To evaluate the efficiency of the sepsis risk calculator and the serial clinical observation in the management of late preterm and term newborns with infectious risk factors.
MethodSingle-center, observational, two-phase cohort study comparing the rates of neonates born ≥35 weeks' gestation, ≥2000 g birthweight, and without major congenital anomalies, who were screened and/or received antibiotics for early-onset neonatal sepsis risk at our center during two periods, before (January/2018–June/2019) and after (July/2019–December/2020) the implementation of the sepsis risk calculator.
ResultsA total of 1796 (Period 1) and 1867 (Period 2) patients with infectious risk factors were included. During the second period, tests to rule out sepsis were reduced by 34.0 % (RR, 95 %CI): 0.66 (0.61, 0.71), blood cultures by 13.1 %: 0.87 (0.77, 0.98), hospital admissions by 13.5 %: 0.86 (0.76, 0.98) and antibiotic administration by 45.9 %: 0.54 (0.47, 0.63). Three cases of early-onset neonatal sepsis occurred in the first period and two in the second. Clinical serial evaluation would have detected all true cases.
ConclusionsThe implementation of a sepsis risk calculator in the management of newborns ≥35 weeks GA, ≥2000 g birthweight, without major congenital anomalies, with infectious risk factors is safe and adequate to reduce laboratory tests, blood cultures, hospital admissions, and antibiotics administration. Serial clinical observation, in addition, could be instrumental to achieve or even improve this goal.
Early-onset neonatal sepsis (EONS) is a life-threatening condition in the immediate neonatal period, defined as bacteremia or bacterial meningitis occurring within the first 72 h after birth. Commonly, a positive culture is considered a case of EONS when a known pathogenic species is isolated, the patient exhibits compatible symptoms, and receives an appropriate course of antibiotics. Traditional infectious risk factors (IRF) are maternal vagino-rectal colonization by group B streptococci (GBS), prolonged rupture of membranes (≥ 18 h), intrapartum maternal fever, and maternal chorioamnionitis (MCA). MCA has received special attention since it affects 0.1–2 % of all pregnancies, its incidence is higher at lower gestational ages (GA) and increases the risk of EONS.1,2 However, over the past two decades, as the use of intrapartum antibiotics has increased, a decrease in the incidence of EONS has been observed, even among infants born to mothers with chorioamnionitis.3 On the other hand, several studies indicate that the use of antibiotics in most newborns is probably unnecessary and could even be harmful. Broad-spectrum antibiotics can alter the patient's microbiota with long-term consequences.4,5 Furthermore, admission of newborns for administration of antibiotics could interfere with breastfeeding and the establishment of the mother-infant bond and increase the risk of errors related to healthcare or medication side effects.6,7 In addition, other studies with large populations of term or near-term infants with IRF who were asymptomatic showed that the risk of developing EONS was extremely low.8 Based on all this evidence, current guidelines for the management of at-risk patients have been updated.9,10 Although the assessment and management of infants with IRF is common in clinical practice, it remains a great challenge to identify which babies are at high risk of EONS and, therefore, to establish the indication to initiate early broad-spectrum antibiotic therapy.11 As a consequence, there is still a wide variability in clinical practice.12
A body of evidence has accumulated to date showing that the use of a multivariate risk assessment, such as the Northern California Kaiser-Permanente Neonatal Sepsis Risk Calculator (SRC)13 is associated with a significant reduction in the use of empirical antibiotics in neonates with IRF. However, the available evidence regarding its safety is limited, so more studies are needed, especially outside the US, where the SRC was initially developed.14 In our center, a previous retrospective study seeking to optimize the use of antibiotics in newborns ≥35 weeks GA showed that the use of the SRC would have decreased significantly the number of infectious workups and hospital admissions of newborns with IRF, as well as the proportion of infants receiving antibiotics.15 The objective of the present study was to prospectively evaluate the efficiency of the implementation of the SRC in our setting and to know if a management based on clinical observation would have been equally safe and efficient. The authors hypothesized that the implementation of the SRC would reduce laboratory draws, hospital admissions, and the administration of broad-spectrum antibiotics to neonates ≥35 weeks GA with IRF and that the management based on clinical observation would not be inferior.
MethodsThe authors carried out a systematic, observational, two-phase cohort study, of the management of newborns ≥35 weeks GA with IRF, born in our maternity hospital comparing two 18-month periods. During Period 1 (January 1, 2018 to June 30, 2019), before the introduction of the SRC, management criteria were based on local protocols, mostly derived from international recommendations.15 During the second Period (July 1, 2019 to December 31, 2020), after the introduction of the SRC, an internal management protocol for newborns with IRF was developed based on the SRC. All patients with at least one IRF were evaluated using the protocol, although the final management decision rested with the attending neonatologist. The main outcomes were the proportion of analytical studies, blood cultures, hospital admissions, and antibiotic administration before and after the implementation of the SRC. Maternal and neonatal medical records were reviewed, collecting sociodemographic and clinical data on a predesigned collection form. Outborn patients and newborns with major congenital anomalies were excluded. Newborns weighing less than 2000 g were also excluded since they are admitted regardless of the IRF. The IRFs considered for this study were: positive maternal GBS-vagino-rectal exudate, intrapartum maternal fever ≥38°C, prelabor rupture of membranes ≥18 h, and MCA, as registered by the attending obstetrician in the maternal clinical record. In the studied center, during the period 2017–2019 (3 years), the incidence of early neonatal sepsis was 0.39‰. However, to calculate the risk using the SRC, the slightly more conservative value of 0.5‰ was used. Infants with MCA were considered directly as medium risk and managed accordingly. For these patients, given the increased risk of EONS, a pre-test risk of 4‰ has been recommended.16
Statistical analysisData were collected in an Excel spreadsheet and statistical analysis was performed with the statistical software SPSS v.25 (IBM Corp, Armonk, NY, USA). Continuous variables with a normal distribution are expressed as mean and standard deviation (SD) and the differences between groups were studied using the Student's t test. Continuous variables not normally distributed are presented as the median and interquartile range (IQR) and were compared using the Mann–Whitney U test. Qualitative variables are expressed as proportions (%) and were analyzed using the Chi-square test or Fisher's exact test, as appropriate. A bivariate analysis of patient characteristics, perinatal interventions, and outcomes was performed. The Risk Ratio (RR) for the main outcomes was calculated by Poisson regression with robust estimation of the variance and is expressed as RR and 95 % confidence intervals (95 % CI). All statistical tests were two-tail performed, and statistical significance was set at p-value <0.05.
This project was approved by the Centre's Research Ethics Committee (Registration Number: 2021-238-1). Since this is an observational study with anonymized data from medical records as part of the quality control of care activity and of adherence to the unit protocols, signing a written informed consent form was not deemed necessary.
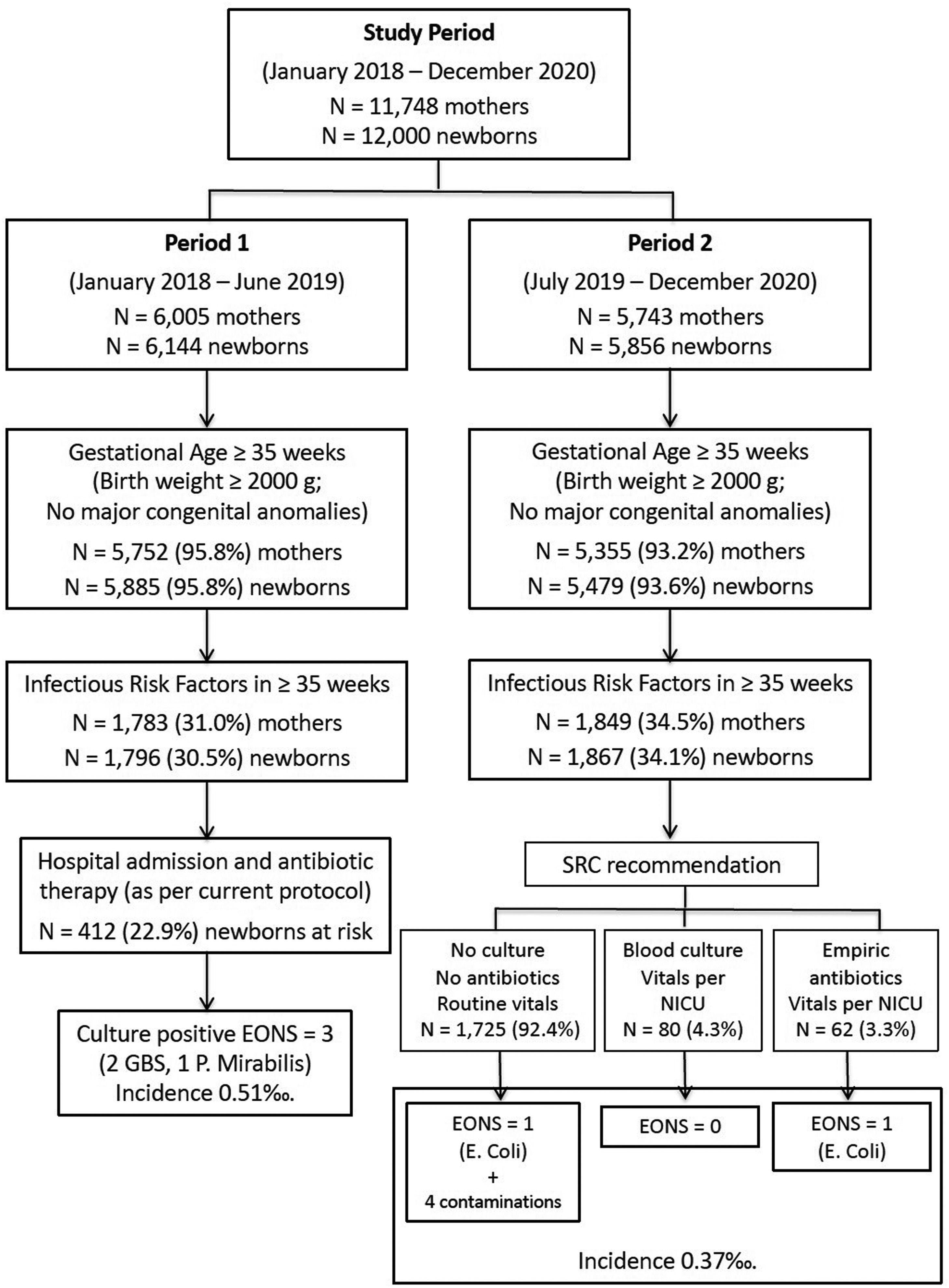
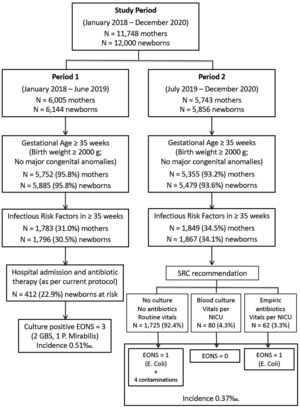
ResultsFigure 1 shows the number of deliveries and newborns assisted in the maternity hospital during the study periods, the management recommendations according to the current protocols, and the incidence of EONS in each period. The results of Period 1 have been published in detail previously.15 For the present study a total of 1796 patients ≥35 weeks GA with at least one IRF (Period 1) are compared to 1867 newborns (Period 2) with similar characteristics.
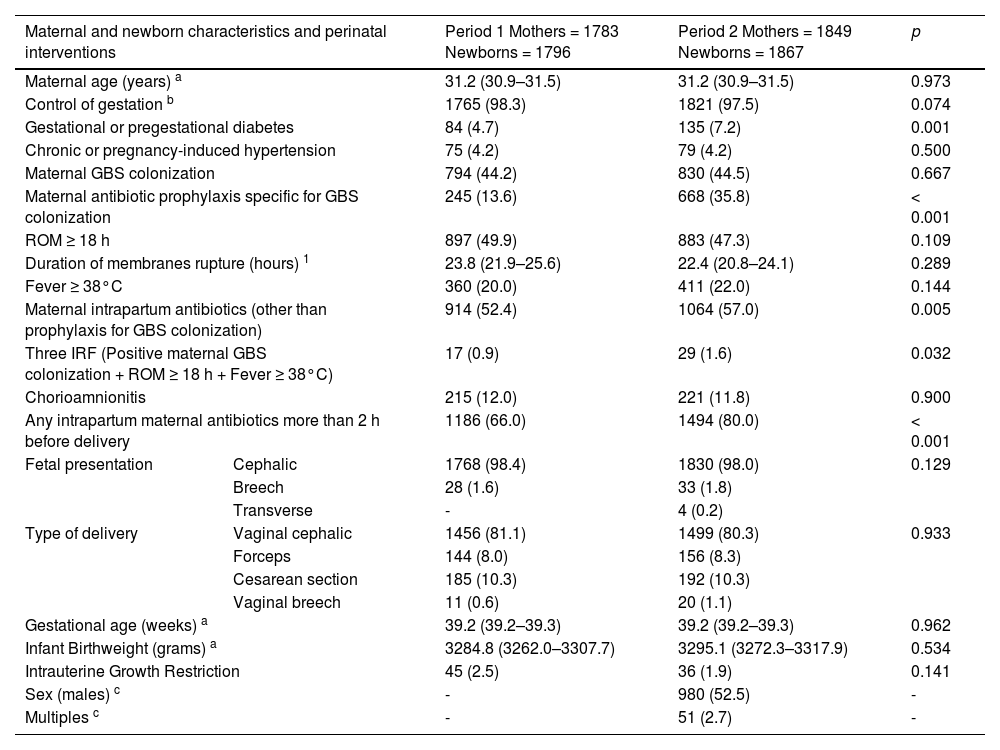
The demographic and clinical characteristics of the mothers and newborns with IRF in the two study periods are summarized in Table 1. The maternal age distribution and the proportion of pregnancy control, at least one prenatal visit to an obstetrical team, was similar in the two groups. Hypertensive states of pregnancy were similar in both groups, but the diagnoses of pregestational or gestational diabetes were higher during the second period. The distribution of IRF was similar in both periods, although the association of at least three of them was more frequent during the second period. The use of both, broad-spectrum antibiotics and specific antibiotics against GBS, was higher during the second period. There were no differences in mode of presentation, type of delivery or infants’ GA or birthweight between periods.
Demographic and clinical characteristics of mothers and newborns from two periods of study: Period 1 (January/2018 to June/2019) and Period 2 (July/2019 to December/2020), before and after the implementation of the Kaiser-Permanente Neonatal Sepsis Risk Calculator.
| Maternal and newborn characteristics and perinatal interventions | Period 1 Mothers = 1783 Newborns = 1796 | Period 2 Mothers = 1849 Newborns = 1867 | p | |
|---|---|---|---|---|
| Maternal age (years) a | 31.2 (30.9–31.5) | 31.2 (30.9–31.5) | 0.973 | |
| Control of gestation b | 1765 (98.3) | 1821 (97.5) | 0.074 | |
| Gestational or pregestational diabetes | 84 (4.7) | 135 (7.2) | 0.001 | |
| Chronic or pregnancy-induced hypertension | 75 (4.2) | 79 (4.2) | 0.500 | |
| Maternal GBS colonization | 794 (44.2) | 830 (44.5) | 0.667 | |
| Maternal antibiotic prophylaxis specific for GBS colonization | 245 (13.6) | 668 (35.8) | < 0.001 | |
| ROM ≥ 18 h | 897 (49.9) | 883 (47.3) | 0.109 | |
| Duration of membranes rupture (hours) 1 | 23.8 (21.9–25.6) | 22.4 (20.8–24.1) | 0.289 | |
| Fever ≥ 38°C | 360 (20.0) | 411 (22.0) | 0.144 | |
| Maternal intrapartum antibiotics (other than prophylaxis for GBS colonization) | 914 (52.4) | 1064 (57.0) | 0.005 | |
| Three IRF (Positive maternal GBS colonization + ROM ≥ 18 h + Fever ≥ 38°C) | 17 (0.9) | 29 (1.6) | 0.032 | |
| Chorioamnionitis | 215 (12.0) | 221 (11.8) | 0.900 | |
| Any intrapartum maternal antibiotics more than 2 h before delivery | 1186 (66.0) | 1494 (80.0) | < 0.001 | |
| Fetal presentation | Cephalic | 1768 (98.4) | 1830 (98.0) | 0.129 |
| Breech | 28 (1.6) | 33 (1.8) | ||
| Transverse | - | 4 (0.2) | ||
| Type of delivery | Vaginal cephalic | 1456 (81.1) | 1499 (80.3) | 0.933 |
| Forceps | 144 (8.0) | 156 (8.3) | ||
| Cesarean section | 185 (10.3) | 192 (10.3) | ||
| Vaginal breech | 11 (0.6) | 20 (1.1) | ||
| Gestational age (weeks) a | 39.2 (39.2–39.3) | 39.2 (39.2–39.3) | 0.962 | |
| Infant Birthweight (grams) a | 3284.8 (3262.0–3307.7) | 3295.1 (3272.3–3317.9) | 0.534 | |
| Intrauterine Growth Restriction | 45 (2.5) | 36 (1.9) | 0.141 | |
| Sex (males) c | - | 980 (52.5) | - | |
| Multiples c | - | 51 (2.7) | - | |
All values are n (%), except when otherwise specified. IRF, Infectious Risk Factors. ROM, Rupture of membranes.
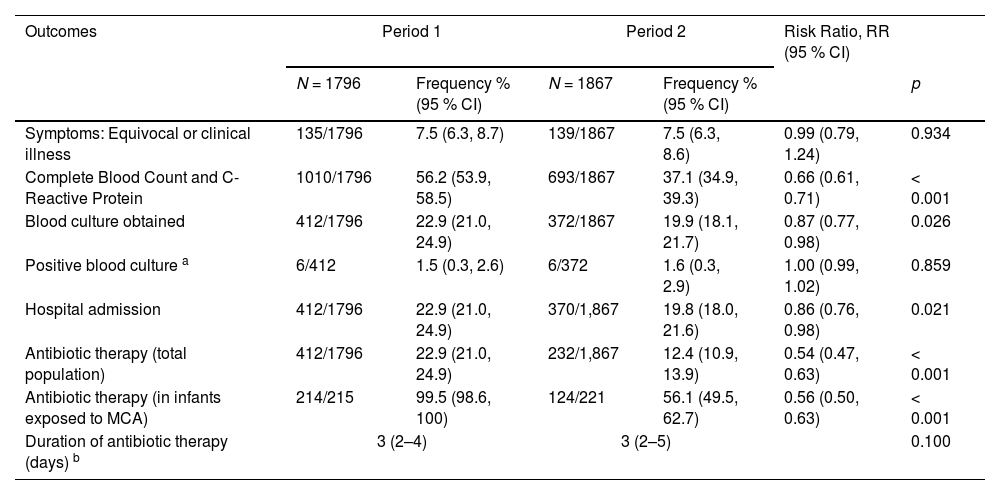
Table 2 shows the proportion of newborns with IRF who underwent laboratory tests, blood cultures, hospital admission, and/or who received antibiotic therapy, empirically or due to suspected EONS, in the two study periods. Although the proportion of patients with equivocal or clinical disease and the proportion of positive blood cultures were similar, during the second-period tests to rule out sepsis were reduced by 34.0 % (from 56.2 % in Period 1 to 37.1 % in Period 2), blood cultures by 13.1 % (from 22.9 % to 19.9 %), hospital admissions by 13.5 % (from 22.9 % to 19.8 %) and the administration of antibiotics by 45.9 % (from 22.9 % to 12.4 %). Attending doctors complied with the SRC recommendations on 1560 (83.6 %) occasions. However, in 240 (12.9 %) patients in whom routine observation was recommended, laboratory tests or hospital admission were carried out. In addition, in 50 (2.7 %) patients in whom laboratory tests and close observation without antibiotic therapy were recommended, antibiotics were finally administered. On the contrary, the remaining 17 patients (0.9 %) were managed more conservatively than recommended by the SRC (Supplementary Table 1).
Main outcomes and comparison between infants born in the two periods, before (January/2018 to June/2019) and after (July/2019 to December/2020), the implementation of the Kaiser-Permanente Neonatal Sepsis Risk Calculator (Risk ratios calculated by Poisson regression; Period 1 as reference).
Values are n/N and % (95 % confidence intervals), except b: median (interquartile range). a Includes 3 possible contaminations during Period 1 and 4 during Period 2. MCA, Maternal corioamnionitis.
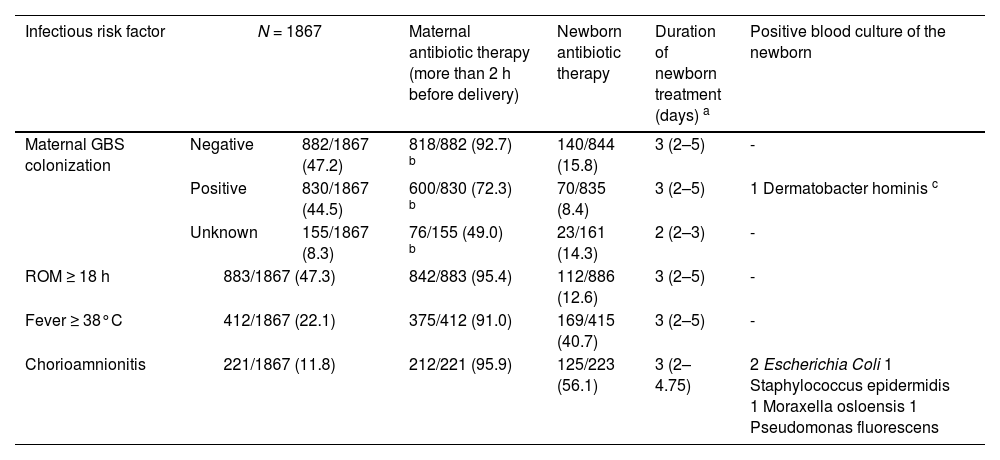
Table 3 shows the distribution of IRF and the maternal and newborn antibiotic therapy administered, together with the positive blood cultures results and the bacteria isolated. Finally, Supplementary Table 2 shows the clinical characteristics of the six patients whose bacteriological results were positive. All patients had normal laboratory tests, including leukocyte count and C-reactive protein. Patient 1 had a positive blood culture for Dermatobacter hominis that was considered a contaminant and received only 4 days of antibiotics. Patients 2 and 3 had positive blood cultures for E. coli. The first was completely asymptomatic throughout his stay, but the second appeared ill immediately after birth. Both received antibiotic therapy with favorable evolution and were discharged without incidents. Patients 4, 5 and 6 did not present symptoms at birth but due to a history of MCA, hospital admission for surveillance was decided and blood cultures were obtained. Given the positive results, although reported as possible contaminations, antibiotic therapy was started. All of them evolved favorably and were discharged healthy.
Distribution of Infectious Risk Factors and relationship with positive blood culture in the newborn (Period 2).
| Infectious risk factor | N = 1867 | Maternal antibiotic therapy (more than 2 h before delivery) | Newborn antibiotic therapy | Duration of newborn treatment (days) a | Positive blood culture of the newborn | |
|---|---|---|---|---|---|---|
| Maternal GBS colonization | Negative | 882/1867 (47.2) | 818/882 (92.7) b | 140/844 (15.8) | 3 (2–5) | - |
| Positive | 830/1867 (44.5) | 600/830 (72.3) b | 70/835 (8.4) | 3 (2–5) | 1 Dermatobacter hominis c | |
| Unknown | 155/1867 (8.3) | 76/155 (49.0) b | 23/161 (14.3) | 2 (2–3) | - | |
| ROM ≥ 18 h | 883/1867 (47.3) | 842/883 (95.4) | 112/886 (12.6) | 3 (2–5) | - | |
| Fever ≥ 38°C | 412/1867 (22.1) | 375/412 (91.0) | 169/415 (40.7) | 3 (2–5) | - | |
| Chorioamnionitis | 221/1867 (11.8) | 212/221 (95.9) | 125/223 (56.1) | 3 (2–4.75) | 2 Escherichia Coli 1 Staphylococcus epidermidis 1 Moraxella osloensis 1 Pseudomonas fluorescens | |
All values are n/N (%), except
Median (interquartile range). ROM, Rupture of Membranes. GBS, Group B Streptococcus.
In “Duration of newborn treatment” only patients who received antibiotics are included.
Antibiotics were administered to some patients because of an indication different from or in addition to GBS colonization.
This isolation was considered a contamination. The mother of this patient did not receive intrapartum antibiotic therapy. The mothers of the other five infant in whom bacteria were isolated in the blood culture had been diagnosed with chorioamnionitis and had received broad-spectrum antibiotics for more than 4 h.
In the present study a total of 11,364 newborns ≥35 weeks GA, 3663 (32.2 %) were exposed to some IRF. Once the use of the SRC was implemented in the Department, there was a reduction in the request for analytical studies from 56.2 % to 37.1 %. Like previous studies,17-21 the present results clearly show the utility of the SRC as part of the protocol for the management of newborns ≥35 weeks GA with IRF to reduce blood draws, hospital admissions, and the use of antibiotics. However, although the authors managed to significantly reduce the rate of antibiotic use by 45.9 %, from 22.9 % to 12.4 %, it is still above the 5 to 8 % rate referred by others.14 Furthermore, a recent study suggests that serial clinical observation could safely reduce the use of antibiotic therapy even to one-third of that recommended by the SRC.22 In that study only 1.9 % of infants received antibiotics for a short period of time. In the present study, if the decision to screen and treat or not had been based on clinical findings and physical examination in the first hours of life, the total number of patients treated could have been between 2 and 6 %. (Supplementary Table 1).
The incidence of MCA in the studied center (3.7 %) is slightly higher than that reported in the literature,1,2,23 and this subgroup of patients is the one that most frequently receives antibiotic therapy in our setting. However, the authors also managed to reduce its administration by 43.6 %, from 99.5 % to 56.1 % in this group of infants (Table 2). In a recent study, Money et al. showed that it would be possible to safely reduce the use of antibiotics in well-appearing infants born to mothers with MCA from 99.7 % to 2.5 % with the use of SRC.24 Some authors recommend attributing a higher risk to these patients, assigning them a pre-test risk of 4‰.16 At the time of the study the authors used a pre-test risk of 0.5‰ for all infants. However, patients with a history of MCA were considered intermediate risk and a blood culture was obtained systematically in these cases. The use of a pre-test risk of 4‰ also gives this recommendation, and it is the one the authors currently use. This strategy allowed us to detect bacteremia in patient number 2. E. coli was isolated and antibiotic therapy started at 14 h of life and the infant evolved favorably. In summary, all the patients diagnosed with EONS in whom a pathogen was isolated in the blood culture presented clinical symptoms in the first 24 h, except in one case in which E. Coli was isolated in the blood culture obtained due to a history of MCA. The significance of a positive blood culture in an asymptomatic newborn has been controversial for a long time.25 It is difficult to establish whether these infants would develop symptoms if they were not treated and to differentiate true EONS from transient bacteremia.
In our setting, many clinicians still rely on white cell count and acute phase reactants, such as CRP or procalcitonin, for guidance in antibiotic administration. As shown in Supplementary Figure 1, providers prescribe antibiotics with significantly lower risk scores than the SRC: median (IQR) 0.4 (0.2–2.3) vs 6.3 (2.3–11.8); p < 0.001 (Panel A). This is mainly due to the prescription of antibiotics in asymptomatic patients with a history of MCA and, although not statistically significant, higher CRP values (Panel B). However, several studies have shown the low predictive value of laboratory tests.26 In fact, when CRP is excluded from the sepsis workup panel, it is possible to reduce around 30 % the exposure of patients to antibiotics without additional risks.27
The present study has limitations. Firstly, this study reflects the experience of a single center and, since patient management is a provider-dependent decision, the recommendations derived from the SRC are not always rigorously followed. The authors achieved 83.6 % compliance with the SRC recommendations, which means that further improvements are still possible. Sometimes, after the successful introduction of new practices, if strict quality control is not maintained, customs are relaxed, and less motivated clinicians return to old practices. A Quality Improvement project on this subject is currently underway in our Unit.
The present study has also some strengths. The prospective data from this study add to existing evidence showing the successful use of the SRC in infants admitted to a regional referral level III teaching hospital. The authors studied systematically all newborns during a specific period of time, including those with any of the IRFs considered in the SRC. MCA is considered an important IRF and is given special consideration. Our digital system for recording medical histories makes data collection and patient follow-up easy and reliable. Finally, the hospital has a Maternal-Neonatal Joint Hospitalization Unit, so all patients with intermediate-risk could be admitted with their mother for close clinical monitoring until the result of the blood culture is available.
ConclusionsThe implementation of the SRC in the management guidelines for newborns ≥35 weeks GA and ≥2000 g birthweight with IRF is safe and adequate to reduce laboratory tests, blood cultures, and hospital admissions, as well as the use of antibiotics. In our setting, strategies are still needed to further reduce the overuse of antibiotics. Given that not every case of EONS is predictable,9 serial clinical observation of at-risk infants remains the most important aspect of clinical practice. As almost all infants with proven sepsis will develop symptoms in the first 48 h of life, close observation could be instrumental in achieving or even improving all those goals.28
Funding sourcesThere was no funding for this research.
The authors are greatly indebted to all the personnel, mostly doctors and nurses, participating in the care of the babies and their families.
Data regarding the first period mentioned in this study were already published as a “hypothetical study”, as is stated in the text (Mazabanda López DA, et al. Management of neonates with 35 weeks of gestational age or more with infectious risk factors at birth: opportunities for improvement. J Perinat Med 2022;50:1150-6.). However, the present study is a prospective analysis that shows the real improvements derived from the intervention of interest.
Deidentified individual data are available on request.