(a) To determine the 3rd, 50th, and 97th percentiles of weight, length, and head circumference of newborns from the Ribeirão Preto BRISA cohort, according to gender and gestational age, and compare them with the Intergrowth-21st standard; (b) To estimate the small for gestational age (<3rd percentile), large for gestational age (>97th percentile), stunting (length<3rd percentile), and wasting (body mass index<3rd percentile).
MethodsObservational study of a cohort of 7702 newborns between 01/01/2010 and 12/31/2010 in the city of Ribeirão Preto, SP, Brazil. The 3rd, 50th, and 97th percentiles were determined for the anthropometric measurements using fractional polynomial regression.
ResultsThe weight difference between Ribeirão Preto and Intergrowth-21st was small, being more pronounced in preterm infants (mean difference between the two populations of +266g); for full-term newborns, there was a mean difference of +66g, and for post-term infants, of −113g. For length, the mean variation was always <1cm; whereas for head circumference, preterm newborns showed a variation >1cm, and full-term and post-term newborns showed a variation of <1cm. The small and large for gestational age detection rates were 2.9% and 4.3%, respectively. Stunting affected 6.5% of all newborns and wasting, 1.5%, with a predominance in girls and in full-term pregnancies; both conditions were present in 0.4% of the sample.
ConclusionsNewborns from Ribeirão Preto, when compared to the Intergrowth-21 standard, are heavier, longer, and have a larger head circumference until they reach full-term.
Determinar os percentis 3, 50 e 97 de peso, comprimento e perímetro cefálico de recém-nascidos da Coorte BRISA Ribeirão Preto, segundo sexo e idade gestacional, e comparar com o padrão Intergrowth-21st; b) Estimar os fenótipos pequeno para idade gestacional ( < percentil 3), grande para idade gestacional ( > percentil 97), stunting (comprimento < percentil 3) e waisting (índice de massa corporal < percentil 3).
MétodoEstudo observacional de uma coorte de 7.702 recém-nascidos entre 01/01 e 31/12/2010 na cidade de Ribeirão Preto, SP, Brasil. Os percentis 3, 50 e 97 para as medidas antropométricas foram determinados com regressão polinomial fracionária.
ResultadosA diferença de peso entre Ribeirão Preto e Intergrowth-21st foi pequena, mais acentuada nos recém-nascidos pré-termo (diferença média entre as duas populações foi de + 266 gramas); para os recém-nascidos a termo a diferença média foi de + 66 gramas e para os pós-termo de -113 gramas. Para comprimento, a variação média foi sempre < 1cm; enquanto que para perímetro cefálico os recém-nascidos pré-termo apresentaram variação > 1cm e os recém-nascidos a termo e pós-termo tiveram variação < 1cm. As taxas de detecção de pequeno e grande para idade gestacional foram 2,9% e 4,3%, respectivamente. Stunting afetou 6,5% de todos os recém-nascidos e waisting 1,5%, com predomínio em meninas e em gestações a termo; ambas as condições estavam presentes em 0,4% da amostra.
ConclusõesOs recém-nascidos de Ribeirão Preto, quando comparados com o padrão Intergrowth-21st, apresentam-se mais pesados, mais longos e com maior circunferência craniana até chegarem a termo.
Intrauterine growth is understood as the result of several factors, including the expression of genetic information, maternal nutrition,1 maternal diseases such as preeclampsia, socioeconomic factors, and environmental factors, such as hypoxia.2 The universally accepted indicator of fetal malnutrition is low birth weight (<2500g),3 but it does not differentiate between stunting (low length for age) and wasting (low body mass index [BMI] for age). Other existing terms to describe prenatal malnutrition include: small for gestational age (SGA), intrauterine growth restriction (IUGR), and placental insufficiency. Traditionally, the Battaglia and Lubchenco classification has been used to classify newborns (NB) into small, adequate, and large for gestational age (GA) in relation to the 10th and 90th percentiles regarding the weight variable.4 Information such as being born SGA is important from a clinical and social point of view because it shows a correlation with important outcomes, such as neonatal and infant mortality, low childhood growth, and chronic diseases in adulthood.5,6
Neonatal reference curves have been published in some countries, but none of them can be used as an international standard for growth.7 Currently, the use of these curves is disputed, because they were created to be used as references for a country, aiming to compare the anthropometric characteristics of NB with those of the overall population, i.e., without the prescriptive nature of a standard applicable to different populations.8
The International Fetal and Newborn Growth Consortium for the 21st Century (INTERGROWTH-21st) is a cross-sectional, multicenter, and transcultural study of NB growth, carried out with the same prescriptive approach and methodological design as that used for the creation of the World Health Organization (WHO) standard currently used for child and adolescent growth evaluation in Brazil.9 The INTERGROWTH-21st (IG-21), which allows the anthropometric analysis of NB, was designed to be a “prescriptive” fetal growth pattern, i.e., to be used as the basis for recommending how healthy fetuses born to healthy mothers in healthy settings “should” grow, rather than a mere reference to how fetuses born in the same countries “actually grow”.10
In this context, the aims of the present study were: (a) to compare the anthropometric data (weight, length, and head circumference) of the birth cohort of Ribeirão Preto 2010 (BRISA-RP) with the IG-21 standard, and (b) to estimate NB phenotypes (SGA, large for gestational age [LGA], stunting, and wasting) according to gender and GA.
MethodsThis was a cross-sectional secondary analysis with a descriptive approach of data obtained from a cohort study (Brazilian Ribeirão Preto and São Luís Birth Cohort Studies – BRISA), whose objective was to find new risk factors for preterm birth.11 All newborns who were alive between 01/01/2010 and 12/31/2010 in the city of Ribeirão Preto (PR), São Paulo, Brazil were assessed. During this period, 7702 newborns were evaluated, equivalent to 98% of hospital deliveries in the municipality.
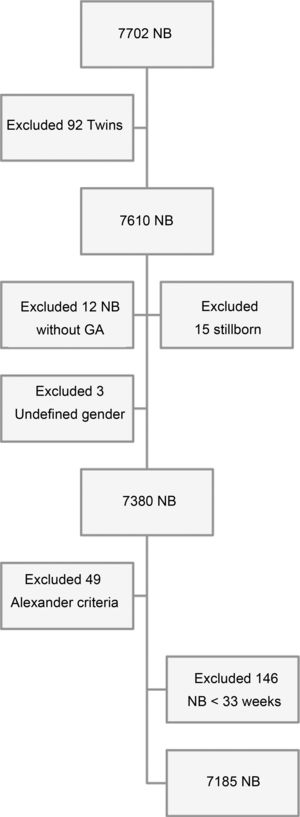
Inclusion criteria were children born at hospitals, to mothers living in RP, whereas the exclusion criteria were twin births, stillborn births, those without GA defined by the adopted criteria, those with no defined gender, and NB who had birth weight values incompatible with gestational age12 (Fig. 1).
Data were obtained from an interview with the mothers after delivery and from information contained in the newborns’ medical records.
Assessed variablesMaternal age (years), ethnicity (white or non-white), level of schooling (years), marital status (with or without partner), occupation of the head of the family (according to the classification proposed by Olsen and Frische13), Brazilian economic classification criterion according to the Brazilian Association of Research Companies (CCEB-ABEP),14 number of cigarettes smoked daily during pregnancy, parity, arterial hypertension (prior or gestational), diabetes (previous or gestational), body mass index (BMI, kg/m2) before pregnancy (categorized as low weight [<18.5kg/m2], adequate weight [≥18.5kg/m2 and <25kg/m2], overweight [≥25kg/m2 and <30kg/m2], obesity [≥30kg/m2), and prenatal care (adequate≥4 consultations).
The neonatal variables studied were gender, birth weight (BW), birth length, and head circumference (HC). Stunting was defined as length<3rd percentile, wasting as BMI<3rd percentile, SGA as birth weight<3rd percentile, and LGA as birth weight>97th percentile of the IG-21 study.10,15
GA was estimated based on date of the last menstrual period (LMP) and information from the earliest ultrasonography (USG) examination. A margin of error of ±7 days was calculated for the date found of the first USG.16
Statistical analysesThe descriptive statistics included the mean value, proportion, standard deviation (SD), and 95% confidence interval (95% CI). Fractional polynomials were used to fit the models of the three anthropometric measurements (BW, length, and HC). The generalized additive models for location, scale, and shape (GAMLSS) provide the option of different distributions other than normal (bias distributions and kurtosis), as well as the modeling of other parameters of a distribution that determines scale and shape by using fractional polynomials.17 In all cases, the technique of fractional polynomial smoothing by gender was applied for the 3rd, 50th, and 97th percentiles. For comparison of the curves with the IG-21 standard, the lower limit of 33+0 weeks was adopted, given that the IG-21 has a very questionable curve below 33 weeks considering the small number of newborns included.
According to the IG-21 study, the smoothed percentiles of weight, length, and HC for the newborns are shown at exact weeks of gestational age only to avoid the publication of long tables for each day of pregnancy. For instance, the exact GA for the 36th week value is 36 weeks plus 0 days.18 All models and goodness-of-fit evaluations were performed using the statistical software R (R Foundation for Statistical Computing, Vienna, Austria) using GAMLSS support. All graphs were produced using R software.
The project was approved by the Research Ethics Committee of the HCFMRP (process no. 11157/2008). All mothers who agreed to participate in the study signed the informed consent.
ResultsThe mean maternal age was 27 years (SD=6.2), and the percentage of young and old mothers was similar. Most of the population self-reported as white, approximately half of the mothers had a level of schooling between 9 and 11 years, and the vast majority of the mothers declared having a partner. In most families the occupation of the head of the family was unskilled or semi-skilled manual worker. The upper and intermediate social classes showed a similar representation (Table 1, Supplementary Material).
About 1/3 of the mothers were overweight and obese, slightly more than 10% said they were smokers, and about 13% of the mothers were hypertensive and 6% were diabetic.Most of the mothers were primiparous and only 2 % of pregnant women presented inadequate prenatal care (Table 2, Supplementary Material). The distribution by gender was equivalent. Preterm births represented 13% and almost 8% of infants had low birth weight. The SGA rate was 2.9%, while the LGA rate was 4.3%, with an inverse association with GA. Stunting affected 6.5% of the sample and wasting, 1.5% (Table 1), with a predominance of girls and full-term pregnancies; both conditions were present in 0.4% of the sample.
Newborn phenotypes according to gender and gestational age (33–42 weeks). BRISA-PR cohort, 2010.
| Phenotype | n | % | 95% CI |
|---|---|---|---|
| Stunting | |||
| Boys | 214 | 6.1 | 5.3–6.9 |
| Girls | 246 | 6.3 | 6.1–7.8 |
| Total | 460 | 6.5 | 5.9–7.1 |
| Per gestational age (weeks) | |||
| 33–37 | 78/1208 | 6.4 | 5.1–8.0 |
| 38–42 | 382/5887 | 6.5 | 5.9–7.2 |
| Wasting | |||
| Boys | 46 | 1.3 | 1.0–1.7 |
| Girls | 56 | 1.6 | 1.2–2.0 |
| Total | 102 | 1.5 | 1.2–1.8 |
| Per gestational age (weeks) | |||
| 33–37 | 19/1208 | 1.6 | 1.0–2.4 |
| 38–42 | 83/5887 | 1.4 | 1.1–1.7 |
| SGA | |||
| Boys | 93 | 2.6 | 2.1–3.2 |
| Girls | 112 | 3.1 | 2.6–3.7 |
| Total | 205 | 2.9 | 2.5–3.3 |
| Per gestational age (weeks) | |||
| 33–37 | 35/1222 | 2.7 | 2.0–3.9 |
| 38–42 | 170/5899 | 2.9 | 2.5–3.3 |
| LGA | |||
| Boys | 153 | 4.3 | 3.7–5.1 |
| Girls | 153 | 4.3 | 3.6–4.9 |
| Total | 306 | 4.3 | 3.8–4.8 |
| Per gestational age (weeks) | |||
| 33–37 | 131/1222 | 10.7 | 9.0–12.6 |
| 38–42 | 175/5899 | 3.0 | 2.6–3.4 |
NB, newborns.
Stunting, length<3rd percentile of IG-21.
Wasting, BMI<3rd percentile of IG-21.
SGA (small for gestational age): birth weight<3rd percentile of IG-21.
LGA (large for gestational age): birth weight>97th percentile of IG-21.
Fig. 1 (Supplementary Material) shows the BRISA-RP histograms of birth weight according to gender. The distributions are very similar, although that of the girls shows greater kurtosis.
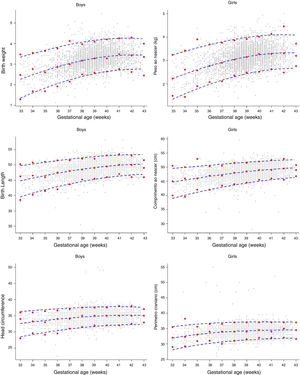
Distribution curves of BW, length, and HC of the BRISA-RP cohort were constructed, according to GA and gender. Fig. 2 shows the individual values (observed values) and the smoothed 3rd, 50th, and 97th percentiles (estimated values), using fractional polynomials. Few outliers were observed after 37+0 weeks of GA for BW and almost identical values were observed for length and HC at birth.
The 3rd, 50th, and 97th smoothed percentiles curves (blue lines) for birth weight, birth length, and head circumference at birth according to gestational age. Empirical values are shown for each gestational week (red circles) and the actual observations (gray circles). BRISA-RP Cohort, 2010.
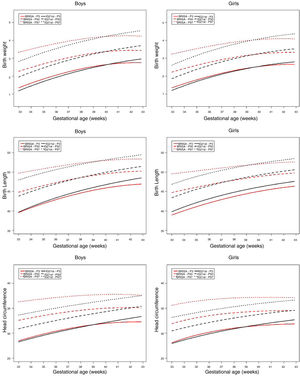
The curves of the 3rd, 50th, and 97th percentiles of the BRISA-RP cohort were compared with IG-21 by GA and gender. For weight in both genders, it was observed that between weeks 33+0 and 39+6, the 50th and 97th percentiles of the BRISA-RP cohort were higher than IG-21; then both percentiles cross the IG-21 pattern and decrease up to week 42+6. For the 3rd percentile, the behavior is similar, but smoother (Fig. 3). In numerical terms, the divergence between BRISA-RP and IG-21 is more pronounced among preterm (33–36 weeks) NB, in which the mean difference between the two populations is +263g for boys and +270g for girls; in full-term infants (37–40 weeks), the mean difference was +81g for boys and +52g for girls; and for post-term (41 and 42 weeks) NB, the mean difference was −120g for boys and −106g for girls (Table 3, Supplementary Material).
Regarding length, the growth curve (50th and 97th percentiles) increased steadily from 33 weeks gestation to 37–39 weeks in the BRISA-RP cohort and then crossed the IG-21 curve. The 3rd percentile always remained below the standard (Fig. 3). Numerically, the mean variation was always<1cm in both boys and girls (Table 4, Supplementary Material).
Finally, the HC for both genders showed a large gap between the 50th and 97th percentiles of the BRISA-PR Cohort and the IG-21 curve until reaching the post-term period; however, the 3rd percentile trajectory was parallel with the IG-21 standard until week 39, when it crossed it (Fig. 3). Regarding the numerical differences, it was observed that preterm NB showed a variation>1cm (1.33cm for boys and 1.39cm for girls), while for full-term and post-term NB, the variation was<1cm (Table 5, Supplementary Material).
DiscussionTo the best of the authors’ knowledge, this is the first Brazilian study that compared a cohort at birth with the international standard IG-21 regarding the size of the newborn for gestational age and gender.
Of the 7185 single live births, those below the 3rd percentile (SGA) and above the 97th percentile (LGA) of the IG-21 standard for birth weight had detection rates of 2.9% and 4.3%, respectively. For stunting and wasting, the values were 6.5% and 1.5%, respectively.
Adjusted percentiles were calculated based on individual data from more than 7000 infants using fractional polynomial regression and data were reported according to full weeks of gestation as smoothed percentiles (Fig. 2), which were consistent with the raw data, increasing the confidence in the curves. SGA was defined as birth weight for age and gender<3rd percentile (instead of the more frequently used 10th percentile), indicating a severe insult; this eliminates the chance of erroneous inclusion of a normal newborn into the lowest birth weight distribution.
It is evident that the birth weight curves of the BRISA-RP cohort and the IG-21 standard, although similar in the full-term period, are very different at the earliest gestational ages. While at the 40 weeks of GA there was no difference in the medians, at 33 weeks gestation the median BRISA-RP was almost 300g higher. This makes the prevalence of SGA in preterm births higher in the BRISA-PR cohort than in the standard curve. The probable reasons for a higher frequency of LGA in the BRISA-PR cohort were increases in the maternal pre-gestational body mass index, parity, weight gain, and gestational diabetes, as well as the reduction in maternal smoking, which are associated with higher birth weight and preterm birth.19 These frequent factors in the BRISA-PR cohort (Table 2, Supplementary Material) were exclusion criteria in the IG-21 study.10
The scant birth weight divergence found in the curve of full-term pregnancies may suggest that women who reached full-term tend to be healthier and have a lower risk, with a profile similar to those included in the IG-21 study.
Other possible explanations for the differences found between the BRISA-RP and IG-21 studies can be attributed to the prescriptive approach of IG-21 (populations with low risk of fetal growth impairment; the population was not selected in the BRISA-RP cohort); gestational age estimate (in the BRISA-RP cohort the GA was defined by a combination of clinical methods and estimated by the earliest USG examination; in the IG-21 study, GA was estimated early through the USG performed before the 14th week of gestation), sample size (the BRISA-RP cohort included 7702 mother-child pairs, whereas the IG-21 evaluated 20,486 NB). Overall, small samples affect the estimate of percentiles in extreme GA20; this is reflected in Fig. 3, where the greatest differences between the reference and the standard were observed in the extreme GA. Moreover, there was a difference in the frequency of preterm birth; IG-21 had a very low rate (5.5%), consisting mainly of late preterm deliveries (33–37 weeks gestation) and full-term low birth weight infants (3.2%), probably due to their prescriptive approach. In the BRISA-PR study, preterm birth was 13.2%, more than two-fold the IG-21, although the rate of low birth weight in full-term infants was similar (3.3%).
In Latin America, there is no comparable data on the prevalence of nutritional phenotypes in newborns who were evaluated using IG-21, except in a study carried out in Argentina, where the prevalence of the SGA phenotype was 3.6% compared with a local reference and 2.0% with IG-21, quite similar to that of the present study.21 A Peruvian study of newborns 3400m above sea level found no significant differences with the IG-21 standard, but the phenotypes were not evaluated.22 In a study carried out in New Zealand, the proportions of SGA and LGA were 1.06% and 6.2%, respectively.23
The authors of the cross-sectional NB study IG-21 admitted that the use of a single cut-off point to define growth restriction has limitations, and that assessment of the growth velocity or functional assessment of the newborn can provide better means to identify malnourished NB.10
In this sense, stunting constitutes a global indicator of child well-being, reflecting social inequalities and describing specific outcomes of the neonatal period.24 Stunting can be related to organic conditions and is broadly considered a cumulative and long-term process, analogous to chronic malnutrition in children, which requires exposure to one or more risk factors for several months or during pregnancy.25 For this reason, recently the evaluation of this indicator in newborns has gained prominence from the perspective of the first 1000 days of life. This study incorporates, for the first time in neonatal care routine, a method for the early diagnosis of stunting that can be monitored during childhood, using the corresponding WHO Child Growth Standards.9
Alternatively, it is likely that neonatal wasting reflects acute exposures in the weeks prior to delivery, a time when fat deposition is more important. Some authors, however, suggest that differences in severity, rather than the time and duration of insults, result in distinct fetal growth impairment phenotypes, with wasting representing the most severe cases.26 In an earlier study, stunting affected 3.8% and wasting 3.4% of a low-risk population.15
The observed prevalence rates of newborn phenotypes were relatively low, especially for SGA and waisting, as they are also lower than the cut-off points, with clinical significance suggested by the WHO.3 Stunting at birth seems to have a relatively low prevalence, even in low-income countries, but it increases with gestational age (Table 1).27 A possible explanation for the lower waisting value observed in the present study (1.5%) would be elective pregnancy termination due to fetal growth restriction. This is confirmed by the 17% higher cesarean rate at 33–37 weeks than at 38–42 weeks (p<0.001, data not shown).
From the point of view of clinical practice, the IG-21 standard could underestimate the frequency of full-term and post-term LGA newborns in the BRISA-RP cohort, with implications for the decline of controls and biochemical tests. On the other hand, using IG-21 would reduce the diagnosis of SGA up to full-term. Although the mean variation in length was always less than 1cm, using IG-21 would increase the diagnosis of stunting throughout pregnancy.
Finally, whereas the trajectory of the 3rd percentile of the HC parallel up to week 40, the preterm NB showed a variation>1cm with the BRISA-RP reference, which may influence an overestimation of microcephaly, currently the first step in the diagnosis of Zika virus infection.
It is recommended that the reference curves of each country be compared graphically and numerically with a standard (such as the IG-21) to detect adverse perinatal outcomes (such as the phenotypes used in this study) and their long-term effects.28,29 As with any international standard, local validation is required before implementation.
The main strength of the study is its sample size, with relatively recent data collected from a cohort study (BRISA-RP) following stringent and standardized methods, from an unselected population of postpartum women. The study demonstrates the practical application of recommended international growth standards to estimate the prevalence of four phenotypes. However, it is limited to a single municipality, which prevents its generalization.
Despite the importance of comparing local populations with standard curves, the use of both reference and standard curves for epidemiological studies still requires their validation through indicators of morbidity and mortality in newborns, as well as the occurrence of chronic non-communicable diseases throughout the life cycle.
The NB of the BRISA-RP study, when compared to the IG-21 standard, are heavier, longer, and have a larger HC until they reach full-term.
FundingFundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP): Process No. 08/53593-0. Fundação de Apoio ao Ensino, Pesquisa e Assistência do Hospital das Clínicas da Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo (FAEPA). Institutional Scientific Initiation Grant from USP.
Conflicts of interestThe authors declare no conflicts of interest.
Please cite this article as: Pimenta JR, Grandi C, Aragon DC, Cardoso VC. Comparison of birth weight, length, and head circumference between the BRISA-RP and Intergrowth-21st cohorts. J Pediatr (Rio J). 2020;96:511–9.
Study conducted at Universidade de São Paulo (USP), Faculdade de Medicina de Ribeirão Preto (FMRP), Ribeirão Preto, SP, Brazil.