Determine the frequency of dermatological diagnoses in preterm newborns up to 28 days of life and associated perinatal factors.
Methoda cross-sectional analytical study with a convenience sample and prospective data collection, was conducted between November 2017 and August 2019. Overall, 341 preterm newborns who had been admitted to a University hospital – including those admitted to the Neonatal Intensive Care Unit – were evaluated.
Results61 (17.9%) had less than 32 weeks gestational age (GA), with a mean GA and birth weight of 33.9 ± 2.8 weeks and 2107.8 ± 679.8g (465 to 4230g), respectively. The median age at the time of evaluation was 2.9 days (4 h to 27 days). The frequency of dermatological diagnoses was 100% and 98.5% of the sample had two or more, with an average of 4.67+1.53 dermatoses for each newborn. The 10 most frequent diagnoses were lanugo (85.9%), salmon patch (72.4%), sebaceous hyperplasia (68.6%), physiological desquamation (54.8%), dermal melanocytosis (38.7%), Epstein pearls (37.2%), milia (32.2%), traumatic skin lesions (24%), toxic erythema (16.7%), and contact dermatitis (5%). Those with GA< 28 weeks showed more traumatic injuries and abrasions, whereas those with ≥ 28 weeks had physiological changes more frequently, and those with GA between 34-366/7 weeks, had transient changes.
ConclusionDermatological diagnoses were frequent in our sample and those with higher GA showed a higher frequency of physiological (lanugo and salmon patch) and transient changes (toxic erythema and miliaria). Traumatic lesions and contact dermatitis were among the 10 most frequent injuries, reinforcing the need to effectively implement neonatal skin care protocols, especially in preterm.
The skin is a complex and fundamental organ for the transition between intra- and extra-uterine life, protecting newborns mechanically and immunologically, reducing insensible transepidermal water loss, providing transcutaneous absorption of substances, body thermoregulation and perception of the environment. Neonatal extrauterine exposure triggers numerous adaptation and maturation mechanisms, which accelerate after birth.1 The skin barrier depends on the stratum corneum maturation, which develops according to gestational age (GA).2
Around the 34th week of gestation, the skin is functionally mature but still can be easily damaged. The earlier the birth, the thinner and more immature the newborn's skin, resulting in greater sensitivity to manipulation and development of skin changes in preterm (PT) babies, which, in most cases, are benign and transient. However, it is important to differentiate these conditions from those associated with diseases or iatrogenic conditions.1,3
Researchers have been studying neonatal dermatological conditions for over 100 years. However, few authors have evaluated PT neonates and, when they were included, small samples were studied, mostly composed of late PT newborns or those not admitted to Neonatal Intensive Care Units (NICU).4,5
The prevalence of neonatal dermatological diagnoses ranges from 67.3% to 100%.6-8 The early recognition of skin diseases in the newborn can determine appropriate diagnosis, treatment, and counseling. Thus, this study aimed to determine the frequency of dermatological diagnoses in PT newborns and their associated perinatal factors.
Materials and methodsThis is a cross-sectional, analytical study with prospective data collection, which was conducted between November 2017 and August 2019 and evaluated 341 neonates. Our convenience sample was composed of all PT infants admitted to the rooming-in facility or the NICU of a university hospital. This study was approved by the Research Ethics Committee on Humans of the Clinical Hospital, with CAAE n 74272417.1.0000.0096.
PT newborns with up to 28 days of life, whose legal guardians signed informed consent forms, were included. Neonates whose guardians were unreachable due to absence, those who were discharged from the hospital, or who died before contact with the researchers were excluded.
The wide range, considering the entire neonatal period, is due to the attempt to include extremely premature infants, who would only be able to be evaluated after a few days or weeks of life. Also, there are neonatal dermatoses that appear after the first days of life and that are not evaluated in studies that include only the first 72 h of life, allowing the evaluation of early and late alterations.
Newborn skin evaluation occurred from Monday to Friday, but even those who were born on the weekend were included in the study. Their skin, mucous membranes, and skin appendages were examined while participants were in their cribs, naked, in a heated and appropriately lit environment. Diagnoses were clinically performed by one pediatric dermatologist and corroborated by the advisor. The diagnoses were signed when there was an agreement between the two researchers.
Participants’ medical records were reviewed via a standard research protocol for the name; gender; age (in hours); prenatal data (risk factors, serologies, use of maternal medications); birth data (GA, mode of delivery, need for resuscitation, Apgar score, weight, and weight to GA classification); and comorbidities (diagnoses before or after birth).
For statistical analysis, prenatal and neonatal characteristics were grouped in hours of life: < 450h and ≥ 450h; GA: < 28, 28 to < 32, 32 to < 34; and 34 to 366/7 weeks; 1- and 5-minute Apgar score: 1-6, 7-10, and unevaluated (in the absence of these data); birth weight: < 1000 g, 1000-1499 g, 1500-2499 g, 2500-4000 g; weight categories: small for GA (SGA), appropriate for GA (AGA), and large for GA (LGA).9-12
Central tendency and dispersion were expressed in means and standard deviation, whereas categorical variables, were in absolute and relative frequencies.
To estimate the difference between continuous and symmetric distribution variables, the Student's t-test was applied. For differences among nominal categorical variables, Pearson's chi-square tests with Yates correction were applied in 2 × 2 tables and, for tables other than 2 × 2 and linear trends for ordinal variables, Pearson's chi-square test was employed.
A backward multivariate logistic regression model was used to identify the variables associated with groups of dermatological diagnoses and with the 10 most frequent cutaneous lesions. The sample was estimated considering a minimum effect magnitude of 10%, a significance level of 5%, and a type II error of 5%. In the sample calculation, the assumptions of the multivariate binary logistic regression model with a minimum frequency of 5 cases for each variable studied and the proportion of 50 cases per variable included in the model were also considered.
The Backwise Multivariate Binary Logistic Regression model was applied to identify the variables associated with the categories of dermatoses and the 10 most prevalent dermatoses in the sample. In the models used, the tolerance coefficients were greater than 0.01 and the variance inflation factors were less than 10, indicating the absence of multicollinearity, that is, the absence of confounding factors, given the lack of significant correlation between the independent variables of the model.
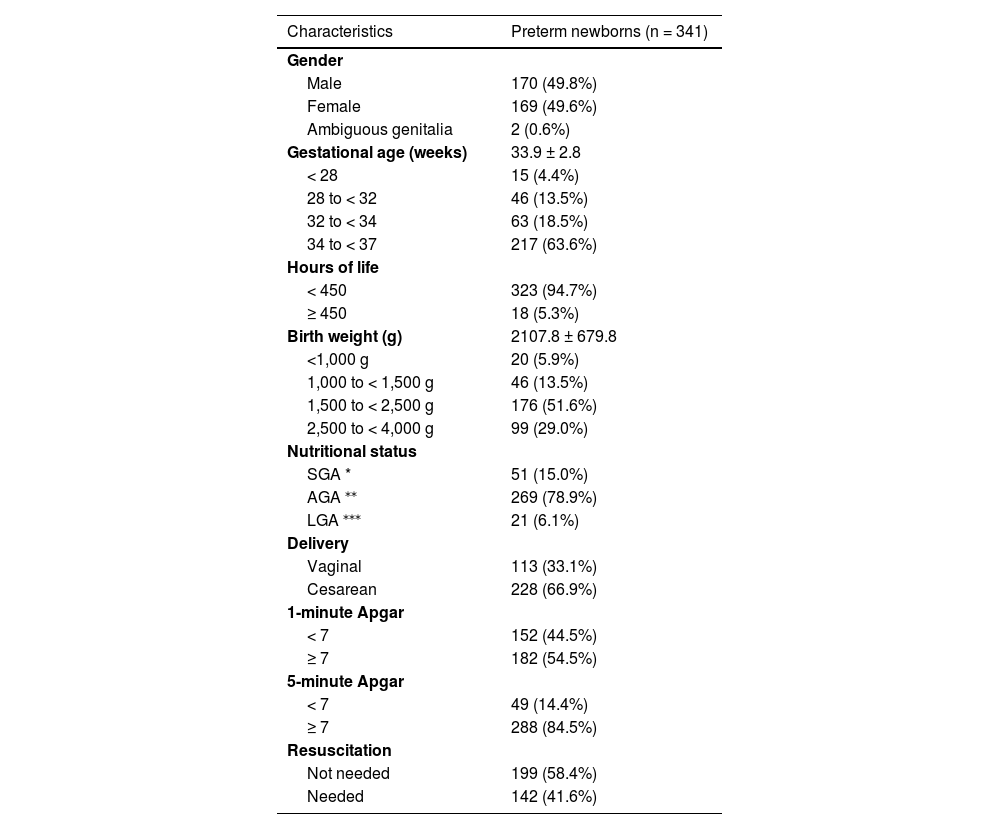
ResultsThe authors included 341 neonates from 576 premature infants born during the study period, 170 of which (49.8%) were boys and 169 (49.6%), were girls; and 2 (0.6%) had ambiguous genitalia. Of these, 61 (17.9%) had less than 32 weeks GA. The mean GA was 33.9 ± 2.8 weeks. Table 1 shows the remaining data.
Characteristics of preterm newborns.
| Characteristics | Preterm newborns (n = 341) |
|---|---|
| Gender | |
| Male | 170 (49.8%) |
| Female | 169 (49.6%) |
| Ambiguous genitalia | 2 (0.6%) |
| Gestational age (weeks) | 33.9 ± 2.8 |
| < 28 | 15 (4.4%) |
| 28 to < 32 | 46 (13.5%) |
| 32 to < 34 | 63 (18.5%) |
| 34 to < 37 | 217 (63.6%) |
| Hours of life | |
| < 450 | 323 (94.7%) |
| ≥ 450 | 18 (5.3%) |
| Birth weight (g) | 2107.8 ± 679.8 |
| <1,000 g | 20 (5.9%) |
| 1,000 to < 1,500 g | 46 (13.5%) |
| 1,500 to < 2,500 g | 176 (51.6%) |
| 2,500 to < 4,000 g | 99 (29.0%) |
| Nutritional status | |
| SGA * | 51 (15.0%) |
| AGA ⁎⁎ | 269 (78.9%) |
| LGA ⁎⁎⁎ | 21 (6.1%) |
| Delivery | |
| Vaginal | 113 (33.1%) |
| Cesarean | 228 (66.9%) |
| 1-minute Apgar | |
| < 7 | 152 (44.5%) |
| ≥ 7 | 182 (54.5%) |
| 5-minute Apgar | |
| < 7 | 49 (14.4%) |
| ≥ 7 | 288 (84.5%) |
| Resuscitation | |
| Not needed | 199 (58.4%) |
| Needed | 142 (41.6%) |
Participants’ mean birth weight was 2,107.8±679.8 g (465 to 4,230 g). Of these, 269 (78.9%) were AGA; 51 (15.0%), SGA; and 21 (6.1%) LGA. The authors found that 287 (84.1%) showed maternal comorbidity – mainly endocrine diseases (36.9%), such as diabetes and thyroid diseases; hypertensive syndromes (32%); and gynecological-obstetric conditions (16.4%), such as endometriosis, isthmic cervical insufficiency, and premature placental abruption.
Newborns’ median age at evaluation was 2.9 days (4 hours to 27 days). The authors found dermatological diagnoses in 100% of them. In total, 1.5% showed a single alteration, whereas 98.5%, two or more, with an average of 4.67 ± 1.53 dermatological diagnoses for each PT newborn.
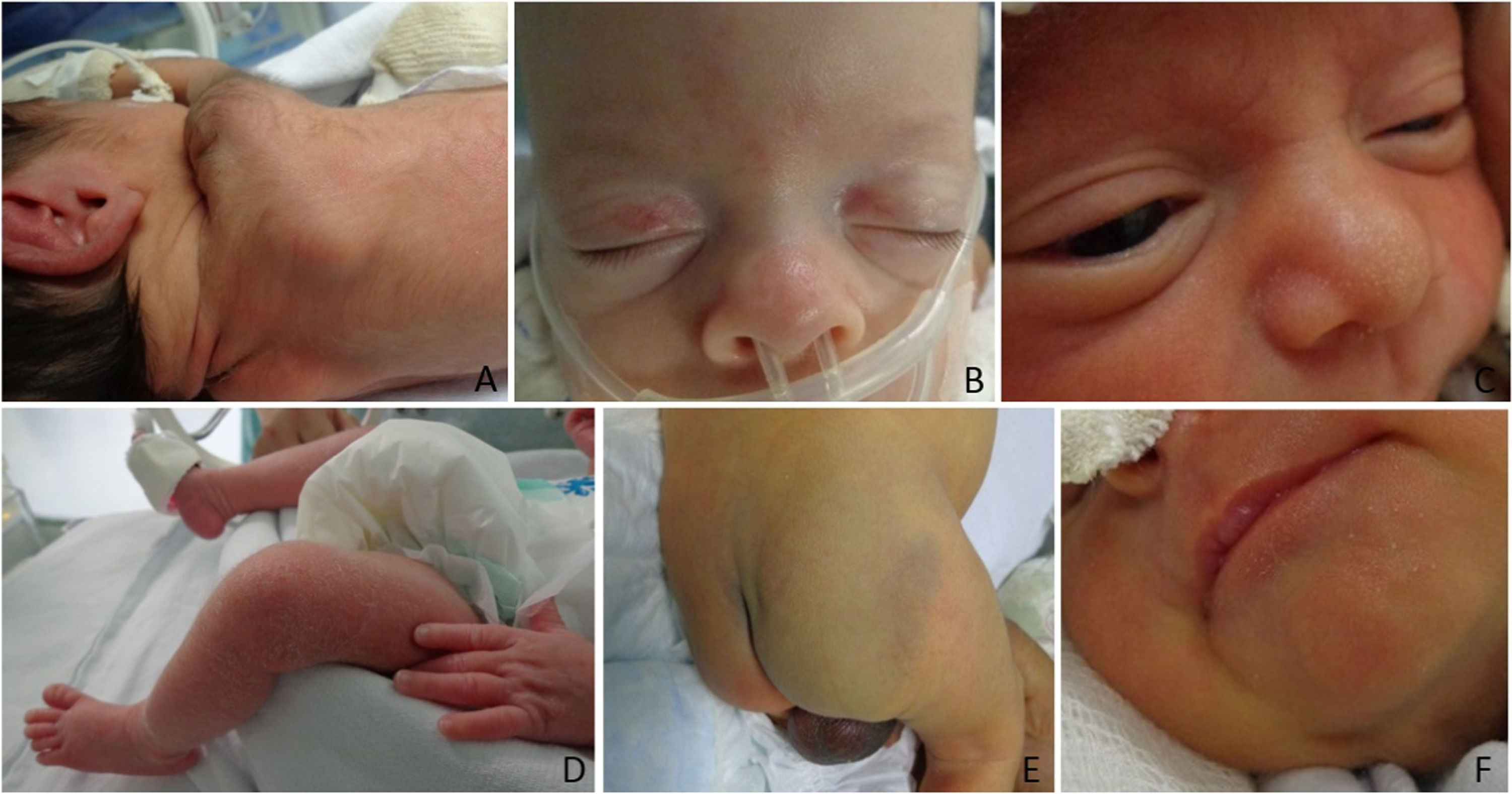
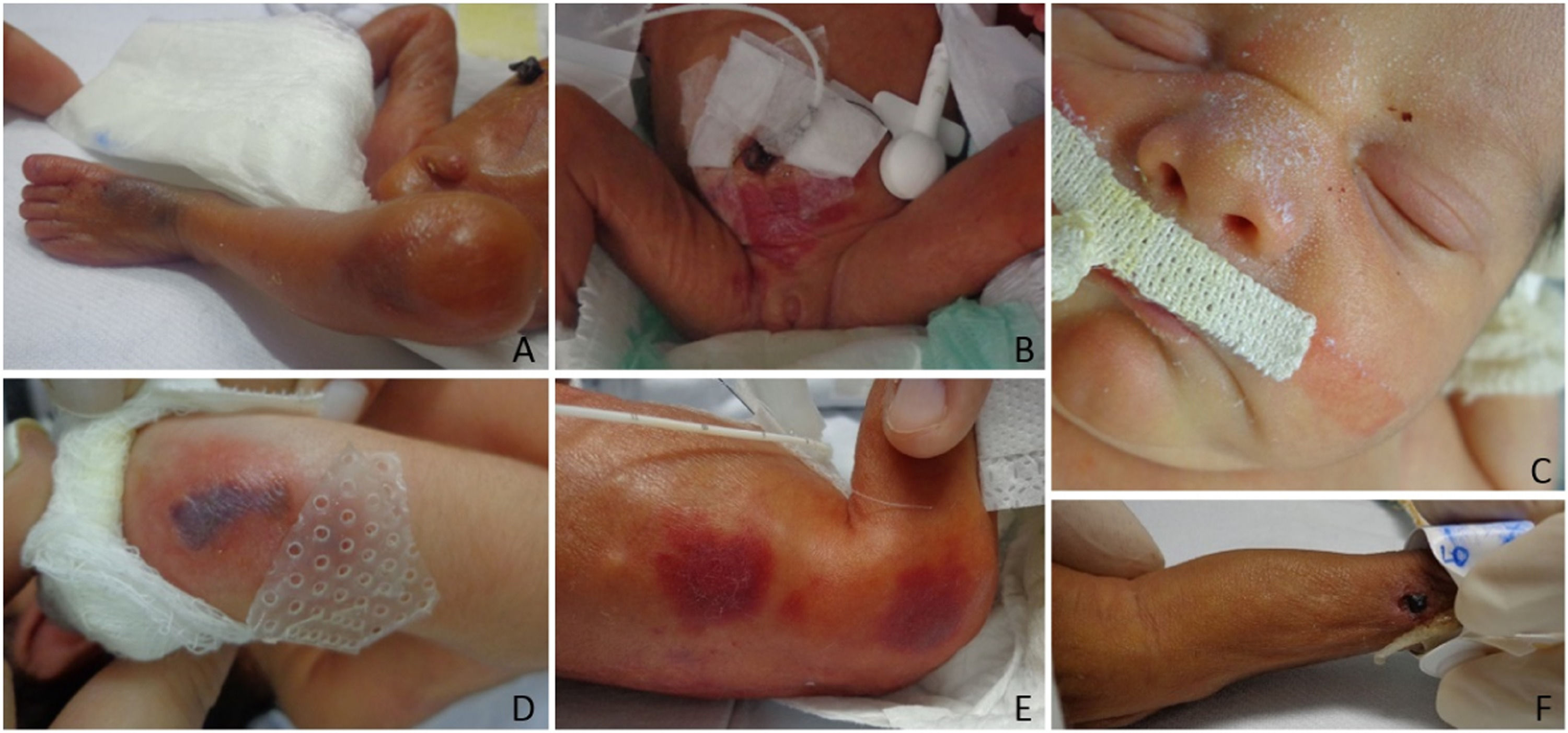
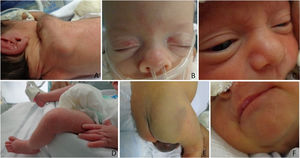
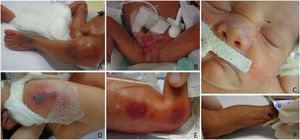
The authors classified diagnoses into physiological changes (physiological desquamation, Epstein pearl, Bohn nodule, sebaceous hyperplasia, salmon patch, dermal melanocytosis, milia, lanugo, vernix caseosa, and cutis marmorata), transient ones (neonatal toxic erythema and miliaria), birthmarks (melanocytic nevi, hypo or hyperchromic lesions, and nevus sebaceous), vascular ones (infantile hemangioma, capillary malformation, telangiectasias), traumatic wounds (abrasions and birth injuries), skin diseases (contact dermatitis, seborrheic dermatitis, intertrigo, candidiasis, and ichthyosis), and sacral dimple/myelomeningocele.
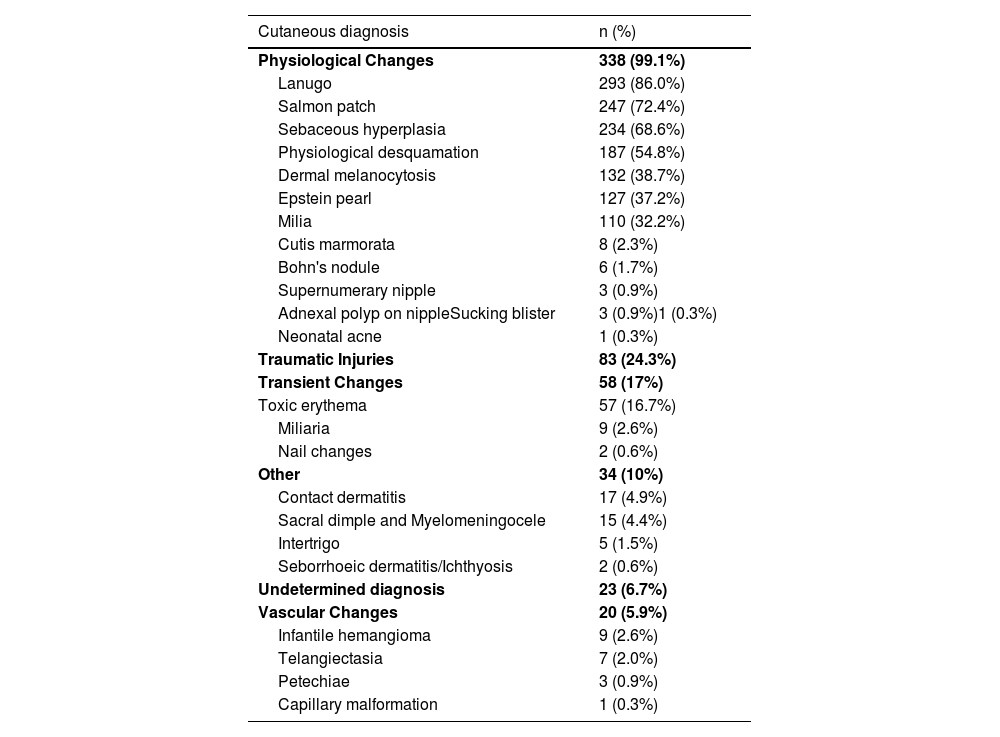
The 10 most frequent diagnoses found were lanugo (85.9%), salmon patch (72.4%), sebaceous hyperplasia (68.6%), physiological desquamation (54.8%), dermal melanocytosis (38.7%), Epstein pearls (37.2%), milia (32.2%), traumatic cutaneous lesions (24%), toxic erythema (16.7%) and contact dermatitis (5%). The authors observed capillary malformations, miliaria, adnexal polyp in the nipple, Bohn nodules, intertrigo, infantile hemangioma, cutis marmorata, supernumerary nipples, among others less frequently. Table 2 shows newborns’ diagnoses and their frequencies.
Frequency distribution of newborns’ cutaneous diagnosis.
Physiological changes occurred in all GA ranges, most frequently in neonates with a ≥ 28-week GA (p < 0.001). The authors observed transient changes in newborns with a GA between 34-366/7 weeks (p < 0.001), and skin traumas and dermatopathies in those with < 28 weeks (p < 0.01) (Figs. 1 and 2). The authors found no difference in GA ranges for the frequencies of the other groups (p > 0.05).
The authors observed milia, toxic erythema and Epstein pearls more frequently in neonates with a ≥32-week GA (p < 0.01); trauma and abrasions, in those with a ≥ 28-week GA (66.7% vs 19.6% vs 20.6% vs 23.5%, p < 0.001); sebaceous hyperplasia and salmon patch, in those with a ≥ 28-week GA (p < 0.01 and p < 0.001); and infantile hemangioma in newborns with a < 32-week GA (p < 0.001).
The authors found a significant difference in the occurrence of sebaceous hyperplasia in the first 168 hours of life and after this period (74.3% vs. 52.3%, p < 0.001). Moreover, physiological desquamation was more frequent after 168 hours (48.6% vs. 72.7%, p < 0.001). Sebaceous hyperplasia occurred almost three times more frequently in the first hours of life (OR = 2.64, 95% CI 1.59-4.37, p < 0.001), as did physiological desquamation (OR = 1.49, 95% CI 1.18-1.87, p < 0.001). The authors found Epstein pearls and milia more frequently in the first 168 hours of life (OR = 1.70, 95% CI 1.00-2.88, p = 0.04; OR = 1.73, 95% CI 0.99-3.01, p = 0.04).
Vascular alterations, dermatopathies and traumatic cutaneous lesions were more frequent in neonates weighing < 1,000 g (p < 0.001). The authors found traumatic injuries, abrasions, or bruises in 55.0% of those with < 1000 g; 19.6%, between 1000 and < 1500 g; 21.0%, between 1,500 and < 2,500 g; and 26.3%, in newborns with ≥ 2500 g (p < 0.01). Infantile hemangioma was most frequent in those with < 1000 kg (p < 0.001).
Toxic erythema was more frequent in those who had vaginal births (OR=2.25, 95% CI 1.25-4.06, p = 0.01) and transient dermatological alterations, in boys (OR = 1.92, 95% CI 1.10-3.35, p = 0.02) and in their first hours of life (OR = 1.89, 95% CI 0.94-3.82, p = 0.02).
In Figure 3 (supplementary material) there are graphs illustrating the frequency of the 10 most frequent dermatoses per gestational age, gender, hours of life and birth weight.
DiscussionThe authors found neonatal dermatological diagnoses in 100% of PT neonates. In total, 98.5% had two or more, averaging 4.53 dermatological diagnoses. A Brazilian study4 with 2,530 newborns showed that 95.8% had dermatological diagnoses, averaging 3.23 dermatological findings. According to Moosavi,6 an Iranian study with 1,000 neonates, evaluated in the first 48 hours of life (158 preterm), showed that 96% of its sample had dermatological diagnoses; and 84.5%, had two or more. A Uruguayan study,8 conducted in the first 72 hours of life, assessed 2,811 participants (122 preterm), showing that 100% had dermatological diagnoses and 99.8% (2,795), two or more.
Lanugo was the most frequent dermatological diagnosis in this study (86%), a lower frequency than the 98.3% (2,763) in De María et al.,8 but higher than other studies,4,5,7 in which it ranges from 2.8 to 38.9%. This variation may occur due to specific characteristics of the population evaluated, in the present study, this finding was more frequent since the authors exclusively evaluated PT neonates.
The authors found a higher frequency of salmon patch (72.4%) than other studies, which ranged from 12.8 to 55.6%,4,6-8 but lower than the 83% of the American study by Kanada et al.,13 which evaluated 594 neonates, 71 of whom were preterm; was twice as frequent in those who had vaginal births (p = 0.04). Some authors have associated the term GA and ethnic factors with salmon patch occurrence. This information does not reflect our findings, which may indicate just an ethnic relation since the authors only evaluated PT infants and found a frequency similar to that found by authors who predominantly evaluated term newborns.
Sebaceous hyperplasia (68.6%) and milia (54.8%) occurred more frequently in ≥ 32-week GA newborns (p < 0.001). Moosavi6 found a relation between sebaceous hyperplasia (affecting 43.7% of their sample) and NB's maturity. Gorur,5 who evaluated 1,260 NB, of which 66 were preterm, reported a 25% prevalence, also associated with full-term birth and consequent skin maturity, whereas De María et al.8 found a 73.3% prevalence. This study found a relation between sebaceous hyperplasia and time of life at the time of evaluation. It occurred more frequently in the first seven days of life (p < 0.001) and, after the multivariate logistic regression, it was almost three times more frequent in this period (p < 0.001).
Previous studies associated milia with skin maturity, which ranged from 7.5 to 38.7%,4-6,8,14 results lower than our 54.8%. This indicates that this skin change may occur even in PT neonates.
Epstein Pearls affected 37.2% of neonates, a lower frequency than in Moosavi6 and De María et al.8 (70.2% and 66.3%, respectively). During the dermatological examination of intubated participants, it was impossible to properly explore their oral cavity, which may explain the lower frequency the authors obtained. It may also be associated with GA since the premature infants evaluated in Moosavi6 and De María et al.8 represented a small percentage of their sample.
Epstein pearls and milia were almost twice more frequent in participants’ first seven days of life (p = 0.04), probably due to the characteristic evolution of epidermal inclusion cysts, which tend to spontaneous resolution over the course of days.
Physiological desquamation affected 54.8% of PT neonates and was more frequent after seven days of life (p < 0.001). The authors found a greater frequency than other studies, ranging from 1.9 to 23.3%,4,6,7 but lower than in De María et al. (79.7%). This wide variation directly depends on study designs. The authors included older newborns than in other studies – which often evaluate newborns in their first 48 or 72 hours of life.
Neonatal toxic erythema (16.7%) was more frequent in newborns with a ≥ 34-week GA (p < 0.001), whereas Moosavi6 and De María et. al.8 found it in 11.1 and 55.3% of their sample, respectively. The frequency the authors found is identical to that in Monteagudo et al.,15 which evaluated 1,000 healthy newborns in northern Spain (of which 75 were preterm) in their first 72 hours of life. This dermatological diagnosis occurs in the first days of life and, besides ethnic factors and delivery mode, newborns’ age during evaluation also interferes with its identification. It was twice more frequent in those who were born vaginally (p = 0.01). Previous studies have identified the same association,15,16 a relation with cesarean sections,7 and no association with delivery mode.4 These studies evaluated a small number of PT neonates. Although some relate it to skin maturity and term birth, this study shows a similar frequency to studies with a majority of term newborns in their sample.5-7,15
Infantile hemangiomas were more frequent in neonates with a < 32-week GA (p < 0.001). Moosavi et al.6 found it in 1.3% of neonates. De María et al.,8 in 0.5% of their sample. The authors found a 2.6% prevalence. Prematurity is a risk factor for the development of Infantile hemangioma and so we expected a higher value than that found in other studies.
Traumatic cutaneous lesions, abrasions or bruises, and contact dermatitis were more frequent in < 28-week newborns (p < 0.001). They occurred in 29.3%, a higher frequency than the 18% in Csoma et al.17 (mean 34.5 ± 4.3-week GA), the 12.8% in De María et al.8 (mean 38.8±1.33-week GA), and the 2% in Reginatto et al.4 (mean 38.4-week GA). The fragility of the immature skin justifies the high frequency found and, the more PT, the more frequent the lesions caused by NICU care. Although caution in neonates’ daily handling and care is already established, extremely preterm infants require even more attention.
The use of antiseptics, materials for fixing devices (probes, catheters, and cannulas), sensors to monitor vital signs (body temperature and oximetry), venous and/or arterial puncture, exposes the neonate to mechanical, thermal, and chemical trauma, immediately breaking their skin barrier and generating consequences, such as medical adhesive-related skin injuries18 and long-term sequelae in case of lesions which form scars.19
Physiological changes were more frequent in newborns with a ≥ 28-week GA (p < 0.001) and transient ones, in those between 34-366/7 weeks (p < 0.001). They are probably associated with skin maturity and its appendages, the closer to term, the greater the occurrence of these dermatological diagnoses.
The sample loss occurred due to the difficulties found in the NICU environment, either obtaining the consent form, since the guardians did not remain in the place full time, also depended on the visiting hours (in which the guardians were not always present); it is common for the puerperal woman to be absent to express breast milk, among others; as well as in many cases there was no clinical stability for the evaluation of these neonates.
A limiting factor of this study was the time when the evaluation was performed, since according to the type of dermatosis observed, there is a relationship between its beginning, end, and hours of life. Some occur more frequently in the first hours and others in the first weeks. Thus, a single evaluation results in limited data on transient conditions.
Another limitation is our difficulty in comparing our findings with other studies on newborns’ skin, most of which evaluated healthy babies, excluding those PTs who required NICU. The different adopted methodologies and lack of consensus to classify the evaluated dermatological diagnoses also restrict our results.
There are few studies that evaluated neonatal dermatosis according to GA subgroups, hours of life, and birth weight as in the present study. As well as the dermatological evaluation of newborns, which is commonly performed by several professionals, in this study it was performed by a single evaluator and corroborated by the advisor and team of the Pediatric Dermatology service.
This study included a significant number of PT newborns (a high-risk population that studies on neonatal dermatological diagnoses often exclude), covered the entire neonatal period and observed neonates’ dermatological diagnoses profile. Neonatal dermatological diagnoses in PT infants occur frequently and some frequencies differ from term newborns.
Skin changes occurred in 100% of PT newborns. Traumatic injuries predominated in < 28-week; physiological changes, in those ≥ 28 weeks; and transient ones, in those 34-366/7 weeks of GA.
Degree of prematurity and time of life during the evaluation were the main factors determining skin changes in this population.
This work was supported by the Coordination for the Improvement of Higher Education Personnel (CAPES) Master's Scholarship.
Work-related service: Hospital de Clínicas da Universidade Federal do Paraná, setor de Ciências da Saúde, Curitiba, Paraná, Brasil.