This study aims at determining the relationship between prednisolone cumulative dose and linear growth in pre-pubertal children with idiopathic nephrotic syndrome.
MethodThis cross-sectional study was conducted on all children with idiopathic nephrotic syndrome registered to the pediatric nephrology department at the main referral children's hospital in Southwestern Iran. Inclusion criteria included age (males <12 years; females <10 years), >6 months of use, and the minimum prednisolone cumulative dose of 152mg/kg. The exclusion criteria were individuals who had entered puberty or had other diseases affecting linear growth. Based on the prednisolone cumulative dose of ≥550mg/kg (four or more relapses), the children were divided into two groups. All data regarding age, height, and weight at disease onset and the last visit, bone age, and the parents’ height were collected. Secondary variables including mid-parental target height and predicted adult height were also calculated. Height data were compared between the different rates of relapse.
ResultsA total of 97 children (68% male) were enrolled. Their post-treatment mean height Z-score was less than that obtained before treatment (−0.584 vs. −0.158; p=0.001). Subjects with higher prednisolone cumulative doses were found to have more reduction in height Z-score (p=0.001). Post-treatment height prediction also showed less growth potential compared to pre-treatment target height (p=0.006). Thirty-three children (34.4%) had four or more relapses, among whom more mean-height Z-score decreases were found compared to those with less-frequent relapses (−0.84 vs. −0.28; p=0.04).
ConclusionThis study showed the negative effect of cumulative dosages of prednisolone on linear growth, which was greater in children with four or more relapses.
Determinar a relação entre a dose cumulativa de prednisolona e o crescimento linear em crianças pré-púberes com síndrome nefrótica idiopática.
MétodoEstudo transversal conduzido em todas as crianças com síndrome nefrótica idiopática registradas no departamento de nefrologia pediátrica no principal hospital infantil para encaminhamento no sudoeste do Irã. Os critérios de inclusão incluíram idade (meninos<12 anos; meninas<10 anos), >6 meses e a dose cumulativa de prednisolona mínima de 152mg/kg. Os critérios de exclusão foram indivíduos que entraram na puberdade ou tinham outras doenças que afetam o crescimento linear. Com base na dose cumulativa de prednisolona de ≥550mg/kg (≥4 recidivas), as crianças foram divididas em dois grupos. Foram coletados todos os dados relacionados a idade, estatura e peso no início da doença e na última visita, idade óssea e estatura dos pais. Também foram calculadas as variáveis secundárias, inclusive estatura-alvo e estatura adulta prevista. Os dados de estatura foram comparados entre as diferentes taxas de recidivas.
ResultadosForam inscritas 97 crianças (68% do sexo masculino). Seu escore z de estatura média pós-tratamento foi inferior ao obtido antes do tratamento (−0,584 em comparação com −0,158; p=0,001). Os indivíduos com maiores doses cumulativas de prednisolona mostraram maior redução no escore z para estatura (p=0,001). A estatura pós-tratamento também foi preditiva de menor potencial de crescimento em comparação com a estatura-alvo pré-tratamento (p=0,006); 33 crianças (34,4%) apresentaram ≥4 recidivas, entre as quais foram encontradas mais reduções médias no escore z para estatura em comparação com as recidivas menos frequentes (−0,84 em comparação com −0,28; p=0,04).
ConclusãoEste estudo mostrou o efeito negativo das doses cumulativas de prednisolona sobre o crescimento linear, que foi maior em crianças com ≥4 recidivas.
Prednisolone (PDN) has changed the prognosis of nephrotic syndrome (NS) since 1950. Nearly 12–24% of patients are steroid-resistant and about 70% experience at least one episode of relapse.1 Besides the great benefits, there are many concerns about the disadvantages of PDN, particularly about its negative effects on linear growth. Several mechanisms have been proposed regarding the effects of NS on growth.2
Some studies have confirmed the short-time inhibitory effects of PDN on growth by comparing height Z-score (Ht.Zs) before and after treatment between frequent and infrequent relapsers.1,3 Other studies have investigated its negative impact in long-term use,4,5 some others have followed patients longitudinally to compare their final height with their peers,6,7 and finally, some have used a combination of methods.8
Theoretically, growth potential can be evaluated using final height prediction in childhood. Several methods have been proposed for this purpose. The chronological age (CA)-based system decreases the prediction as bone age (BA) increases. However, the bone age-based system does not do this. In the BA-based system, the predictions are less affected by bone age discrepancies, but the effect is not symmetrical. Greulich-Pyle (GP) and Tanner-Whitehouse (TW) methods for skeletal age determination can be used for predicting adult height, which is helpful in diagnosing and evaluating the effects of treatment. In this method, the predicted adult height (PAHt.) is compared to the expected adult height (target range of height; target height [THt.], calculated as mid-parental height [MPHt.]).9
By newer TW Mark systems, the errors of prediction are generally a little smaller than those arising from the Bayley-Pinneau predictions, and considerably smaller in the case of postmenarcheal girls.10
The aim of this study was to evaluate potential short-term and long-term effects of PDN on linear growth by comparing: (a) Ht.Zs before and after treatment, (b) post-treatment predicted final height compared with (potential pre-treatment) mid-parental target height, and (c) low and high total cumulative doses of PDN according to the growth variables.
MethodThis cross-sectional study was conducted on all children (>2 yr.) and adolescents with the diagnosis of idiopathic nephrotic syndrome (INS) registered to the pediatric nephrology department at the main tertiary (Abuzar) children's hospital in Southwestern Iran from January 2011 to December 2012. All new admissions and recurrences were treated in the in-patient ward and were followed at the out-patient clinic at the same center.
Definitions: INS was diagnosed if urine protein was >40mg/m2/hr., serum protein <2.5g/dL, edema, hyperlipidemia (more than age-appropriate limit) with the absence of gross hematuria, renal failure, prolonged hypertension, and normal complement.11 Relapse was defined as reappearance of proteinuria ≥2+ by dipstick for three consecutive days. Patients who responded during 8 weeks of PDN treatment (and did not relapse frequently before 3 months of drug discontinuation) were defined as SSNS (steroid-sensitive nephrotic syndrome). They were considered as SRNS (steroid-resistant nephrotic syndrome) if there was no response during this period. SDNS (steroid-dependent nephrotic syndrome) was defined if recurrence occurred during the alternate-day treatment or 3 months after discontinuation of treatment.
Inclusion criteria were: age <12 yr. for boys and <10 yr. for girls, more than 6 months having passed the beginning of treatment, receiving at least 152mg/kg – equal to a complete initial course of treatment, and parental agreement. The exclusion criteria were: abnormal blood pressure, chronic kidney disease, any sign of pubertal development, and the presence of any other substantial disease affecting linear growth, such as hypothyroidism and celiac disease (discovered by a full history taking, precise physical examination, and a basic laboratory panel obtained during the first assessment).
All subjects were initially treated with 2mg/kg/d of PDN with a maximum of 80mg/day over 4–6 weeks, followed by 4 weeks of the same steroid dose given on alternate days, and then the dose was tapered over about 3 months. Relapses were treated with PDN 40–60mg/kg/d up to 5 days after disappearance of proteinuria, and then were tapered during the next 8 weeks. SDNS and SRNS patients also received other immunosuppressive drugs, including levamisole, cyclophosphamide, mycophenolate mofetil, and cyclosporine.
The following data were prepared for analysis:
- -
Date of birth, to calculate exact chronological age (CA) at each visit.
- -
Age at diagnosis and the time passed after the diagnosis.
The first and the last weight and height (before and after treatment) measured by a clinic nurse using instruments with an accuracy of 0.1kg and 0.1cm, respectively (Seca®, SP, USA). Height measurements were expressed as height standard deviation score (Ht.-SDS) based on the Tanner-Whitehouse reference growth curves.12
Parents’ heights at the beginning of treatment were used to calculate MPHt. as pre-treatment Ht. prediction.
Bone age: radiographs of the left hand and wrist were taken and rated according to the GP system.13 All bone X-rays were reviewed by two radiologists separately and the mean values were used as the final report.
Medications, duration, and dosesNumber of relapses.
Total cumulative dose of PDN was considered as the sum of 150mg/kg for initial treatment and 100mg/kg for each relapse for SSNS subjects. For SDNS and SRNS individuals, all doses of PDN used between the relapses were also added to the calculation. Based on this variable, to assess the possible relation of total PDN dose and linear growth, patients were divided into two groups: less than 550mg/kg [150+(3×100)] equal to three or fewer relapses; and patients with ≥550mg/kg equal to four or more relapses.
Secondary variables were calculated as follows:
- -
Height standard deviation score (Ht.-SDS) was calculated as: Ht.SDS=(x−x¯)/SD, where "x¯" and “SD” are the age-matched population mean height and standard deviation, respectively, and “x” is the patient's height (population data according to Tanner et al.12). For example, based on CDC charts, the 50th percentiles for boys and girls at 20 years of age (used for parental Ht.-SDS determination) were considered as 177cm and 163.3cm, respectively. The Ht.-SDSs at this age were 7.0cm and 6.5cm, respectively.
- -
MPHt. was calculated as the target height (the potential height range that a child is expected to reach if no postnatal disorder or environmental factor has interacted with the genetic height potential). THt. was calculated as: mean parental height+6.5cm in males and mean parental height−6.5cm in females.
- -
The PAHt. was calculated using TW mark-2 equations.14 The variables Ht., CA, BA, and MPHt. are used in this equation.
The equations permit a prediction of adult height, which, in 95% of instances, is within ±7cm of true adult height in boys aged 4–12 and ±6cm in girls aged 4–11.
With this equation, only two coefficients are provided for each chronological year (i.e., for 6, 6.5, 7, 7.5, etc.). Therefore, the coefficient of each child referring to the nearest half-year point has been used in this study. This system was limited to the ages more than 6 years. Consequently, their previous coefficients (TW mark-1) were used10 for younger subjects. In comparison with the 1975 system (TW mark-1) and with the newer version, the standard errors of prediction (actual fewer estimates) are about the same or, on occasion, larger (near half a centimeter). Recent standard errors are therefore preferred.14
For assessment of short-time side-effects of PDN on linear growth, statistical differences between the first and the last Ht.-SDS were analyzed by comparing 95% confidence intervals.
Finally, predicted adult height (±3.5cm as the standard errors of prediction) and the mid-parental target height of each patient were compared; less values of predicted heights will be expressed as the negative effect of PDN on the potential linear growth.
Delta Ht.Z-score and Delta Ht. Prediction were calculated as follow:
All variables including numbers of relapses (cumulative PDN dose) were compared between the different types of NS, and between the boys and girls. Student's t-test was used to compare ΔHt.Zs in different groups. Correlation between height changes and prednisolone doses was evaluated by Pearson's correlation test. Data were expressed as mean±SD. All p-values were two-sided and considered statistically significant for <0.05.
This study was approved by the research ethics committee of Ahvaz Jundishapur University of Medical Sciences (U-91156).
ResultsNinety-seven patients were enrolled (68% male; male/female ratio=2.1). The patients were aged 1–11 yr. (3.9±2.0 yr.) at the onset of disease and 2.5–11.8 yr. (6.95±2.41 yr.) at the end of the study (only one pre-pubertal 14-year-old boy was included). Mean duration of the follow-up was 2.87 years (ranging from 8 months to 9.9 years). This mean was 3.9±1.8 yr. for SDNS and SRNS patients. All collected and calculated data are shown in Tables 1 and 2.
Mean difference of growth indices and their values in 90 children with nephrotic syndrome before and after treatment with prednisolone.
| N | Mean dif.a | R | p | |
|---|---|---|---|---|
| All subjects | 90 | −0.42 | – | 0.001 |
| Boys | 61 | −0.500 | – | 0.001 |
| Girls | 29 | −0.270 | – | 0.231 |
| <550mg | 60 | −0.302 | – | 0.022 |
| ≥550mg | 28 | −0.686 | – | 0.011 |
| Correlation with PCD | ||||
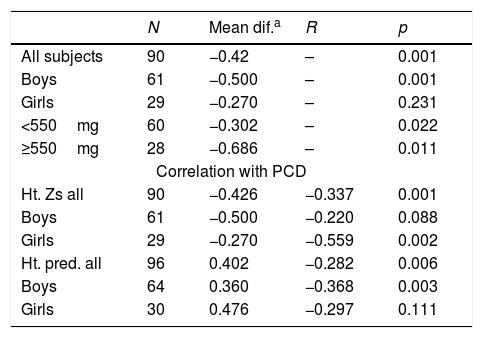
| Ht. Zs all | 90 | −0.426 | −0.337 | 0.001 |
| Boys | 61 | −0.500 | −0.220 | 0.088 |
| Girls | 29 | −0.270 | −0.559 | 0.002 |
| Ht. pred. all | 96 | 0.402 | −0.282 | 0.006 |
| Boys | 64 | 0.360 | −0.368 | 0.003 |
| Girls | 30 | 0.476 | −0.297 | 0.111 |
PCD, prednisolone cumulative dose.
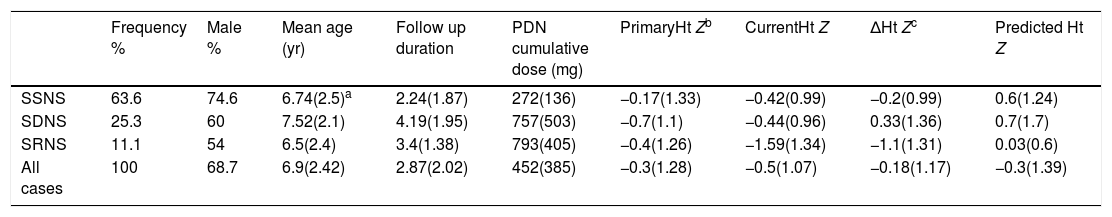
Height Z-scores of 99 children treated for different types of nephrotic syndrome.
| Frequency % | Male % | Mean age (yr) | Follow up duration | PDN cumulative dose (mg) | PrimaryHt Zb | CurrentHt Z | ΔHt Zc | Predicted Ht Z | |
|---|---|---|---|---|---|---|---|---|---|
| SSNS | 63.6 | 74.6 | 6.74(2.5)a | 2.24(1.87) | 272(136) | −0.17(1.33) | −0.42(0.99) | −0.2(0.99) | 0.6(1.24) |
| SDNS | 25.3 | 60 | 7.52(2.1) | 4.19(1.95) | 757(503) | −0.7(1.1) | −0.44(0.96) | 0.33(1.36) | 0.7(1.7) |
| SRNS | 11.1 | 54 | 6.5(2.4) | 3.4(1.38) | 793(405) | −0.4(1.26) | −1.59(1.34) | −1.1(1.31) | 0.03(0.6) |
| All cases | 100 | 68.7 | 6.9(2.42) | 2.87(2.02) | 452(385) | −0.3(1.28) | −0.5(1.07) | −0.18(1.17) | −0.3(1.39) |
SSNS, steroid-sensitive nephrotic syndrome; sDNS, steroid-dependent nephrotic syndrome; SRNS, steroid-resistant nephrotic syndrome.
In the SSNS group, 26 patients had several episodes of relapses and the remaining 37 patients had none (six were new cases and before 10-month follow up). The median for relapse rate was two (1–6 relapses) in the SSNS group and five (3–24 relapses) in the SDNS group.
The mean PDN cumulative dosage was 452.6mg/kg and the median was 350mg/kg. The mean PDN cumulative dosages in SSNS, SDNS, and SRNS groups were 272, 757, and 793mg/kg, respectively. Thirty-six patients (36.3%) needed other immunosuppressive drugs, such as steroid sparing agents (SSA): levamisole (n=25), cyclophosphamide (n=8), MMF (n=11), and cyclosporine (n=8).
Effect of cumulative dosage of prednisolone on heightSeventy-one (66%) of all 97 patients had less than four relapses (<550mg/kg PDN; group 1) and the remaining 33 subjects, group 2, had four or more relapses. The second group (frequent relapsers) showed more decrease in mean Ht.Zs compared to the first group (−0.84 vs. −0.28; p=0.04).
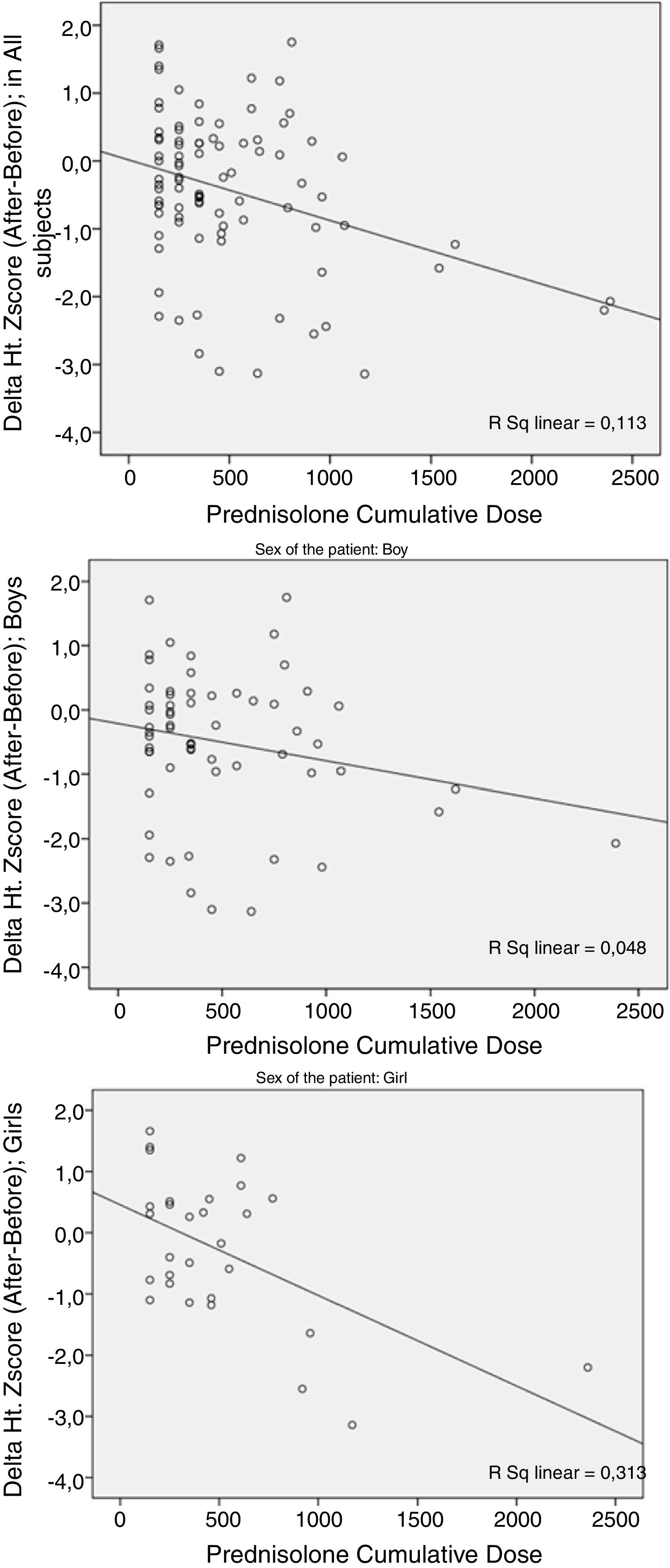
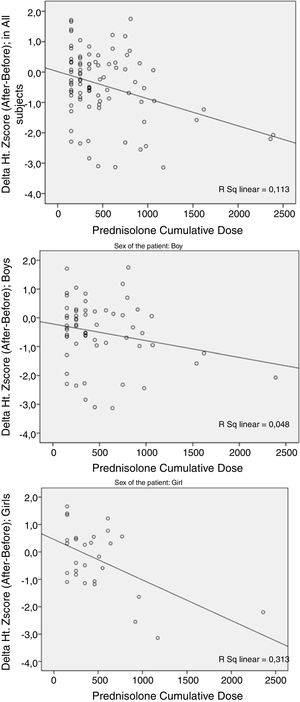
The cumulative dosage of PDN had a meaningful relationship (p=0.001) with the deterioration of ΔHt.Zs (the difference between post- and pre-treatment Ht.Zs). This deterioration was larger in female patients (Fig. 1).
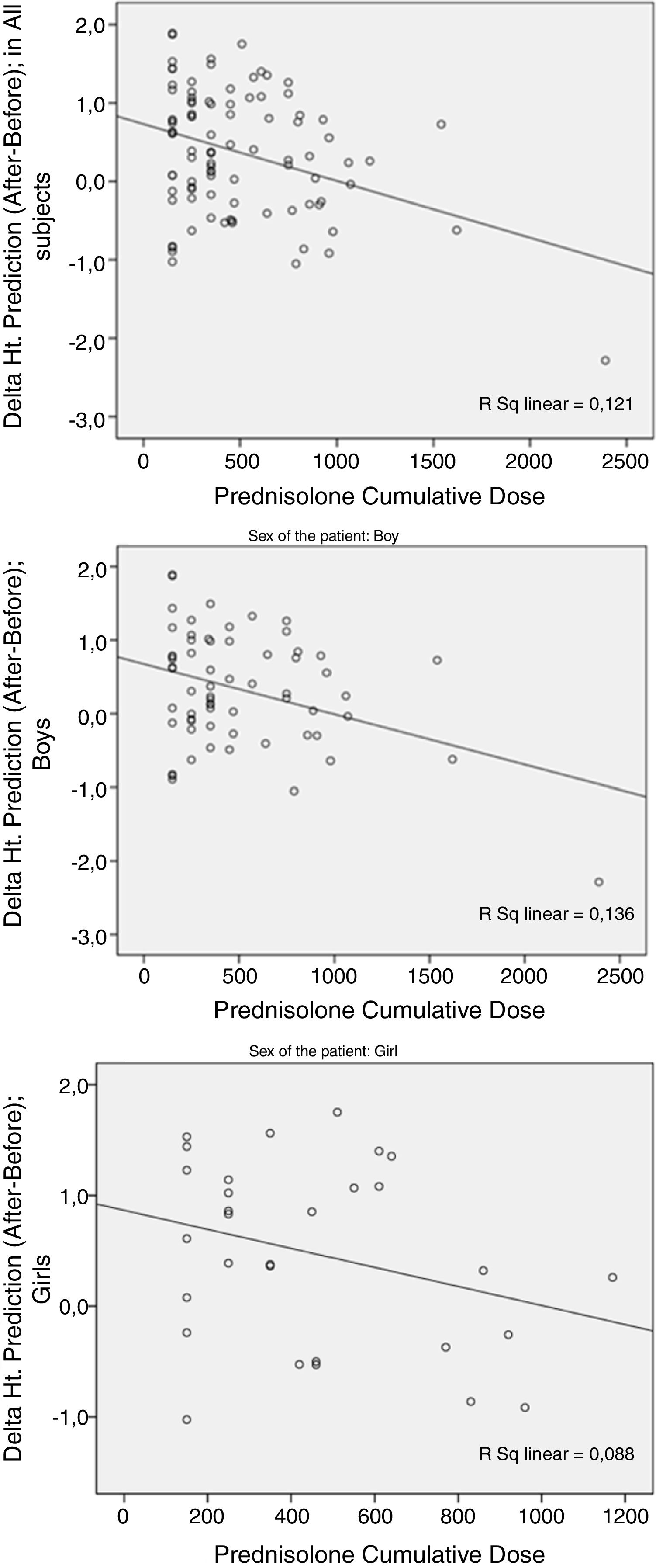
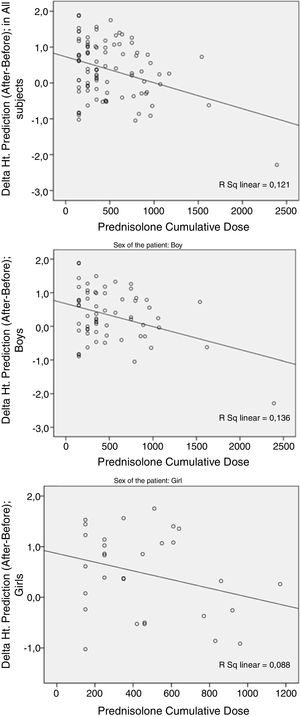
Compared to the mean of target Ht.Zs (pre-treatment predicted height), the mean of post-treatment predicted Ht.Zs was increased to almost 0.4 SD, but again more PDN cumulative dosages were associated with greater decrease in the post-treatment prediction compared to the pre-treatment level. However, statistical significance was found only in male patients (p=0.006; Fig. 2).
As is clear in the figures, these two variables (ΔHt.Zs and ΔHt. prediction) differed by sex. Pearson's test revealed a higher correlation of prednisolone cumulative dose (PCD) and ΔHt.Z score in girls than boys; p=0.002 vs. 0.088. Regarding the Δprediction, there was higher correlation in boys than girls; p=0.003 vs. 0.111.
Patients who needed other immunosuppressive drugs had lower values of ΔHt.Zs (p=0.047), and this effect was more significant in the group of four or more relapses (p=0.03). In those with less than four relapses, the cumulative dosage of PDN did not have any meaningful relationship with the outcome measures.
Deterioration of the mean Z-score had no meaningful relationship with the type of nephrotic syndrome (p>0.05), allergy (p>0.05), family history of renal and non-renal diseases (p>0.05), presence of hypertension (p>0.05), and parental consanguinity (p>0.05).
DiscussionThis study was designed to assess the possible short- and long-term effects of steroids on the linear growth of pre-pubertal children with nephrotic syndrome. Results showed its negative effect on patients with SDNS and four or more relapses. While the effect was more noticeable in girls over the short term, it was more noticeable in boys over the long term. The results of this study were similar to previous ones in many aspects. This study showed that the post-treatment Ht.Zs of patients receiving PDN ≥550mg/kg had reduced significantly, and that the deterioration of Ht.Zs was associated with an increase in cumulative PDN dose.
The study by Donatti et al. on 85 SSNS patients, over all, did not show any significant alterations in the Ht.Zs and the mean height percentile between the first and last visit; by dividing patients into two groups, however, if they had improved Ht.Zs, the total dose and duration of PDN, and the chronological age were higher in the last group. This group also had retarded growth and puberty.16
In an opposite direction, Adhikari et al., in a study on an African/Indian population, did not found any significant difference between treated and untreated (symptomatically managed) children in mean Ht.-SDS. They then concluded that courses of daily steroids or persistent proteinuria did not inhibit linear growth.17 That study had not considered the effect of cumulative PDN dose or relapse rate in their evaluation.
Non-uniform results seen in previous studies could mostly be due to differences in their methodologies, and since a cut-off has been used for high- vs. low-dose PDN exposures in this study, it is hard to compare this study with the previous ones.
The suppression of osteoblastogenesis in bone marrow and the promotion of osteocyte and osteoblast apoptosis are consequences of steroids, which lead to a decrease in bone formation. The accumulation of apoptotic osteocytes could account for some definitions, such as osteonecrosis, aseptic necrosis, or avascular necrosis. The negative effect of steroid use on bone mass and short-term growth depends on the type and dose of steroid, and occurs mostly during the first 6 months of treatment. The common belief is that trabecular bones are affected more than cortical ones. However, a recent study in patients with acute lymphoblastic leukemia found a high incidence of cortical bone involvement after glucocorticoid treatment.
However, there is some evidence indicating that steroids induce bone resorption by prolonging osteoclast lifespan. Besides, they promote calcium loss through the kidneys and intestine, producing a negative balance in calcium metabolism that induces more osteoclastic activity due to secondary hyperparathyroidism.2,18
The effect of PDN cumulative dose on linear growth is shown better with longitudinal studies. For example, the study by Emma et al. on children with SDNS (mean follow up=11.7±3.5 years) showed loss of almost 0.5 SD of Ht.-SDS during the pre-pubertal period, and 0.7 SD of their target Ht. (only in male subjects) when they reached their final height. More prolonged treatment was associated with higher risk of Ht.-SDS loss.6 These findings were similar to the present results in that this study also discovered long-term negative effects (lower values for Ht. prediction after treatment) limited to boys. In comparison with the study by Emma et al., using a cut off makes the present approach different, but looking at PDN impact on ΔHt.Zs and height prediction using MPHt.Z could be a point of strength in the present study. It was also shown in the study by Ribeiro et al.4 that long-term PDN, particularly in higher doses, affects height SDS as well as spinal bone density. A longitudinal study by Rees et al. on 41 patients (29 boys) with SSNS also showed a decrease in Ht.-SDS in boys (only after 10 years, and it was associated with delay in puberty) but not in girls.3 This finding requires further research for explanation.
Simmonds et al. determined a cut-off point in which lower cumulative PDN dose was compatible with normal growth. By calculating daily PDN dose and Ht.-SDS velocity between clinics, they found no negative effect on growth at doses of PDN <0.75mg/kg/day; with higher doses, there was a small decline in Ht. SDS velocity.19 In the present study, a similar relationship with the cumulative doses of PDN ≥550mg/kg was found.
Under high-dose steroid treatment, the physiological secretion of growth hormone may be reduced or mediated by somatostatin and, in some cases, growth hormone stimulation tests are unable to produce an appropriate response.20
Attainment of peak bone mass and growth may also be impaired by steroid use because of an altered pituitary-gonadal axis. A reversible hypo-gonadotropic hypogonadism is shown in studies in adults using steroid therapy. Adrenal insufficiency following chronic glucocorticoid therapy may also be associated with depressed levels of other sex steroids, such as androstenedione and estrogen. Steroids may also impair FSH and cause reduction in estrogen secretion.21,22
Puberty occurs between childhood and the time when final height is obtained. Its beginning time varies according to the ethnic and familial genetic basis, and it may last 2–6 years to complete. For each family and sibling, it has an individual pattern of progress regarding the overall rate and stages intervals. Even in a healthy child, the exact prediction of pubertal onset and its growth spurt is not possible. With steroid exposure and its negative effects on pubertal process and bone formation, such predictions become more difficult. Accordingly, the present study completely excluded pubertal stages from follow-up analysis. Longitudinal follow-up of patients after completion of puberty can solve this problem when they have reached the final height. Finding a sex difference in short-term and long-term negative PDN impacts (in girls and boys, respectively) matches with the shorter pre-pubertal years in girls than in the boys.
Finally, it was better to compare pre-treatment and post-treatment PAHt.Z, but because pre-treatment bone ages were not available in subjects, MPHt.Z was used as the basic potential target height, which, theoretically, must not change from childhood to adolescence. Comparing different items before and after treatment to evaluate PDN side effects can cause some errors. Although the number of patients in this study was greater than in the previous ones, a much larger population in a multicenter study for longer period would give better results. Short duration (<2.5 yr.) of follow-up in more than half of the patients may affect the results.
Moreover, the equations used in this study were obtained based on data from a British population. However, they have been checked in a Japanese population. Still, they need to match with the raw data of each population to assess applicability.14
Finally, the authors were more familiar with the GP system for bone age assessment, and then it was used as a part of TW mark-2 system for height prediction. In fact, the original method for bone age rating in TW formula is their own system, and not that of GP.
This study showed the negative effect of cumulative PDN dose on linear growth in nephrotic syndrome; fortunately, it happened mostly in prolonged disease with four or more relapses. This finding is important for giving parental assurance about relative safety of this lifesaving drug. Finally, the importance of earlier growth intervention in frequent relapses is warranted.
FundingThis study was supported by Vice chancellor for research of the Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran.
Conflicts of interestThe authors declare no conflicts of interest.
Please cite this article as: Valavi E, Aminzadeh M, Amouri P, Rezazadeh A, Beladi-Mousavi M. Effect of prednisolone on linear growth in children with nephrotic syndrome. J Pediatr (Rio J). 2020;96:117–24.