This study aimed to investigate the clinical significance of serum microRNA-146a and pro-inflammatory factors in children with Mycoplasma pneumoniae pneumonia after azithromycin treatment. microRNA-146a is known to regulate inflammatory responses, and excessive inflammation is a primary characteristic of MPP.
MethodsChildren with MPP received conventional symptomatic therapy along with intravenous administration of azithromycin for one week. Serum levels of microRNA-146a and pro-inflammatory factors were measured using RT-qPCR and ELISA kits, respectively. The correlation between microRNA-146a and pro-inflammatory factors was analyzed by the Pearson method. Pulmonary function indexes were assessed using a pulmonary function analyzer, and their correlation with microRNA-146a and pro-inflammatory factors after treatment was evaluated. Children with MPP were divided into effective and ineffective treatment groups, and the clinical significance of microRNA-146a and pro-inflammatory factors was evaluated using receiver operating characteristic curves and logistic multivariate regression analysis.
ResultsSerum microRNA-146a was downregulated in children with MPP but upregulated after azithromycin treatment, contrasting with the trend observed for pro-inflammatory factors. MicroRNA-146a showed a negative correlation with pro-inflammatory cytokines. Pulmonary function parameters were initially reduced in children with MPP, but increased after treatment, showing positive/inverse associations with microRNA-146a and pro-inflammatory factors. Higher microRNA-146a and lower pro-inflammatory factors predicted better efficacy of azithromycin treatment. MicroRNA-146a, tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), interleukin-8 (IL-8), and forced expiratory volume in the first second/forced vital capacity (FEV1/FVC) were identified as independent factors influencing treatment efficacy.
ConclusionAzithromycin treatment in children with MPP upregulates microRNA-146a, downregulates pro-inflammatory factors, and effectively improves pulmonary function.
Mycoplasma pneumoniae is a small, cell wall-less, and self-replicating pathogen that commonly causes lower or upper respiratory tract infections, including community-acquired pneumonia (CAP).1 The clinical manifestations of Mycoplasma pneumoniae infection include cough, fever, sore throat, hoarseness, headache, myalgias, chills, and general malaise.2 While most respiratory tract infections caused by M. pneumoniae resolve without complications, a significant proportion progress to pneumonia. In some cases, particularly those that are severe or refractory, complications such as respiratory failure, multi-organ dysfunction, pleural effusion, and long-term sequelae like bronchiectasis and bronchiolitis obliterans can occur.3 MPP is particularly prevalent in school-age children and poses a significant health burden due to the immature development of their autoimmune systems, and its incidence is on the rise.4 The recommended treatment for MPP is macrolide antibiotics, which act by inhibiting protein synthesis in bacterial ribosomes, and possess both direct antimicrobial and anti-inflammatory properties.5 Azithromycin, a widely used macrolide antibiotic, effectively targets the normal metabolism of mycoplasma and suppresses the synthesis of mycoplasma proteins, leading to the successful treatment of pneumonia.6 Despite the extensive use of azithromycin in MPP, the precise effects and underlying mechanism of this antibiotic remain largely unknown.
microRNAs (miRNAs) are small non-coding RNAs that play crucial roles in post-transcriptional gene regulation and are implicated in various diseases and cellular processes, including cell differentiation, growth, and apoptosis.7 In the context of lung diseases, miRNAs have emerged as key regulars of immune and inflammatory responses.8 Among them, miR-146a has garnered significant attention as a critical modulator of immunological tolerance, such that its deficiency in regulatory T cells can disrupt immune homeostasis.9 High levels of miR-146a have been associated with decreased activation of the inflammatory cascade following lung infection and improved survival.10 Notably, overexpression of miR-146a in alveolar macrophages has been shown to reduce the levels of pro-inflammatory cytokines, such as interleukin (IL)−6 and tumor necrosis factor (TNF)-α, highlighting its anti-inflammatory functionality.11 In the context of Mycoplasma pneumoniae infection, airway epithelial cells play a crucial role as the first line of defense by secreting various pro-inflammatory factors, including TNF-α, IL-1β, IL-6, and IL-8, whereas the induction of these pro-inflammatory cytokines contributes significantly to the development of Mycoplasma pneumoniae pneumonia (MPP), leading to local inflammation, airflow obstruction, airway remodeling, emphysema, and impaired lung function.12 The induction of proinflammatory cytokines is extensively involved in MPP development.13 Given the potential role of miR-146a and pro-inflammatory factors in pneumonia, it is essential to investigate their significance in the context of MPP.
Currently, azithromycin is the first-line drug for the treatment of MPP in children. However, there is limited research exploring the changes in miR-146a and inflammatory factors during azithromycin treatment. Therefore, this study aimed to investigate the effects of azithromycin on miR-146a and pro-inflammatory factors in the serum of MPP children. By analyzing the alterations in these molecular factors, the authors hope to provide valuable reference values for their clinical application in the management of MPP. Understanding the impact of azithromycin on miR-146a and pro-inflammatory factors can contribute to elucidating the underlying mechanisms of action and potentially guide the development of novel therapeutic strategies for MPP in children.
Materials and methodsEthics statementThis study was conducted in accordance with the ethical principles outlined by the Academic Ethics Committee of the Children's Hospital of Soochow University. Prior to participation, all children involved in the study were provided with detailed information about the nature of the study, and their voluntary agreement to participate was obtained. In cases where the child was capable of providing informed consent, the consent form was signed by the child and their parents or legal representative. In situations where the child was unable to provide consent, the form was signed by their parents or legal representative.
Study subjectsA total of 173 children diagnosed with Mycoplasma pneumoniae pneumonia (MPP) and admitted to the study's Hospital between January 2021 and June 2022 were included in this study. Additionally, 120 healthy children who underwent physical examination during the same time were recruited as the normal group for comparison.
Inclusion criteriaChildren were eligible for inclusion in the study if they met the following criteria: 1) Diagnosed with MPP based on clinical symptoms, including fever (> 37.5 °C), cough, abnormal auscultation of breath sounds, abnormal chest X-ray, and blood test; 2) Positive M. pneumoniae polymerase-chain-reaction (PCR) testing of nasopharyngeal secretions or bronchoalveolar lavage fluid; 3) Aged between 2 and 12 years; 4) First-time diagnosis of MPP and no prior anti-mycoplasma infection treatment.
Exclusion criteriaChildren who met any of the following criteria were excluded from the study: 1) Allergic to the drugs used in this study; 2) Recent presence of infectious or respiratory diseases, such as tuberculosis and asthma; 3) Detection of other pathogen infections by PCR; 4) Incomplete clinical data; 5) Co-occurrence of bacterial, viral, and chlamydial infectious diseases; 6) Presence of other immune system diseases; 7) Diagnosis of malignant tumors.
TreatmentChildren diagnosed with MPP received conventional symptomatic treatment upon admission, including measures to reduce fever, relieve cough, and eliminate phlegm. In addition to the symptomatic treatment, MPP children were intravenously administered azithromycin (China Resources Double-Crane Pharmaceutical, Beijing, China) at a dosage of 10 mg/kg daily for a duration of one week.
Sample and data collectionPrior to treatment initiation and after completion of the treatment course, a fasting cubital venous blood sample of 4 mL was collected from each child. The blood samples were then centrifuged at 2000 r/min for 20 min, and the resulting upper serum was collected in sterile Eppendorf tubes. The serum samples were subsequently frozen at −80 °C for future analysis. Clinical data, including age, gender, and blood tests (including white blood cell [WBC], neutrophil, and lactate dehydrogenase [LDH] tests) were recorded. The WBC and neutrophils were measured using an automated five-part differential hematology analyzer (SYSMEX, Kobe, Japan). Serum LDH level was determined using LDH assay kits (Mindray, Shenzhen, Guangdong, China).
Reverse transcription-quantitative polymerase chain reaction (RT-qPCR)Total RNA extraction from the serum samples was conducted using the TRIzol reagent (Invitrogen, Carlsbad, CA, USA). The extracted RNA was then reverse-transcribed into cDNA following the instructions provided with the reverse transcriptase kits (Takara, Dalian, Liaoning, China). RT-qPCR was performed on an ABI 7500 Fast Real-Time PCR System (ABI, Foster City, CA, USA) using SYBR Green real-time PCR Master Mix (Takara) and miScript SYBR Green PCR kit (Qiagen, Hilden, German). The PCR cycling conditions consisted of an initial denaturation at 95 °C for 30 s, followed by 35 cycles of denaturation at 95 °C for 25 s, annealing at 64 °C for 25 s, and extension at 72 °C for 30 s. The relative level of miR-146a was normalized to the internal reference U6 and calculated using the 2−ΔΔCt method. The primer sequences are provided in Supplementary Table 1.
Enzyme-linked immunosorbent assay (ELISA)The serum levels of TNF-α, IL-6, and IL-8 were determined using ELISA kits (Sigma-Aldrich, Merck KGaA, Darmstadt, Germany) following the manufacturer's instructions.
Pulmonary function indexesPulmonary function indexes, including forced expiratory volume in the first second (FEV1), peak expiratory flow rate (PEF), and the ratio of FEV1 to forced vital capacity (FEV1/FVC), were measured using a pulmonary function analyzer (Maibang Medical Instrument, Beijing, China).
Efficacy evaluation and groupingThe evaluation of treatment efficacy was based on the improvement of clinical symptoms and chest X-ray results. Patients in whom clinical symptoms such as cough, fever, and pulmonary rale basically disappeared, and chest X-ray showed normal findings were classified as significantly effective. Patients who experienced relief of clinical symptoms and a ≥ 50% reduction in pulmonary shadow on chest X-ray were classified as effective. Patients who showed no significant improvement in clinical symptoms and chest X-ray findings were categorized as ineffective. Based on these criteria, patients classified as significantly effective (100 cases) and effective (38 cases) were grouped together as the effective group, totaling 138 cases. Patients showing no effect (35 cases) constituted the ineffective group.
Statistical analysisStatistical analysis and data visualization were performed using SPSS 21.0 (IBM Corp. Armonk, NY, USA) and GraphPad Prism 8.01 software (GraphPad Software Inc., San Diego, CA, USA). Count data were presented as the number of cases and percentages and analyzed using the Chi-square test. Measurement data were depicted as mean ± standard deviation (SD). Pairwise comparisons were performed using the t-test, and one-way ANOVA was employed for multi-group comparisons. The correlation between miR-146a and pro-inflammatory factors, as well as the correlations of pulmonary function indexes with miR-146a and inflammatory factors after treatment, were using the Pearson correlation method. Receiver operating characteristic (ROC) curves were generated to assess the predictive value of serum miR-146a and inflammatory factors for azithromycin efficacy. Logistic multivariate regression was performed to identify independent factors associated with azithromycin efficacy.
ResultsClinical baseline characteristics of enrolled populationThe study included a total of 173 children with MPP as observation subjects, along with 120 healthy children from the same period as the normal group. Supplementary Table 2 shows no evident differences in gender, age, and WBC level between healthy children and MPP children (p > 0.05). However, notable differences were observed in neutrophil and LDH levels, with MPP children showing higher levels compared to healthy children (all p < 0.05).
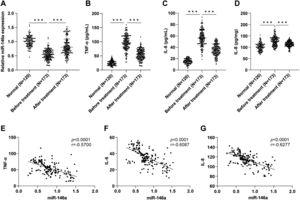
Serum miR-146a upregulation after treatment and negative correlation with TNF-α/IL-6/IL-8To assess the effect of treatment, the authors measured the serum levels of miR-146a. RT-qPCR analysis revealed lower serum miR-146a in MPP children compared to healthy children (p < 0.001). However, significant increases in miR-146a were observed after treatment in MPP children (Figure 1A). The levels of pro-inflammatory factors (TNF-α, IL-6, and IL-8) were measured using ELISA kits, which showed elevated levels of these factors in MPP children, which subsequently decreased after treatment (all p < 0.001) (Figure 1B-D). Furthermore, Pearson correlation analysis demonstrated a negative correlation between serum miR-146a levels and TNF-α, IL-6, and IL-8 in MPP children after treatment (p < 0.0001, r = −0.5700; p < 0.0001, r = −0.6087; p < 0.0001, r = −0.6277) (Figure 1E-G).
Serum miR-146a was upregulated after azithromycin treatment and negatively correlated with pro-inflammatory factors in MPP children. A: Relative expression level of miR-146a in the serum was determined by RT-qPCR; B-C: Levels of TNF-α, IL-6, and IL-8 in the serum were measured by ELISA kits; E-G: Correlation of miR-146a with TNF-α, IL-6, and IL-8 in the serum of MPP children after treatment was assessed by Pearson analysis, respectively. Data were presented as mean ± SD, and one-way ANOVA was used for comparisons among multiple groups, followed by Tukey's test . ***p < 0.001.
Pulmonary function indexes were assessed in the enrolled population using a pulmonary function analyzer. Compared to healthy children, MPP children exhibited significantly reduced levels of FEV1, PEF, and FEV1/FVC levels. However, after treatment, these indexes showed a significant improvement in MPP children (all p < 0.001) (Supplementary Table 3). These findings indicated that azithromycin treatment in children with MPP could enhance pulmonary function parameters. Further analysis using Pearson correlation revealed that FEV1, PEF, and FEV1/FVC levels were positively correlated with miR-146a, while inversely correlated with pro-inflammatory factors in MPP children after treatment (Supplementary Table 4).
Relationship of serum miR-146a and pro-inflammatory factors with azithromycin efficacyTo determine the relationship between serum miR-146a, pro-inflammatory factors, and the efficacy of azithromycin, MPP children undergoing azithromycin treatment were divided into two groups: the effective group and the ineffective group. These findings demonstrated that the effective group exhibited higher miR-146a and lower TNF-α, IL-6, and IL-8 levels compared to the ineffective group (all p < 0.01) (Table 1). These findings suggested that higher levels of miR-146a and lower levels of pro-inflammatory factors were associated with improved efficacy of azithromycin treatment.
Relationship of serum miR-146a and pro-inflammatory factors with treatment efficacy of azithromycin.
Note: TNF, tumor necrosis factor; IL, interleukin.
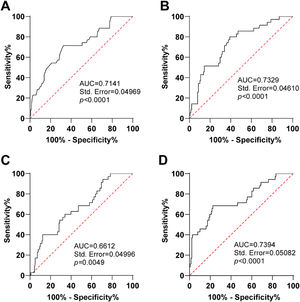
To evaluate the predictive value of serum miR-146a and inflammatory factors in determining azithromycin efficacy, ROC curve analysis was conducted. The area under the curve (AUC) for miR-146a in predicting azithromycin efficacy was 0.7141, with a sensitivity of 71.43% and specificity of 67.39% (Figure 2A). The AUC for TNF-α was 0.7329, with a sensitivity of 80% sensitivity and specificity of 60.87% (Figure 2B). For IL-6, the AUC was 0.6612, with a sensitivity of 40% and specificity of 87.68% (Figure 2C). The AUC for IL-8 was 0.7394, with a sensitivity of 68.57% sensitivity and specificity of 77.54% (Figure 2D). These results indicated that serum levels of miR-146a, TNF-α, IL-6, and IL-8 exhibited good predictive values for azithromycin efficacy.
Predictive values of serum miR-146a and inflammatory factors for efficacy of azithromycin. A, ROC curve of miR-146a for predicting efficacy of azithromycin; B, ROC curve of TNF-α for predicting efficacy; C, ROC curve of IL-6 for predicting efficacy; D, ROC curve of IL-8 for predicting efficacy.
Logistic regression analysis was performed using efficacy as the dependent variable (effective = 1, ineffective = 0) and pulmonary function indexes, miR-146a, and inflammatory factors (as shown in Supplementary Table 3) as independent variables. The analysis revealed that serum miR-146a, TNF-α, IL-6, IL-8, and FEV1/FVC were independent factors influencing the efficacy of azithromycin (Table 2).
Logistic multivariate regression analysis of azithromycin efficacy.
Note: TNF, tumor necrosis factor; IL, interleukin; FEV1, forced expiratory volume in the first second; PEF, peak expiratory flow rate; FVC, forced vital capacity.
Mycoplasma pneumoniae pneumonia (MPP) is a common respiratory infection caused by pathogenic M. pneumoniae and accounts for 10–40% of community-acquired pneumonia (CAP) in children under 18 years of age.14 While MPP is generally benign and mild, severe or prolonged infections can lead to extrapulmonary manifestations, affecting systems such as the digestive, cardiovascular, and nervous systems.15 The host-pathogen interactions play a significant role in signaling and physiological changes within host cells and can induce post-transcriptional regulation of genes involved in immune and inflammatory responses, and the regulation of microRNAs (miRNAs) is closely related to infectious diseases.16 Macrolide antibiotics, particularly azithromycin, are commonly used as the first-line treatment for MPP in clinical practice. Azithromycin offers advantages such as minimal stomach irritation, limited hepatorenal function damage, an extended half-life, and strong inhibition of mycoplasma growth.4 In this study, the authors investigated the effects of azithromycin on miR-146a and pro-inflammatory factors in the serum of MPP children. The results showed that azithromycin therapy increased miR-106a expression and decreased levels of pro-inflammatory factors.
Neutrophils play a crucial role as innate immunity sentinels and cellular responders to inflammation, and in MPP, M. pneumoniae attaches to bronchial epithelial cells, leading to the production of interleukin-8 (IL-8), which drives the recruitment and activation of neutrophils.17 LDH (lactate dehydrogenase), a primary inflammatory marker, is released into the serum during inflammatory processes and serves as a biomarker for monitoring tissue injury.18 Consistent with previous findings, the levels of neutrophil and LDH were found to be elevated in children with MPP.
Various serum biomarkers and inflammatory cytokines play a crucial role in the immunopathogenesis of M. pneumoniae infection, and the release of IL-1β, IL-8, and TNF-α by immune cells results can cause vascular endothelial cell injury, contributing to the progression of MPP.19 Children with refractory MPP often have higher serum levels of IFN-γ and TNF-α.20 Equally importantly, miR-146a is a well-known negative regulator of innate immunity and inflammation.21 Previous studies have shown that miR-146a-5p is dysregulated in pneumonia patients and can mitigate inflammation injury in pneumonia cell models.22 In the present study, the authors observed that children with MPP had decreased miR-146a expression and increased levels of TNF-α, IL-6, and IL-8 in their serum. However, these changes were alleviated by azithromycin therapy. Furthermore, miR-146a showed an inverse correlation with these pro-inflammatory factors after treatment. Similarly, in refractory MPP, reduced levels of miR-146a-5p have been associated with inflammation alleviation through the IRAK-1/RXR/LXR pathway, highlighting its potential as a therapeutic target.23 The gene regulation mediated by miR-146a contributes to the repolarization of human alveolar macrophages, shifting the production of pro-inflammatory towards an anti-inflammatory phenotype.24 Azithromycin treatment has been shown to suppress M. pneumoniae infection-elicited inflammation, decrease cell viability, and inhibit apoptosis, partially through the modulation of the miR-499a-5p/STAT3 axis.25 Collectively, these findings suggest that targeting miR-146a and pro-inflammatory cytokines could hold promise for azithromycin therapy in MPP.
It is important to note that MPP can lead to significant impairment of lung function, affecting both small and large airways to varying degrees.26 Consistent with prior research,27 the present study demonstrated that azithromycin treatment significantly improved lung function in MPP children, as evidenced by increased FEV1, PEF, and FEV1/FVC values. Loss of miR-146a has been associated with severe disruption of the alveolar-capillary barrier, excessive secretion of pro-inflammatory cytokines, and impaired lung function.28 In the present study, the authors observed a positive correlation between post-treatment levels of pulmonary function indexes and miR-146a, while a negative correlation was found with pro-inflammatory factors. These findings suggest that miR-146a may play a role in preserving lung function and modulating the inflammatory response in MPP.
miRNAs have been recognized for their potential to modulate the expression levels of target genes associated with pharmacological responses, and pharmacological therapies can restore altered miRNA levels, indicating their utility as biomarkers for predicting treatment response.29 Similarly, inflammatory factors have been shown to predict the efficacy of treatments and the development of inflammation-induced diseases.30 For instance, in COVID-19 patients, the response to tocilizumab is closely related to increased miR-146a expression and decreased IL-6 levels.31 However, the clinical significance of miR-146a and pro-inflammatory factors in predicting azithromycin efficacy in MPP patients is not well understood. The present findings innovatively revealed that higher levels of serum miR-146a and lower levels of TNF-α, IL-6, and IL-8 were predictive of better efficacy of azithromycin in MPP children. Additionally, miR-146a, TNF-α, IL-6, IL-8, and FEV1/FVC were identified as independent factors influencing the efficacy of azithromycin.
In conclusion, the administration of azithromycin in MPP children led to a significant increase in miR-146a expression, a decrease in pro-inflammatory factors, and an improvement in pulmonary function indexes, ultimately resulting in higher clinical efficacy. However, there are several limitations to consider. The use of miR-146a as a biomarker for Mycoplasma pneumoniae pneumonia in children still presents challenges in daily practice and the hospitalization of pediatricians. Moreover, the number of cases included in the present study was limited, highlighting the need for future studies to expand the sample size and increase confidence in the results. Furthermore, the present analysis focused solely on the expression levels of serum miR-146a and pro-inflammatory factors in MPP children and their correlation, while the precise underlying mechanism between miR-146a and pro-inflammatory factors requires further in-depth exploration.
Ethics approval and consent to participateThis study was approved by the Academic Ethics Committee of Children's Hospital of Soochow University. All children were informed of the study's nature and agreed to participate in it if they could make a decision. If the patient could sign the informed consent, it was voluntarily signed by himself/herself and his/her parents or legal representative. If not, it was signed by his/her parents or legal representative.
Consent for publicationNot applicable.
Availability of data and materialsThe data that support the findings of this study are available from the corresponding author upon reasonable request.
FundingNo funding was received for this study.
Authors' contributionsZW is the guarantor of the integrity of the entire study and contributed to the manuscript preparation; ZW, CYL contributed to the study concepts and manuscript editing; CYL contributed to the study design, definition of intellectual content, manuscript review; YD, CYL contributed to the literature research; CC, YD contributed to the clinical studies; YQL, YD contributed to the experimental studies; YQL, CC contributed to the data acquisition; ZW, YQL contributed to the data analysis, statistical analysis; All authors read and approved the final manuscript.
Not applicable.