define refractoriness. For the definition of therapeutic non-response, studies have considered periods varying from six months to two or three years of clinical follow-up.16,17 In 2014, the duration of symptoms to define refractoriness was established as a period of at least three months, under appropriate treatment, in a consensus of the European and American societies of Pediatric Gastroenterology.4 In Brazil, nonadherence to therapy is a factor that definitely impairs the evolution of children with functional intestinal constipation.18 The systematic outpatient follow-up allowed us to more concretely assess the magnitude of the risks and benefits observed when comparing therapeutic options.
Appendicostomy is an alternative for a minority of patients with functional intestinal constipation, followed-up in tertiary services and who do not respond to consensus therapy. In literature, the procedure success is reported to range between 57% and 100% of the cases, and such rates were obtained based on different outcomes: complete RFI remission, improvement of abdominal pain or quality of life, presence of evacuation after irrigation via appendicostomy, and recovery of regular bowel movements.7,19,20 Dolejs et al.21 observed that, three months after surgery, 89/93 (96%) of the patients had RFI control. In the present series, six months after the appendicostomy, 82% of patients no longer had RFI, and it was also observed that the early occurrence of complications close to the procedure may have contributed to a delay in RFI remission. Dolejs et al.21 also observed that 24% of patients recovered their capacity to evacuate spontaneously and regularly, with appendicostomy reversion after a median time of 40 months (22/93 children). In the group of 25 articles systematically reviewed by Siminas et al.,10 this outcome was verified in 9.5% of the patients. The recovery of spontaneous evacuation movements is an outcome achieved, on average, in less than half of the patients with refractory constipation submitted to appendicostomy, even after long-term follow-up.9,22,23 Colonic manometry studies have shown that the presence of high-amplitude peristaltic contractions (HAPCs) and peristaltic response to bisacodyl can predict success with the use of antegrade irrigation.23 Rodriguez et al. observed that the recovery of manometric indicators of colonic peristalsis function is associated with the possibility of discontinuing the use of the appendicostomy, an outcome observed in 11/40 of the studied patients.9
Other authors have indicated that those patients who do not reach this response may be those with primary colonic motility disorders.20 Appendicostomy evaluation for longer follow-up periods, on average 10 years, showed a loss of efficacy over time, and about 60% of patients maintained the use of antegrade irrigations after this period. These patients, nevertheless, report high rates of satisfaction and several reported that the transition to adult outpatient clinics was associated with a loss of professional support they had as children and abandonment of the technique.24
Unsatisfactory appendicostomy results were associated with three factors: patient's age at the time of the procedure,25 as children under 6 years of age would have more difficulty accepting the procedure due to the incorrect use of enemas, which appear to be more effective if performed in the postprandial period and as infusions with a duration of 30 and 60min, and the undesirable effects of irrigation, such as pain, during catheterization or infusion.7,26 There are no controlled studies to evaluate the best irrigation solution, and different services have been using various guidelines regarding composition and volumes of irrigation solutions, as well as the addition or not of osmotically active agents. Siddiqui et al.20 found that saline solution has a lower risk of electrolytic imbalance and that the addition of osmotic agents (polyethylene glycol) increases the success rates of irrigation.
High rates of surgical complications have been recorded in all evaluated cases.7,20,23,27 The most prevalent complications, which usually require surgical reinterventions, are stoma stenosis and leakage. In an attempt to minimize these complications, several modifications have been proposed to the initial surgical technique: skin flap creation with other types of skin suture, implantation of devices in cecostomies, button, broviacs, stoppers, and left Malone.28 The devices reduce stenoses or leakage, but are associated with other complications such as displacements, infections, and formation of granulation tissue with the need for surgical reintervention in 29% of the cases, besides being visually identifiable.29,30 In this service, the autors chose to make an appendicostomy with an appendix in situ and without use of devices, a technique of choice in the literature.10
In a recently published meta-analysis, Li et al. compared appendicostomy to cecostomy, concluding that the two techniques have similar percentages of incontinence control; the appendicostomy for antegrade irrigation, however, is more often associated with complications requiring surgical reintervention.31
The importance of the colonic transit study in the management and prognosis of refractory functional constipation has not been well defined. Associations of the transit pattern with the type of RFC clinical manifestation have been identified.13,16 Pioneering researchers using this investigation tool have found that patients with a slow colonic transit pattern do not respond to retrograde enemas and may have neurotransmitter deficiencies.16 Data from the pediatric population are not yet sufficient to define surgical indications based on this type of investigation. In the present series, the transit pattern was not a sufficiently adequate tool for patient selection regarding the therapeutic method.
Despite the high frequency of complications and the absence of validated quality of life assessment, the procedure appears to have been accepted by the relatives and patients of the present series, since the majority persisted using the route after the successful management of complications. Benefits of the appendicostomy in the quality of life of children with RFC have been described as significant.32
Appendicostomy, despite the high number of complications, appears to control the fecal leaking resulting from intestinal constipation more frequently and earlier than isolated clinical management. The recovery of the spontaneous evacuation condition was observed in a larger number of patients when compared with the isolated clinical management; however, the small number of participants and the conclusions based on risk estimation suggest that studies with larger samples and longer follow-up periods should be carried out to allow a more definitive assessment of this outcome. For now, it seems possible to suggest that the information of surgical possibility should be provided to the cases selected by systematic evaluation and that the final decision on the choice of any of the methods should be made by the family, after being appropriately informed about the risks and benefits of each alternative.
FundingFAEPEX - Fundo de Apoio ao Ensino, à Pesquisa e à Extensão da Unicamp, Project No. 149/09.
Conflicts of interestThe authors declare no conflicts of interest.
Please cite this article as: Arruda VP, Bellomo-Brandão MA, Bustorff-Silva JM, Lomazi EA. Refractory functional constipation: clinical management or appendicostomy? J Pediatr (Rio J). 2020;96:210–6.
Study carried out at Faculdade de Ciências Médicas, Universidade Estadual de Campinas (Unicamp), Campinas, SP, Brazil.