Diagnosis of central precocious puberty has always been challenging in clinical practice. As an important method in the diagnosis of central precocious puberty, luteinizing hormone-releasing hormone stimulation test is complex and time-consuming. In many cases, clinical traits are inconsistent with luteinizing hormone-releasing hormone stimulation test results, therefore not reliable for diagnosis. In this study, the authors intended to find an indicator that predicts the results of the luteinizing hormone-releasing hormone stimulation test among subjects with early pubertal signs.
MethodsCases of 382 girls with early breast development before 8 years old and luteinizing hormone-releasing hormone stimulation test before 9 years old were included and underwent follow-up tests. Patients with peak luteinizing hormone level ≥5IU/L were considered positive in the luteinizing hormone-releasing hormone stimulation test. Anthropometric data, body mass index, bone age evaluation, blood hormones levels of luteinizing hormone, estradiol, follicle-stimulating hormone, and uterine and ovarian volumes were analyzed.
ResultsSubjects with positive results in the initial test demonstrated early bone maturation, accelerated growth, and elevated basal blood luteinizing hormone, estradiol, and follicle-stimulating hormone levels, when compared with subjects with negative results in the initial test. Subjects with positive results in the follow-up test presented a more advanced bone age and more accelerated linear growth, when compared with subjects with negative results in the follow-up test.
ConclusionsAccording to the statistical analysis, advanced bone age is the most effective predictor of the result of luteinizing hormone-releasing hormone stimulation test.
O diagnóstico da puberdade precoce central sempre foi complicado na prática clínica. Como um importante método no diagnóstico de puberdade precoce central, o teste de estimulação do hormônio liberador do hormônio luteinizante é complexo e demorado. Em muitos casos, as características clínicas são incompatíveis com os resultados do teste de estimulação do hormônio liberador do hormônio luteinizante e, assim, não são confiáveis para o diagnóstico. Neste estudo, visamos constatar um indicador que previsse os resultados do teste de estimulação do hormônio liberador do hormônio luteinizante entre indivíduos com sinais puberais precoces.
MétodosForam incluídos casos de 382 meninas com desenvolvimento precoce das mamas antes dos 8 anos de idade e teste de estimulação do hormônio liberador do hormônio luteinizante antes dos 9 anos e elas foram submetidas a testes de acompanhamento. Os resultados das pacientes com nível máximo de hormônio luteinizante ≥ 5IU/L foram consideradas positivos no teste de estimulação do hormônio liberador do hormônio luteinizante. Foi feita uma análise dos dados antropométricos, do índice de massa corporal, da avaliação da idade óssea, dos níveis sanguíneos de hormônio luteinizante, volumes uterinos e ovarianos de estradiol (E2) e do hormônio folículo-estimulante.
ResultadosOs indivíduos com resultado positive no teste inicial demonstraram maturação precoce do osso, crescimento acelerado e níveis sanguíneos elevados de hormônio luteinizante, estradiol e hormônio folículo-estimulante, em comparação aos indivíduos com resultados negativos no teste inicial. Os indivíduos com resultados positivos no teste de acompanhamento apresentaram um maior avanço na idade óssea e crescimento linear mais acelerado, em comparação aos indivíduos com resultados negativos no teste de acompanhamento.
ConclusõesDe acordo com a análise estatística, a idade óssea avançada é o indicador mais efetivo do resultado do teste de estimulação do hormônio liberador do hormônio luteinizante.
Precocious puberty (PP) is that occurring before age 8 in girls or age 9 in boys.1 In most cases, PP is a result of the premature activation of the hypothalamic-pituitary-gonadal (HPG) axis, which is also called central precocious puberty (CPP). CPP is usually idiopathic except when identifiable causes (e.g. tumors, hydrocephalus, brain lesions, or cranial trauma) are present.2 Early bone maturation, accelerated linear growth, and pelvic ultrasonographical (USG) changes (including abnormal changes in ovarian, uterine, and adrenal volume) are usually present in PP,3,4 in addition to other secondary sex characteristics, such as early growth of pubic hair.5 CPP could lead to early closure of epiphysis, which eventually results in a decrease of height, among other impacts, such as psychological stress.6 The literature reports that girls with early pubertal development present more behavior problems than their peers.7 Thus, a detailed assessment of pubertal development for girls with suspected PP is necessary; if a diagnosis is made, proper treatment should be followed.
Premature thelarche (PT) is defined as an isolated development of breast tissue, with no other signs of sexual maturation; it is usually observed within the first 1–4 years of life. In most cases, PT is observed as a variation of normal physical development, not being considered pathological.8,9 Children with PT present no signs of increased growth or advanced skeletal maturation, and have normal level of basal gonadotropin and E2.10,11 With a prevalence of 4.7%, the causes of PT are largely unknown; 2–13% of cases of PT could progress to CPP,12–14 so the clinical traits, bone age (BA), basal, and stimulated gonadotropin concentrations of the PT cases should be carefully evaluated.
The stimulated LH test is an assistive test that verifies the activity of HPG axis and distinguishes CPP cases from PT cases. For children with early progression of pubertal signs, accelerated linear growth, and early bone maturation, the CPP diagnosis is made when premature activation of HPG axis is confirmed.15 However, the LHRH stimulation test demands a lengthy and complex sampling procedure; and sometimes, multiple tests are needed before a diagnosis of CPP can be made.9,13 It has been reported that LH levels from two samples at 45-minute time point are required for a precise diagnosis of CPP.9 In this study, the authors aimed to find a laboratory or clinical indicator that predicts a positive result in the LHRH stimulation test, since knowledge of such indicators could help decide the timing of the test, thus facilitating the diagnostic procedure of CPP.
In this study, the clinical and laboratory parameters of subjects with early pubertal signs were profiled and compared. Relevant parameters were correlated with the test results in initial and follow-up LHRH stimulation tests to find the most significant predictor of test results.
MethodsSubjects382 girls referred to the Shandong Provincial Hospital, affiliated to Shandong University, between April 2010 and May 2015 for the assessment of suspected PP were selected for the research. Patients selected for the study met the following criteria: (a) female with premature breast development (Tanner stage≥B2) prior to age 8 years; (b) BA≥1 year above chronological age (CA); (c) LHRH stimulation test performed before the age of 9 years; (d) follow-up (≥6 months), each case should be followed-up over age 9. Patients with history of endocrine disorders or intracranial lesions were excluded. This research was approved by the Clinical Research Committee of Shandong Provincial Hospital Affiliated to Shandong University. Informed consent was obtained from the parents/guardians of each participant of the study.
The CPP diagnosis was made when the patient presented: (1) secondary sex maturation before the age of 8 years; (2) advanced BA; (3) peak LH level ≥5IU/L during LHRH stimulation test. Subjects were grouped according to the results from initial and follow-up LHRH stimulation tests.
MethodsCA, BA, weight, height, BMI, and laboratory parameters were collected at the initial LHRH stimulation test. Body weight and height were presented as standard deviation score (SDS). Six-month growth velocity was determined as six-month height increment from the day when LHRH stimulation test (gonadorelin injection) was conducted. Growth velocity ratio was determined by the formula: six-month height increment of subjects/six-month increment of general Chinese girls. A follow-up LHRH stimulation test was performed every three or six months, according to growth velocity and pubertal progression.
Each case was assessed for breast development using the Tanner scale. BA was determined using the Greulich-Pyle method (with radiograph of the wrist and left hand). The predicted adult height (PAH) was calculated using the Bayley-Pinneau method, after the deduction of the mean height of 20-year-old Chinese women (161.2cm).
Blood level of basal FSH and LH was determined with an immunofluorimetric assay kits (Roche®, FIA, IFMA, IN, USA). Electrochemiluminescence immunization (Roche®, Cobas e601, IN, USA) was applied to evaluate the tumor markers including alpha-fetoprotein (AFP) and human chorionic gonadotropin (HCG). E2 blood levels were determined using radioimmuoassay kits (Cisbio Bioassays, United Kingdom). In the LHRH stimulation test, blood samples were collected at 30, 60, and 90min after the administration of LHRH (100μg), and subjected to LH and FSH test. Patients with peak LH>5IU/L were considered to have a positive LHRH stimulation test.
Statistical analysisContinuous variables were expressed as means±standard deviations, and categoric variables were expressed as frequencies and percentages. Student's t-tests were performed on continuous variables, and the chi-squared test or Fisher's exact test was performed on categoric variables. Binary logistic regression analysis and linear regression analysis were performed to assess the significant indicators that could predict LHRH stimulation test results. A ROC curve was also built to identify the cut-off value of the difference between BA and CA (BA-CA) in the prediction of CPP diagnosis. All significant tests were two-tailed, and p-values <0.05 were considered statistically significant. All statistical analyses were performed with the SPSS (SPSS Statistics for Windows, Version 17.0. Chicago, USA).
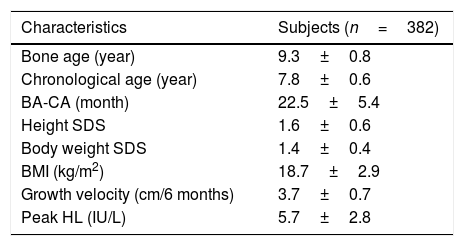
ResultsClinical and auxological features of the subjectsA total of 382 girls were assessed; the clinical and auxological features of the subjects are shown in Table 1. The mean CA of the subjects was 7.8±0.6 years at the initial LHRH stimulation test; mean BA was 9.3±0.8 years; mean BA-CA was 22.5±5.4 months; the weight was 1.4±0.4; mean height SDS of the subjects was 1.6±0.6; mean six-month growth velocity was 3.7±0.7cm; and mean BMI was 18.7±2.9kg/m2.
Baseline subject characteristics.
| Characteristics | Subjects (n=382) |
|---|---|
| Bone age (year) | 9.3±0.8 |
| Chronological age (year) | 7.8±0.6 |
| BA-CA (month) | 22.5±5.4 |
| Height SDS | 1.6±0.6 |
| Body weight SDS | 1.4±0.4 |
| BMI (kg/m2) | 18.7±2.9 |
| Growth velocity (cm/6 months) | 3.7±0.7 |
| Peak HL (IU/L) | 5.7±2.8 |
BA-CA, the difference between the bone age and the chronological age; SDS, standard deviation score; BMI, body mass index.
Data are presented as mean±SD.
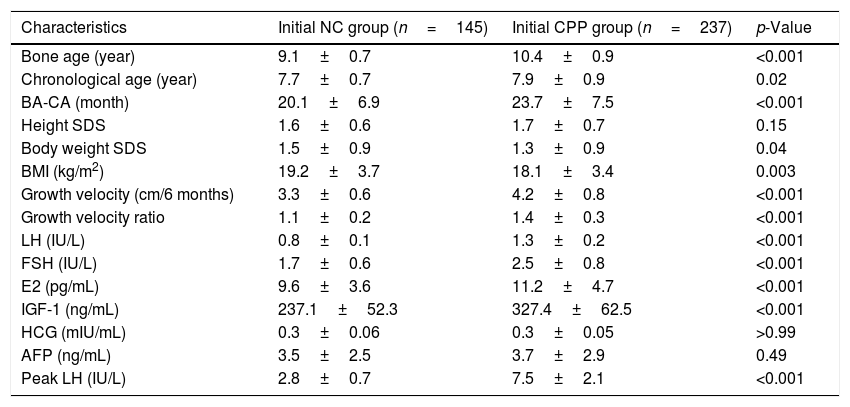
237 subjects were diagnosed with CPP according to the initial LHRH stimulation test and placed into the CPP group, 145 subjects with negative results were treated as negative control (NC group). As shown in Table 2, a higher BA (10.4±0.9 years in the CPP group and 9.1±0.7 years in the NC group; p<0.001) and CA (7.9±0.9 years in the CPP group and 7.7±0.7 years fin the or NC group; p<0.02) were observed between the CPP and NC groups. In addition, a significantly lower BMI (18.1±3.4kg/m2 in the CPP group and 19.2±3.7kg/m2 in the NC group; p=0.003), a higher BA-CA (23.7±7.5 months in the CPP group and 20.1±6.9 months in the NC group; p<0.001), a higher six-month growth velocity (4.2±0.8cm in the CPP group and 3.3±0.6cm in the NC group; p<0.001), and a higher growth velocity ratio (1.4±0.3 in the CPP group and 1.1±0.2 in the NC group; p<0.001) were found in subjects with CPP diagnosis, when compared with the NC group. Furthermore, higher basal LH concentration, basal blood FSH and E2, and blood IGF-1 concentrations were detected in the CPP group, when compared with the NC group. No significant differences were observed between the two groups regarding height and blood AFP and HCG levels.
Clinical and biochemical characteristics of participants in the initial LHRH stimulation test (n=382).
| Characteristics | Initial NC group (n=145) | Initial CPP group (n=237) | p-Value |
|---|---|---|---|
| Bone age (year) | 9.1±0.7 | 10.4±0.9 | <0.001 |
| Chronological age (year) | 7.7±0.7 | 7.9±0.9 | 0.02 |
| BA-CA (month) | 20.1±6.9 | 23.7±7.5 | <0.001 |
| Height SDS | 1.6±0.6 | 1.7±0.7 | 0.15 |
| Body weight SDS | 1.5±0.9 | 1.3±0.9 | 0.04 |
| BMI (kg/m2) | 19.2±3.7 | 18.1±3.4 | 0.003 |
| Growth velocity (cm/6 months) | 3.3±0.6 | 4.2±0.8 | <0.001 |
| Growth velocity ratio | 1.1±0.2 | 1.4±0.3 | <0.001 |
| LH (IU/L) | 0.8±0.1 | 1.3±0.2 | <0.001 |
| FSH (IU/L) | 1.7±0.6 | 2.5±0.8 | <0.001 |
| E2 (pg/mL) | 9.6±3.6 | 11.2±4.7 | <0.001 |
| IGF-1 (ng/mL) | 237.1±52.3 | 327.4±62.5 | <0.001 |
| HCG (mIU/mL) | 0.3±0.06 | 0.3±0.05 | >0.99 |
| AFP (ng/mL) | 3.5±2.5 | 3.7±2.9 | 0.49 |
| Peak LH (IU/L) | 2.8±0.7 | 7.5±2.1 | <0.001 |
CPP, central precocious puberty; NC, negative control; SDS, standard deviation score; BMI, body mass index; BA-CA, difference between the bone age and the chronological age; LH, luteinizing hormone; FSH, follicle-stimulating hormone; E2, estradiol; IGF-1, insulin-like growth factor-I; AFP, alpha-fetoprotein; HCG, human chorionic gonadotropin.
Data are presented as mean±SD.
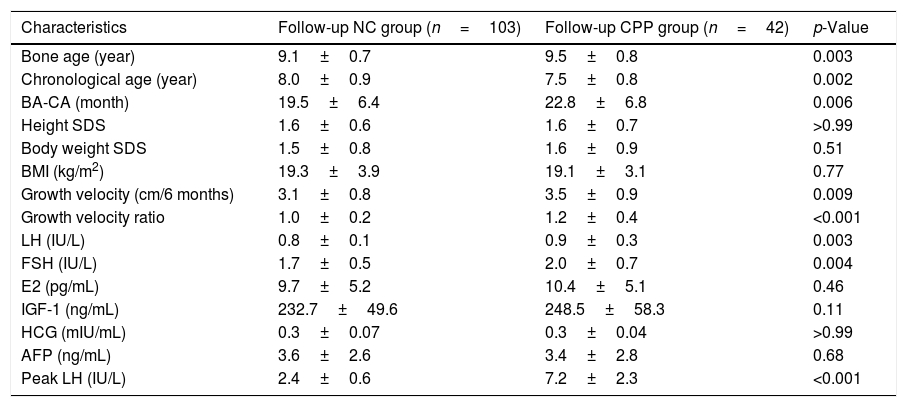
The 145 girls with negative initial LHRH stimulation test results underwent follow-up LHRH stimulation tests. Among the 145 subjects, 42 were diagnosed with CPP in the follow-up LHRH stimulation tests (follow-up positive group) and 103 girls had negative results (follow-up negative group). Higher BA (9.5±0.8 years in the follow-up positive group and 9.1±0.7 years in the follow-up negative group; p<0.005), higher BA-CA (22.8±6.8 months in the follow-up positive group and 19.5±6.4 months in the follow-up negative group; p=0.006), higher six-month growth velocity (3.5±0.9cm in the follow-up positive group and 3.1±0.8cm in the follow-up negative group; p=0.009), and higher growth velocity ratio (1.2±0.4 in the follow-up positive group and 1.0±0.2 in the follow-up negative group; p<0.001) were observed in subjects with positive result in the follow-up test, when compared with subjects with positive result. No statistically significant differences were observed among the groups regarding BMI, body weight SDS, height SDS, basal blood E2, blood IGF-1 concentrations, blood AFP, and HCG levels (Table 3). The BA-CA data of initial examination was used to build an ROC curve and yielded an AUC of 0.605 (0.605±0.03). The use of 19.5 months as BA-CA cut-off value had a sensitivity of 0.7 and a false-positive rate of 0.53 in the prediction of follow-up CPP diagnosis.
Clinical and biochemical characteristics of participants in the follow-up LHRH stimulation test (n=145).
| Characteristics | Follow-up NC group (n=103) | Follow-up CPP group (n=42) | p-Value |
|---|---|---|---|
| Bone age (year) | 9.1±0.7 | 9.5±0.8 | 0.003 |
| Chronological age (year) | 8.0±0.9 | 7.5±0.8 | 0.002 |
| BA-CA (month) | 19.5±6.4 | 22.8±6.8 | 0.006 |
| Height SDS | 1.6±0.6 | 1.6±0.7 | >0.99 |
| Body weight SDS | 1.5±0.8 | 1.6±0.9 | 0.51 |
| BMI (kg/m2) | 19.3±3.9 | 19.1±3.1 | 0.77 |
| Growth velocity (cm/6 months) | 3.1±0.8 | 3.5±0.9 | 0.009 |
| Growth velocity ratio | 1.0±0.2 | 1.2±0.4 | <0.001 |
| LH (IU/L) | 0.8±0.1 | 0.9±0.3 | 0.003 |
| FSH (IU/L) | 1.7±0.5 | 2.0±0.7 | 0.004 |
| E2 (pg/mL) | 9.7±5.2 | 10.4±5.1 | 0.46 |
| IGF-1 (ng/mL) | 232.7±49.6 | 248.5±58.3 | 0.11 |
| HCG (mIU/mL) | 0.3±0.07 | 0.3±0.04 | >0.99 |
| AFP (ng/mL) | 3.6±2.6 | 3.4±2.8 | 0.68 |
| Peak LH (IU/L) | 2.4±0.6 | 7.2±2.3 | <0.001 |
CPP, central precocious puberty; NC, negative control; SDS, standard deviation score; BMI, body mass index; BA-CA, difference between the bone age and the chronological age; LH, luteinizing hormone; FSH, follicle-stimulating hormone; E2, estradiol; IGF-1, insulin-like growth factor-I; AFP, alpha-fetoprotein; HCG, human chorionic gonadotropin.
Data are presented as mean±SD.
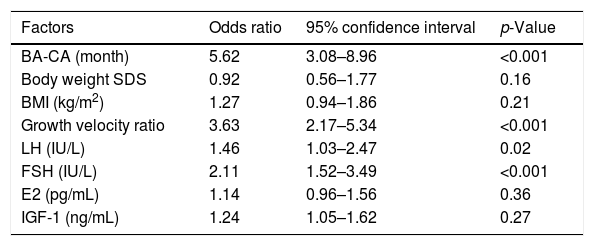
A binary logistic regression model was built to analyze the relevant parameters such as BA-CA; body weight SDS; BMI; growth velocity ratio; basal blood E2, LH, FSH; and blood IGF-1 concentrations, aiming to evaluate the significant indicators that could predict the LHRH stimulation test results. According to the binary logistic regression analysis, four parameters were associated with the LHRH stimulation test results in the initial LHRH stimulation test: BA-CA (OR, 5.62; 95% CI, 3.08–8.96; p<0.001), growth velocity ratio (OR, 3.63; 95% CI, 2.17–5.34; p<0.001), basal blood FSH level (OR, 2.11; 95% CI, 1.52–3.49; p<0.01), and blood basal LH concentration (OR, 1.46; 95% CI, 1.03–2.47; p=0.02; Table 4). In addition, a linear regression analysis on the correlation between BA-CA and peak LH was also conducted. In the linear regression model of initial test data and follow-up data, BA-CA was positively correlated with peak LH, with a coefficient of determination (R2) of 0.78 and 0.65.
Logistic regression analysis of the factors associated with the results of the initial LHRH stimulation test.
| Factors | Odds ratio | 95% confidence interval | p-Value |
|---|---|---|---|
| BA-CA (month) | 5.62 | 3.08–8.96 | <0.001 |
| Body weight SDS | 0.92 | 0.56–1.77 | 0.16 |
| BMI (kg/m2) | 1.27 | 0.94–1.86 | 0.21 |
| Growth velocity ratio | 3.63 | 2.17–5.34 | <0.001 |
| LH (IU/L) | 1.46 | 1.03–2.47 | 0.02 |
| FSH (IU/L) | 2.11 | 1.52–3.49 | <0.001 |
| E2 (pg/mL) | 1.14 | 0.96–1.56 | 0.36 |
| IGF-1 (ng/mL) | 1.24 | 1.05–1.62 | 0.27 |
SDS, standard deviation score; BA-CA, difference between the bone age and the chronological age; E2, estradiol; LH, luteinizing hormone; FSH, follicle-stimulating hormone; IGF-1, insulin-like growth factor-I.
Factors of the initial positive results group (n=237) were compared to those of the initial negative results group (n=145).
In this study, 382 girls were referred to this hospital with initial LHRH stimulation tests; 145 of them had negative test results. Knowing that some diagnosis might have been missed (who are negative in the initial LHRH stimulation test), these 145 cases were regularly followed-up. Using the initial data of 382 cases, a binary regression analysis was performed, demonstrating a strong correlation between BA advance and LHRH stimulation test result (and CPP diagnosis). Higher basal blood levels of LH, E2, and FSH were also observed when compared with subjects with negative LHRH stimulation test. With the initial data of BA-CA and CPP diagnosis, a ROC curve was constructed, and the cut-off value was used to predict the CPP diagnosis (with a sensitivity of 0.7 and false-positive rate of 0.53). In the follow-up test, 42 girls were diagnosed with CPP. These 42 girls were compared with the 103 peers with negative result, and a significantly higher BA advance was observed among CPP girls. BA advance alone effectively predicts a significant amount of CPP diagnoses.
Reviews of trends in timing of puberty indicated a trend toward earlier puberty in girls worldwide. According to a research conducted in 1997 on 17,077 girls from the United States by the Pediatric Research in Office Settings network, the mean age of thelarche onset was 9.96 years.16 With a trend toward earlier pubertal timing, an increasing number of girls were referred to the pediatric endocrinology clinic for the assessment of pubertal precocity signs. In some cases, diagnosis of PP could be challenging, due to the inconsistency between the clinical traits and the LHRH stimulation test results.17,18 Although the LHRH stimulation test is the golden standard in the diagnosis of CPP, it is time-consuming and requires frequent sampling.19 In this study, 382 investigated girls presented a series of early pubertal signs, such as premature breast development, advanced skeletal maturation, and accelerated linear growth. In the initial LHRH stimulation test, 237 subjects had positive CPP diagnoses. Subjects with positive results in the initial test presented lower BMI, higher BA-CA, faster growth velocity, higher basal blood LH, FSH, and E2, and higher blood IGF-1 levels, when compared with those with negative results in the initial test.
According to Della Manna et al.,20 accelerated linear growth, increased uterine and ovarian size, advanced BA, and high spontaneous LH concentration were observed in subjects with PP. Differences in clinical findings and biochemical parameters between both groups were also found, but no further investigation was conducted on these correlations.15 The significant increase of year growth, early skeletal maturation, basal blood LH, peak LH and peak FSH of LHRH stimulation test were also found in diagnosed with PP.21 In this study, it was observed that the BA was the most sensitive indicator of LHRH stimulation test among subjects with early pubertal signs. Studies demonstrated a positive correlation between early pubertal maturation and increased BMI.22,23 In the present study, a significant difference in BMI among the subjects with different initial test results was observed; all values were within the range of general children. It is worth mentioning that some of the breast development signs observed in subjects with negative test results in the initial test were only adipose tissues and not true thelarche.
Studies assessing the differentiation diagnosis of PP and PT have suggested that basal blood LH concentration could reveal the activity of the HPG axis in most CPP patients.24 A higher blood level of IGF-1 in CPP subjects was also observed when compared with PT subjects.25 The peak LH level during LHRH stimulation test was found to be a significant indicator of CPP and subjects with premature breast development, which is presented in the early stages of both PT and PP cases; therefore, attention should be paid to cases with abnormal thelarche.26 A recent study demonstrated that accelerated linear growth is a powerful indicator for positive LHRH test results in girls with early pubertal signs.17 In the present study, 29% of subjects with negative results in the initial test had a CPP diagnosis during the follow-up test. A significantly higher BA-CA and faster growth were observed in subjects with positive results on the initial test when compared with subjects with negative test results in the follow-up test. No significant differences were observed among the groups regarding BMI, basal blood LH, basal blood FSH, basal blood E2 level, and blood IGF-1 concentration. It is difficult to differentiate CPP from cases with an initial negative result (PT, for example) through a mere evaluation of clinical and laboratory traits. The findings of the present study suggest that a detailed evaluation of BA is necessary to determine whether patients need a follow-up LHRH stimulation test and LHRH agonist treatment.
Further research is needed to address the limitations of this study. First, a limited number of cases led to a relatively small sample size; second, this trial included only Chinese participants, and therefore the findings might not apply to the global population; third, this study was a single-center trial. A multicenter cohort study with larger sample size and patients from different ethnicities is necessary. It is also worth mentioning that, although advanced BA is a useful parameter in diagnosing CPP or PP, clinicians should never overemphasize any single parameter in clinical practice. A good decision-making in clinical practice should be flexible and include all useful parameters. Future studies should assess more parameters for a more accurate diagnosis.
In conclusion, CPP patients with positive results on the LHRH stimulation test presented more advanced BA, faster linear growth, and elevated blood gonadotrophin concentrations. Advanced BA, in particular, is the most significant predictor of the LHRH stimulation test result when diagnosing a suspected CPP case.
Conflicts of interestThe authors declare no conflicts of interest.
Please cite this article as: Xu Y-Q, Li G-M, Li Y. Advanced bone age as an indicator facilitates the diagnosis of precocious puberty. J Pediatr (Rio J). 2018;94:69–75.