To assess the prevalence of metabolic syndrome-like symptoms in a population of preterm infants with very low birth weight (<1500g) at 2 years of corrected age and identify the occurrence of associated risk factors.
MethodsCross-sectional study during a five-year period, including preterm infants born with very low birth weight evaluated at 2 years of corrected age. Metabolic syndrome-like symptoms was defined by the presence of three or more of these criteria: abdominal circumference≥90th percentile, fasting blood glucose≥100mg/dL, triglycerides≥110mg/dL, HDL cholesterol≤40mg/dL, and blood pressure≥90th percentile.
ResultsA total of 214 preterm infants with birth weight<1500g were evaluated. The prevalence of metabolic syndrome-like symptoms at 2 years of corrected age was 15.1%. Arterial hypertension was present in 57.5%, HDL≤40mg/dL in 29.2%, hypertriglyceridemia in 22.6%, and abdominal circumference above the 90th percentile in 18.8%. Only 3.7% had hyperglycemia. The presence of periventricular leukomalacia was an independent risk factor for arterial hypertension at this age (OR 2.34, 95% CI: 0.079–0.69, p=0.008). Overweight and obesity at 2 years of corrected age were independently associated with metabolic syndrome-like symptoms (OR 2.75, 95% CI: 1.19–6.36, p=0.018).
ConclusionMetabolic syndrome-like symptoms can be observed in very low birth weight preterm infants as early as 2 years of corrected age. Overweight and early-onset obesity are significant risk factors for metabolic syndrome-like symptoms, which deserves appropriate intervention for this high-risk population.
Avaliar a prevalência de síndrome metabólica “like” em população de crianças prematuras com muito baixo peso de nascimento (< 1500 gramas) aos dois anos de idade corrigida e identificar a ocorrência de fatores de risco associados.
MétodosEstudo transversal que incluiu prematuros nascidos durante um período de cinco anos com muito baixo peso de nascimento, avaliados aos dois anos de idade corrigida. A síndrome metabólica “like” foi definida pela presença de três ou mais desses critérios: circunferência abdominal ≥ percentil 90, glicemia de jejum ≥ 100mg/dL, triglicerídeos ≥ 110mg/dL, colesterol HDL ≤ 40mg/dL e pressão arterial ≥ percentil 90.
ResultadosForam avaliados 214 prematuros abaixo de 1.500 gramas. A prevalência de síndrome metabólica “like” aos dois anos de idade corrigida foi 15,1%. Hipertensão arterial esteve presente em 57,5%, HDL ≤ 40mg/dL em 29,2%, hipertrigliceridemia em 22,6% e aumento da circunferência abdominal acima do percentil 90 em 18,8%. Apenas 3,7% apresentaram hiperglicemia. A presença de leucomalácia periventricular foi um fator de risco independente para hipertensão arterial nessa idade (OR 2,34; IC 95% 0,079-0,69; p=0,008); sobrepeso e obesidade aos dois anos de idade corrigida foram independentemente associados com síndrome metabólica “like” (OR 2,75, IC 95% CI 1,19-6,36; p=0,018).
ConclusãoSíndrome metabólica “like” ocorre em prematuros de muito baixo peso tão precocemente quanto dois anos de idade corrigida. Sobrepeso e obesidade de início precoce são fatores de risco significativos para síndrome metabólica “like”, merecem intervenção apropriada para essa população de alto risco.
Metabolic syndrome prevention is a challenge in the pediatric clinical practice, as it is associated with a 1.5-fold increase in overall mortality and 2.5-fold increase in mortality due to cardiovascular diseases.1,2 In adults, metabolic syndrome is defined as a group of findings including obesity, insulin resistance, dyslipidemia and arterial hypertension associated with the subsequent development of cardiovascular disease and diabetes.2 However, there is a lack of reference values for its components in children, which makes it difficult to diagnose metabolic syndrome in the pediatric age range. Therefore, arbitrary cutoff points, adapted from standard curves for anthropometry assessment and for diagnosis of hypertension to laboratory abnormalities are alternatively used.3
In young adults born at less than 34 weeks gestation, the cardiometabolic risk was 3.7-fold higher and, in late preterm infants, 2.5-fold higher when compared with those born at term.4 Singhal et al. investigated the effects of promoting early growth on the body composition in two studies with children born small for gestational age (SGA; weight below the 10th percentile in study 1 and below the 20th percentile in study 2) and found a causal association between quick and early weight gain and risk for obesity later in life.5 Preterm children appear to present a metabolic profile similar to that found in SGA children born at term, possibly with similar risk for metabolic syndrome.5,6
Several studies have suggested an association between low birth weight and metabolic syndrome in adulthood, but there is a scarcity of data evaluating preterm birth. Studies with adolescents and adults born prematurely evaluated isolated components of metabolic syndrome, such as obesity, arterial hypertension or insulin resistance.6–12
Because the prevalence of metabolic syndrome in the early stages of life of very low birth weight (VLBW) preterm infants remains unknown, the present study aimed to evaluate the prevalence of metabolic syndrome-like symptoms (MSL) in VLBW preterm infants at 2 years of corrected age (CA) and to identify its association with perinatal and/or neonatal risk factors.
MethodsThis was a cross-sectional study including VLBW preterm infants (birth weight 500–1499g) born during a five-year period in a tertiary public university hospital who were discharged from the neonatology unit to the institution's follow-up outpatient clinic. The exclusion criteria were: presence of major congenital malformations and inborn errors of metabolism or chromosomal abnormalities, congenital syphilis, toxoplasmosis, rubella, cytomegalovirus, herpes (STORCH) and HIV infections; attending fewer than three medical consultations at follow-up after discharge; or death in the period. At the time of hospital discharge, the protocol form of eligible patients had been filled out with all the perinatal data and neonatal hospitalization morbidities by one of the researchers.
Outcomes and variablesThe maternal variables assessed were type of delivery, use of antenatal corticosteroids, maternal pre-eclampsia, chorioamnionitis (presence of clinical signs such as maternal fever, uterine hypertonia, purulent or foul-smelling amniotic fluid, maternal leukocytosis, or fetal tachycardia) or presence of urinary infection (positive urine culture). Family income (in Brazilian minimum wages, which were converted to dollars at the time of data collection, due to fluctuations in the purchasing power of the country's currency) and the primary caregiver's level of schooling were also analyzed.
The neonatal variables assessed were birth weight, gestational age (determined by the best obstetric estimate with early obstetrical ultrasound and/or date of last menstrual period and confirmed by the pediatric physical examination immediately after birth), being SGA (below the 10th percentile), use of amino acids in the first 24h of life, presence of bronchopulmonary dysplasia (need for supplemental oxygen at 36 weeks postconceptional age), and presence of periventricular leukomalacia (diagnosed by brain ultrasonography and confirmed by magnetic resonance imaging of the brain).13
The outpatient follow-up evaluation was carried out according to the routine ambulatory care of the institution: once a month up to 6 months of CA, every two months between 7 and 12 months of CA and every three months thereafter, until 24 months of CA. Rates of exclusive breastfeeding and supplemented breastfeeding at 6 months of CA were prospectively recorded, as well as anthropometric measures according to CA and gender, using the World Health Organization curves, including the calculated body mass index (BMI) for each child at 2 years of CA, which defined overweight/obesity, when above the percentile of each reference age.
All blood samples were collected by venipuncture, after 12h of fasting, as part of the routine ambulatory follow-up, which performs examinations at 24 months of CA. The laboratory tests included measurements of triglycerides (mg/dL), HDL (mg/dL), and glucose (mg/dL) levels.
Blood pressure (BP) was measured on the right upper limb, using a cuff of adequate size and with the child in the sitting position. After at least half an hour of consultation, and when the child was already calmer and playing, he/she was placed in the sitting position for BP measurement; three measurements were obtained, and their mean was used in the analysis (mmHg), being compared with table values according to height percentile, age, and gender, according to guidelines of the National High Blood Pressure Education Program (NHBPEP) Working Group.14 BP was measured noninvasively using the automatic oscillometric monitor with a variation of 10–200mmHg and resolution of 1mmHg (Mindray VS-800, Seattle, WA, USA).
The abdominal circumference (AC) was measured in duplicate using an inelastic measuring tape (in cm) placed at the umbilical scar level, at the end of the child's expiration, and in the orthostatic position. AC was measured on all children at 2 years of CA and the cutoff value greater than or equal to the 90th percentile for gender and age was considered altered.15
To determine the prevalence of metabolic syndrome in the study population, the authors proposed a new term for this study: MSL. MSL was defined as the presence of an alteration in three or more of the following criteria proposed by Cook et al.: AC≥90th percentile, fasting glucose≥110mg/dL, triglycerides≥110mg/dL, HDL cholesterol≤40mg/dL, and blood pressure≥90th percentile.16 In 2003, the American Diabetes Association reduced the glucose value to ≥100mg/dL, which was adopted in the present study.17
Ethical considerationsThe study project and the data collection protocol were approved by the Ethics and Research Committee.
Statistical analysisThe sample size was calculated based on two previous complementary studies; the first with children born at term and estimated for the degree of prematurity, and the second a meta-analysis that allowed the calculation of the prevalence in adulthood related to the lowest birth weight, thus allowing us to assume an estimated prevalence of MSL of 9% in children born with VLBW and estimating to detect 0.5 standard deviation in these children when compared with preterm infants at 2 years of CAage.7,18 A total number of 126 children was estimated as necessary for a confidence level of 95% and a 5% error.
The statistical program SPSS (PASW Statistics for Windows, Version 18.0. Chicago, USA) was used. Frequencies and percentages were used for categorical variables; mean and standard deviation (mean±SD) or median and interquartile range (P25-P75). The Mann–Whitney, chi-squared, and Student's t-tests were used in the statistical analysis, as well as multiple linear regression (relative risk [RR] and confidence interval calculation), and the Hosmer–Lemeshow test. The variables with clinical relevance and p<0.20 in the univariate analysis were included in the multiple linear regression model: maternal chorioamnionitis or maternal urinary infection, periventricular leukomalacia, supplemented breastfeeding (mixed feeding) at 6 months of CA, weight at 2 years of CA, and overweight or obesity at 2 years of CA. The model was adjusted for birth weight, gestational age, and gender. The level of significance was set at p<0.05.
ResultsDuring the study period, 446 preterm infants weighing≤1500g were born at the institution, of whom 133 (29.8%) died during admission to the neonatal intensive care unit and 78 (24.9%) were excluded: six due to major congenital malformations, two due to inborn errors of metabolism, five due to congenital toxoplasmosis, two due to cytomegalovirus, one with herpes, seven due to syphilis, six due to HIV-positive; 10 deaths were observed during outpatient follow-up, and 39 other children attended fewer than three consultations. Thus, 235 patients with VLBW were considered eligible for outpatient follow-up, of whom 23 did not have data to define MSL (9.4%).
A total of 212 (90.6%) of the VLBW eligible preterm infants were studied, whose mean birth weight was 1170±232g and gestational age of 30±2 weeks; 48.9% of the children were SGA. Considering the presence of alterations in three or more of the criteria proposed by Cook et al., the prevalence of MSL was 15.1% (32 children); 51 children had two altered criteria (21.7%), 80 had only one altered criterion (34%), and 49 children (20.8%) did not meet any of the proposed criteria for the diagnosis of MSL.
Presence of arterial hypertension was the most frequent isolated finding in 57.5% of the study population, followed by low levels of HDL (29.2%) and hypertriglyceridemia (22.6%). The diagnosis of abdominal obesity occurred in 18.8% and only 3.7% of the population had hyperglycemia.
The individualized analysis of each MSL criterion adjusted for birth weight and gestational age showed that the presence of periventricular leukomalacia was an independent risk factor for arterial hypertension (OR 2.34, 95% CI: 0.079–0.69, p=0.008); bronchopulmonary dysplasia was an independent risk factor for HDL cholesterol levels≤40mg/dL (OR 2.1, 95% CI: 0.063–0.7, p=0.012); male gender and VLBW (OR 2.48, 95% CI: 1.09–5.61, p=0.03) were independent risk factors for the presence of hypertriglyceridemia (triglycerides≥110mg/dL). The presence of periventricular leukomalacia (OR 1.9, 95% CI: 1.59–6.12, p=0.05), overweight or obesity (OR 2.3, 95% CI: 1.38–2.95, p=0.04), and low maternal level of schooling (OR 1.27, 95% CI: 1.062–1.56, p=0.09) were independent risk factors for AC≥90th percentile. Despite the small number of children with hyperglycemia, overweight or obesity was an independently associated risk factor (OR 2.4, 95% CI: 1.22–4.84, p=0.04), and being born adequate for gestational age had a protective effect against fasting glycemia≥100mg/dL (OR 0.063, 95% CI: 0.003–0.82; p=0.046).
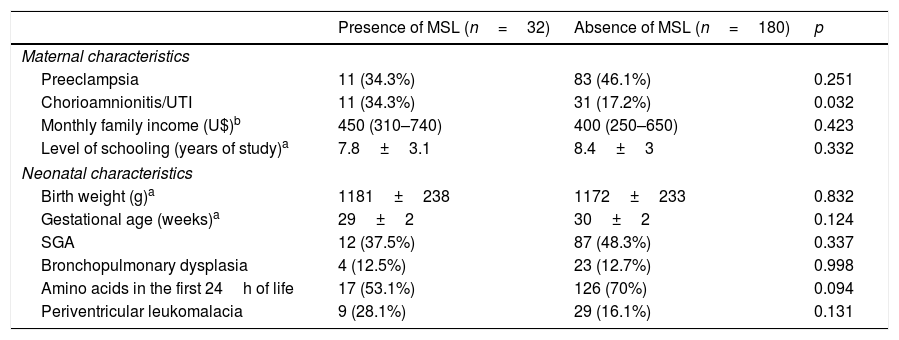
The characteristics of VLBW infants who were associated with MSL are described in Tables 1 and 2. Overweight or obese children at 2 years of CA and those whose mothers had clinical chorioamnionitis or urinary tract infection during pregnancy showed a higher prevalence of MSL in the univariate analysis. This association was not observed in relation to the feeding pattern at 6 months of CA, weight and length at 2 years of CA (Table 2).
Perinatal characteristics of very low birth weight preterm infants according to the presence of metabolic syndrome-like symptoms (MSL).
| Presence of MSL (n=32) | Absence of MSL (n=180) | p | |
|---|---|---|---|
| Maternal characteristics | |||
| Preeclampsia | 11 (34.3%) | 83 (46.1%) | 0.251 |
| Chorioamnionitis/UTI | 11 (34.3%) | 31 (17.2%) | 0.032 |
| Monthly family income (U$)b | 450 (310–740) | 400 (250–650) | 0.423 |
| Level of schooling (years of study)a | 7.8±3.1 | 8.4±3 | 0.332 |
| Neonatal characteristics | |||
| Birth weight (g)a | 1181±238 | 1172±233 | 0.832 |
| Gestational age (weeks)a | 29±2 | 30±2 | 0.124 |
| SGA | 12 (37.5%) | 87 (48.3%) | 0.337 |
| Bronchopulmonary dysplasia | 4 (12.5%) | 23 (12.7%) | 0.998 |
| Amino acids in the first 24h of life | 17 (53.1%) | 126 (70%) | 0.094 |
| Periventricular leukomalacia | 9 (28.1%) | 29 (16.1%) | 0.131 |
UTI, urinary tract infection; SGA, small for gestational age.
Data shown as mean±standard deviation a or median (interquartile range).
Follow-up of very low birth weight preterm infants at 2 years of corrected age (CA).
| Presence of MSL (n=32) | Absence of MSL (n=180) | p | |
|---|---|---|---|
| Exclusive breastfeedinga at 6 months of CA | 4 (12.5%) | 27 (15%) | 0.631 |
| Mixed feedingb at 6 months of CA | 16 (50%) | 72 (40%) | 0.395 |
| Weight at 2 years of CAc (g) | 11,850 (±2411) | 11,190 (±1502) | 0.144 |
| Length at 2 years of CAc (cm) | 84.9 (±0.7) | 84.4 (±0.2) | 0.501 |
| Overweight/obesity at 2 years of CA | 12 (37.5%) | 29 (16.1%) | 0.005 |
The presence of overweight or obesity at 2 years of CA was an independent risk factor for MSL, with a 2.75-fold higher risk of MSL in VLBW preterm infants (Table 3).
Factors associated with the early presence of metabolic syndrome-like symptoms in very low birth weight preterm infants.
| Factors | OR | 95% CI | p-Value |
|---|---|---|---|
| Maternal chorioamnionitis or maternal urinary tract infectiona | 1.91 | (0.79; 4.56) | 0.146 |
| Periventricular leukomalaciaa | 2.42 | (0.92; 6.36) | 0.074 |
| Mixed feeding at 6 months of CAa | 0.75 | (0.32; 1.74) | 0.194 |
| Weight at 2 years of CA (g)a | 1.0 | (1.0; 1.0) | 0.872 |
| Overweight/obesity at 2 years of CAa | 2.75 | (1.19; 6.36) | 0.018 |
OR, odds ratio; 95% CI, 95% confidence interval; CA, corrected age.
Logistic regression analysis.
In the present study, MSL is a proposed new term that includes a warning regarding risk predictors that are found in a continuous scale and interact in a complex way in metabolic syndrome in childhood and, consequently, its own definition in early childhood is a challenge, especially in the population of preterm infants with VLBW. The present study found a prevalence of 15.1% at 2 years of CA in preterm VLBW infants, a four-fold higher prevalence than that found in a study carried out with children aged 6–16 years, in which the prevalence of metabolic syndrome was only 3.6%,18 and in another sample of adolescents from the United States, which found a prevalence of 4.2% using the same criteria used in the present study.19
Metabolic syndrome tends to be more prevalent among children at higher risk, such as preterm infants and/or those with intrauterine growth restriction.20 It is pertinent to seek to determine the prevalence of this condition in the early stages in life. The association between low birth weight or early obesity and later coronary disease, diabetes, or diagnosis of metabolic syndrome has been demonstrated in previous studies, in addition to the association of these factors with isolated metabolic abnormalities.20,21 However, until the present study, the early identification of the individual components of metabolic syndrome and their association with perinatal and postnatal risk factors had been little explored. A systematic review and meta-analysis of 27 studies including preterm infants evaluated outcomes associated with metabolic syndrome, but not all individual components of metabolic syndrome at an early age were considered, as outlined in the present study.22
Metabolic syndrome is not a disease, but a cluster of metabolic disorders and thus, to date, it has not yet been possible to determine whether it is associated with a poorer prognosis than its components individually. In the present study, 76.9% of the preterm infants had at least one altered individual component among those that characterize metabolic syndrome; arterial hypertension was the most frequent alteration (57.5%), followed by low HDL (29.2%) and hypertriglyceridemia (22.6%), which may have overestimated the diagnosis of MSL, even considering that, when these components were assessed individually, studies found comparable results, especially taking into account the fact that they were not designed for this population of VLBW preterm infants. Moreover, in a cohort of male patients with a long-term follow-up, the presence of metabolic syndrome was not able to predict cardiovascular mortality better than the sum of its individual components, reinforcing the importance of this individual evaluation.6,23,24
The prevalence of MSL in infants born SGA and adequate for gestational age was similar. Preterm birth, regardless of intrauterine growth restriction, is a risk factor for hypertension and dyslipidemia, the two most prevalent individual components in this study.6 Preterm children born SGA do not necessarily undergo the same issues as those born SGA without being preterm; being born with intrauterine growth restriction reflects adverse exposure in utero, whereas preterm birth is multifactorial and involves environmental and extrauterine insults, in addition to adverse intrauterine exposure. Both conditions, preterm birth or nutrient deprivation in a critical intrauterine period, promote epigenetic changes resulting in dyslipidemia, obesity, and insulin resistance, the main components of metabolic syndrome.4,12,20,21
The presence of overweight/obesity at 2 years of CA was independently associated with MSL. This is a very significant finding for prevention, because as the prevalence of childhood obesity increases, so will that of MSL. Childhood obesity is currently associated with a higher occurrence of metabolic syndrome, reinforcing the importance of an additional metabolic programming starting at the preterm birth.3,4,20,25 In preterm infants weighing less than 1000g, the prevalence of hypertension and obesity at 3 years of age was high, and obesity was more prevalent among hypertensive children.26 Thus, there is a previously described association between arterial hypertension and overweight/obesity for this population.
The high prevalence of overweight/obesity can be explained in part by the fact that insulin resistance is the common pathophysiological mechanism between metabolic syndrome and obesity in Brazilian children and adolescents.27 A previous study with Brazilian school-age children born at term reported a metabolic syndrome rate of only 0.3%, in normal-weight children, 10.7% in those overweight, and 34.5% in those who were obese.18 The present findings are similar to those of the NHANES study, which showed a prevalence of MS of 14.5% in overweight adolescents and only 0.9% when weight was normal.28
The association between the presence of hypertension in childhood or adolescence and prematurity has been previously described: at 16 years of age, 13.5% of adolescents whose birth weight ranged from 600g to 1250g were already hypertensive, when compared to only 4% of adolescents born with adequate weight and at term.10 Hack et al. found higher rates of arterial hypertension in VLBW preterm infants, when compared to their peers born at term in adulthood.11 In a cohort of extremely preterm infants (gestational age<28 weeks), systolic and diastolic pressures were significantly higher than in controls, with this result persisting from 8 to 18 years of age; similarly to the present study, these authors found an association with overweight at 2 years of age.29 Posod et al. found systolic and diastolic hypertension, hypercholesterolemia, fasting hyperglycemia, and insulin resistance indicators in children born preterm, allowing the conclusion that premature infants have a cardiovascular disease risk as early as school age.30
The presence of periventricular leukomalacia was an independent risk factor for arterial hypertension, reinforcing the known hypothesis that experiences involving a fetal inflammatory response and intrauterine stress such as maternal chorioamnionitis and/or maternal infection involved in the pathogenesis of leukomalacia are present very early in life and progress into the postnatal period in a sustained manner with a set of morbidities, including periventricular leukomalacia; these have lasting consequences for the preterm infant, such as arterial hypertension in childhood, adolescence or adulthood.20,31,32
Despite the importance of the present data, some limitations must be considered, particularly regarding the early diagnosis of metabolic syndrome in preterm infants, since there is no reference in the literature that supports this diagnosis as early as at 2 years of CA.20 For this reason, the authors developed a definition for MSL based on practical, accessible, and reproducible criteria used for adults and adolescents, since the diagnosis of metabolic syndrome remains controversial.3,24 Despite the presence of several combinations of the five components to define metabolic syndrome in adults, it is a different matter in children, as the presence of obesity was the main component.23,32 The present study did not assess insulin resistance, a detectable abnormality in individuals with low weight. However, the glucose levels recommended by the American Diabetes Association were used, because they are easily applicable and reproducible in studies and clinical practice.14,17,33
Eating habits and the family feeding history, two important factors related to the metabolic profile, could not be adequately assessed after 6 months of CA due to limitations of food recall questionnaires used with mothers or caregivers after 6 months of age, as well as in a previous cohort that evaluated the blood pressure of very low weight preterm infants without correlating it with food consumption.11 Nevertheless, the sample was homogeneous for these factors. All caregivers received the same dietary guidance (including guidance on the time of introduction of complementary foods) and the children received the same follow-up and nutritional guidance according to the routine outpatient practice of this institution, which is why attending fewer than three follow-up consultations after discharge was one of the exclusion criteria.
It was possible to reliably establish the diagnosis and prevalence of MSL in VLBW preterm infants as early as at 2 years of CA, being already elevated at this age. The same applied to its individual components, of which arterial hypertension was the most prevalent in VLBW preterm infants at 2 years of age. The presence of overweight and obesity at 2 years of CA are important early warning signs for the early presence of MSL and should be continuously prevented.
FundingThe study received financial support from FIPE-HCPA and the Postgraduate Program in Child and Adolescent Health, UFRGS.
Conflicts of interestThe authors declare no conflicts of interest.
To Vania Naomi Hirakata and Luciano S P Guimarães for their assistance with statistical analysis; to the multiprofessional team from the Ambulatory of Neonatology of HCPA; to the patients and their families; and to Fundo de Incentivo à Pesquisa (FIPE), for the financial support.
Please cite this article as: Heidemann LA, Procianoy RS, Silveira RC. Prevalence of metabolic syndrome-like at 2 years of corrected age in very low birth weight preterm infants and associated factors. J Pediatr (Rio J). 2019;95:291–7.