To evaluate if there are differences regarding disease location and mortality of necrotizing enterocolitis, according to the gestational age at birth, in newborns submitted to surgery due to enterocolite.
MethodsA historical cohort study of 198 newborns submitted to surgery due to necrotizing enterecolitis in a tertiary hospital, from November 1991 to December 2012. The newborns were divided into different categories according to gestational age (<30 weeks, 30–33 weeks and 6 days, 34–36 weeks and 6 days, and ≥37 weeks), and were followed for 60 days after surgery. The inclusion criterion was the presence of histological findings of necrotizing enterocolitis in the pathology. Patients with single intestinal perforation were excluded.
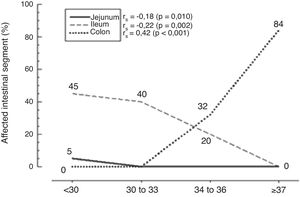
ResultsThe jejunum was the most commonly affected site in extremely premature infants (p=0.01), whereas the ileum was the most commonly affected site in premature infants (p=0.002), and the colon in infants born at term (p<0.001). With the increasing gestational age, it was observed that intestinal involvement decreased for the ileum and the jejunum (decreasing from 45% to 0% and from 5% to 0%, respectively), with a progressive increase in colon involvement (0% to 84%). Total mortality rate was 45.5%, and no statistical difference was observed in the mortality at different gestational ages (p=0.287).
ConclusionsIn newborns submitted to surgery due to necrotizing enterocolitis, the disease in extremely preterm infants was more common in the jejunum, whereas in preterm infants, the most affected site was the ileum, and in newborns born close to term, it was the colon. No difference in mortality was observed according to the gestational age at birth.
Avaliar se há diferença de localização e de mortalidade da enterocolite necrosante de acordo com a idade gestacional ao nascimento, em neonatos operados por enterocolite.
MétodosCoorte histórica de 198 neonatos operados por enterocolite necrosante em hospital terciário, de novembro de 1991 a dezembro de 2012. Os recém-nascidos operados foram divididos em diferentes categorias de acordo com a idade gestacional (<30 semanas, 30 a 33 semanas e 6 dias, 34 a 36 semanas e 6 dias e ≥37 semanas), e foram seguidos por 60 dias depois da cirurgia. O critério de inclusão foi a presença de achados histológicos de enterocolite necrosante no anatomopatológico, e de exclusão foi a presença de perfuração intestinal única.
ResultadosO jejuno foi mais acometido pela ECN nos prematuros extremos (p=0,01); o íleo mais afetado nos recém-nascidos prematuros (p=0,002), e o cólon nos recém-nascidos a termo ou próximos ao termo (p<0,001). Com o aumento da idade gestacional, observa-se redução do acometimento do jejuno e do íleo (regrediu de 45% para 0% e de 5% para 0%, respectivamente), e aumento progressivo do acometimento do cólon (0% para 84%). A mortalidade total das crianças operadas por ECN foi de 45,5%; não existiu diferença estatística na mortalidade nas diferentes idades gestacionais (p=0,287).
ConclusõesEm recém-nascidos operados por enterocolite necrosante, a doença no jejuno foi mais comum no prematuro extremo, no íleo no prematuro, e a doença no cólon nos recém-nascidos próximos ao termo. Não foi observada diferença de mortalidade de acordo com a idade gestacional ao nascimento.
Necrotizing enterocolitis (NEC) is a disease that especially affects preterm infants and those with low birth weight (<2500g).1 Studies show the existence of an inverse association between mortality and birth weight.2 The incidence of NEC is variable, depending on the geographic area, ethnicity, and genetic and environmental factors; despite the improved care and improved survival of an increasing number of preterm infants, its incidence remains high in some countries.3 NEC is the most devastating gastrointestinal disease in neonates, occurring in 0.7 per 1000 patients, and in up to 7% of those hospitalized in neonatal intensive care units (NICUs), with a mortality ranging from 20% to 50%.4–6 There is still a controversy regarding the disease's etiology, pathophysiology, and the importance of all possible causal factors.
During the 1980s, before the age of surfactant's and improved neonatal care, studies on NEC showed that the patients most severely affected by the disease were preterm infants between 33 and 35 weeks of gestation.7,8 However, more recent studies have shown a higher frequency of the disease in preterm infants born at 29–31 weeks of gestation.8
In addition to the increasing survival of preterm infants, studies also show that the disease's etiology may differ between preterm and full-term infants. In the latter, the heart diseases and other conditions that predispose to intestinal ischemia seem to be more associated with the disease than the intestinal immaturity and greater permeability to the bacteria and food substrates observed in preterm infants.
Several studies have attempted to elucidate the etiology of NEC,9,10 and others have investigated potential prevention and treatment strategies.11,12 Few studies have attempted to associate gestational age to the intestinal segment most affected by the disease. The present study sought to identify an association between disease location and the gestational age at birth in newborns submitted to surgery due to NEC.
Materials and methodsNewborns submitted to surgery due to NEC (historical cohort) at Hospital da Criança Conceição, Grupo Hospitalar Conceição, Porto Alegre, state of Rio Grande do Sul, Brazil, between November 1991 and December 2012 were evaluated. Inclusion criteria were the histological confirmation of NEC in patients submitted to laparotomy and resection of intestinal loops, such as ischemia, necrosis, or perforation, as well as the histological findings of coagulation necrosis, inflammation, tissue repair alterations, or the presence of intestinal pneumatosis in the anatomopathological examinations. Cases of single perforation associated with prematurity were excluded. Eligible patients were followed for 60 days regarding the outcomes of death, survival, and presence of complications associated with the disease and the surgery.
The patients were divided into four categories according to gestational age: <30 weeks; 30–33 weeks and 6 days; 34–36 weeks and 6 days, and ≥37 weeks. Gestational age was measured according to the date of the last menstrual period, ultrasonography findings, and according to the methods of Ballard et al.13 (preterm infants <32 weeks) and Capurro et al.14
Exploratory laparotomy was indicated in cases of pneumoperitoneum, deterioration of the clinical status after peritoneal cavity drainage, positive paracentesis (presence of stool or bacteria in the peritoneal fluid of patients with two relative indications of surgery), or presence of three or more relative indications of laparotomy.5 Relative indications were: fixed abdominal mass, clinical deterioration (hypotension, poor perfusion and skin pallor, respiratory function worsening, thermal instability, tachycardia, and worsening of laboratory tests), abdominal wall cellulitis, presence of a fixed abdominal loop in a series of abdominal X-rays, diffuse intestinal pneumatosis (present in the four quadrants), and air portogram.5
Data were stratified according to gestational age and analyzed using the statistical software SPSS (IBM SPSS Statistics for Windows, version 19.0, NY, USA). Comparisons between groups were performed using the chi-squared test, Fisher's exact test, and analysis of variance (ANOVA). Tukey's post hoc test was utilized when ANOVA was used to evaluate the statistical difference between the groups. Spearman's coefficient was utilized to demonstrate the correlation between gestational age and the most affected intestinal segment. A p-value <0.05 was considered significant.
The study was approved by the Research Ethics Committee of the institution (study number 09/079), and was carried out according to the rules of the Declaration of Helsinki. Informed consent was obtained from the patients’ parents or guardians prior to surgery.
ResultsA total of 198 newborns submitted to laparotomy due to NEC, with a median gestational age of 32 weeks (24–42), were evaluated. The NICU where the study was performed receives on average 670 patients a year, with an incidence of NEC ranging from 2.7% to 4.3% in the study years, requiring surgical treatment in 27% to 50% of patients.
Birth weight showed a mean of 1535±697g (range: 555–4160g). The patients were distributed according to the gestational age: 36 (18.1%) were born at <30 weeks, 79 (39.9%) between 30 and 33 weeks, 55 (27.7%) between 34 and 36 weeks, and 28 (14.4%) at ≥37 weeks. The demographic and clinical characteristics of the studied population are shown in Table 1.
Demographic and clinical characteristics of patients submitted to surgery due to necrotizing enterocolitis, according to gestational age.
| Variable | Total | Gestational age (weeks) | p-value | |||
|---|---|---|---|---|---|---|
| n=198 | <30 | 30–33 | 34–36 | ≥37 | ||
| n=36 | n=79 | n=55 | n=28 | |||
| Gender (M/F) | 109/89 (55.1/44.9) | 17/19 (47.2/52.8) | 50/29 (63.3/36.7) | 26/29 (47.3/52.7) | 16/12 (55.1/42.9) | 0.217a |
| Ethnicity (W/nW) | 159/39 (80.3/19.7) | 31/5 (86.1/13.9) | 60/19 (75.9/24.1) | 45/10 (81.8/18.2) | 23/5 (82.1/17.9) | 0.616a |
| Vaginal delivery | 109 (55.1) | 20 (57.1) | 43 (59.7) | 31 (57.4) | 15 (53.6) | 0.9481a |
| BW (g) | 1535±697 | 905±184a | 1309±445b | 1653±405c | 2754±697 | <0.001b |
| SGA | 72 (36.4) | 10 (27.8) | 30 (38.0) | 25 (45.5) | 7 (25.0) | 0.193a |
| Neonatal asphyxia | 58 (29.3) | 9 (25.0) | 29 (37.2) | 17 (31.5) | 3 (10.7) | 0.059a |
| Apgar at the 5th min. | 8.0 (1–10) | 7.4 (4–10) | 7.8 (2–10) | 8.4 (1–10) | 8.6 (4–10) | 0.001b |
| UA catheterization | 77 (38.9) | 32 (88.9) | 40 (50.6) | 4 (7.3) | 1 (3.6) | <0.001a |
| Heart disease | 24 (12.1) | 12 (33.3) | 6 (7.6) | 1 (1.8) | 5 (17.9) | <0.001c |
| Associated disease | 73 (36.9) | 12 (57.1) | 40 (58.0) | 13 (26.5) | 8 (32.0) | 0.002a |
| HMD | 80 (40.4) | 32 (88.9) | 41 (51.9) | 7 (12.7) | 0.0 | <0.001a |
| Short intestine | 17 (8.6) | 2 (5.6) | 5 (6.3) | 8 (14.5) | 2 (7.1) | 0.332c |
| Death | 90 (45.5) | 18 (50.0) | 33 (418) | 22 (40.0) | 17 (60.7) | 0.258a |
Data are shown as mean±standard deviation, median (IQR: P25–P75) or percentage (%).
M, male; F, female; W, white; nW, non-white; BW, birth weight in grams; SGA, small for gestational age; min., minute; UA, umbilical artery; HMD, hyaline membrane disease.
It can be observed that only the extremely preterm children, with less than 30 weeks gestation, had jejunal involvement, at a median of 5% of intestinal involvement, with a statistically significant difference observed when compared with the other gestational age groups (p=0.010), as shown in Fig. 1.
In preterm infants, it was observed that the ileum was the intestinal segment most affected by the disease (p=0.002), as shown in Fig. 1. The extent of the ileum affected by the disease was 45% in neonates born at <30 weeks, 40% in those born between 30 and 33 weeks and 6 days, and 20% disease extension in those born at 34–36 weeks and 6 days. It was observed that the closer to the term, the greater the involvement of the diseased colon (p<0.001), as seen in Fig. 1. An 84% extension of the affected colon was observed in children born at ≥37 weeks, and 32% of the affected colon in preterm infants born at 34–36 weeks and 6 days, with no disease in this segment in the other gestational age ranges. A weak correlation was observed between jejunal disease and gestational age at birth, a correlation that became moderate in the ileum and quite significant when the presence of the disease in the colon was evaluated, with the highest strength of association (Fig. 1).
Of the total cohort, 90 (45.52%) patients died during the study. No statistical difference was observed in relation to the deaths between the different gestational ages (p=0.287). NEC was fatal in 18 (50%) neonates at <30 weeks, in 33 (41.8%) born between 30 and 33 weeks, in 22 (40%) born between 34 and 36 weeks, and in 17 (60.7%) born at ≥37 weeks.
DiscussionNEC remains the most frequent surgical emergency in the NICU, due to the increased survival of extremely preterm infants, intensive care improvement, and the use of total parenteral nutrition and pulmonary surfactant.
The most affected patients in this study were preterm infants, which in the literature represent 90% of the patients submitted to surgery due to NEC.1,15 Differences between the groups studied in the Fig. 1 comparison of some clinical and epidemiological characteristics of newborns submitted to surgery due to NEC were observed, but they were due to the difference in the patients’ gestational age, and not due to NEC.
This study showed that of the newborns submitted to surgery due to NEC, the extremely preterm infants had significant jejunal involvement, whereas the preterm infants showed ileal involvement, and in the full-term or near full-term infants, the disease was more common in the colon. This difference observed in the site of NEC may be due to the different disease mechanisms, especially regarding the importance of risk factors in full-term or near full-term infants.16
The exact pathophysiology of NEC is not fully understood, and it has not been defined whether a single initial event is responsible for the activation of the cascade of events that culminates with the intestinal lesion, or whether the interaction of several factors is responsible for disease evolution. Disagreements still exist regarding whether the hypoxic/ischemic event is primary or secondary, or if both are initial factors or are the results of insults.
After birth, the intestinal tract is exposed to many types of bacteria, and the diet, be it formula or breast milk. In preterm infants, the intestine must maintain its growth and development until it reaches the length and maturity found in full-term infants. They have increased metabolic demands, which make the intestine even more vulnerable to direct insults to the intestinal wall, as well as bacterial invasion and difficulties in intestinal wall repair and regeneration. This fragility of the intestinal wall and its increased permeability,17,18 associated with reduced motility and consequent stasis and the repair difficulties after insults to the intestinal wall, make the preterm infant more susceptible to NEC; however, no study has clearly defined whether there are differences regarding the site most often affected by the disease, either the ileum or the colon.19,20
The most common site of NEC occurrence is the terminal ileum and the proximal colon, sites with lower vascularity, suggesting a mismatch in the circulatory system that could be responsible for the disease. Studies on NEC pathogenesis show the importance of ischemia, with the hypoxic/ischemic insult acting directly on the intestinal mucosal damage, breaking the intestinal barrier and promoting bacterial invasion, culminating with the inflammatory cascade activation.
Ischemic insults are more relevant for NEC onset in full-term newborns, with ischemia being the initial event for the triggering of the inflammatory cascade. In preterm infants, in contrast, the probable disease mechanism would be intestinal immaturity, which causes susceptibility to NEC due to reduced intestinal wall protection and repair.17,18
Although this study's postoperative mortality agrees with the literature,2,5 no difference in mortality according to gestational age at birth was observed. The high mortality in neonates born at ≥37 weeks, although not statistically different from the other groups, may have been due to other severe associated comorbidities, or due to delayed NEC diagnosis and treatment onset.
This study has limitations, as it was performed in a single center and featured a historical cohort. Other studies with larger samples, or even multicenter ones, could corroborate these findings that in neonates submitted to surgery due to NEC, gestational age influences the specific intestinal segment affected by the disease. This could further contribute to better clarify the disease's pathophysiology and the importance of the causal factors in its genesis.
The present study showed that the disease location in newborns submitted to surgery due to NEC was different according to the gestational age. Jejunal involvement was more common in extremely preterm infants, whereas the ileal involvement was more significant in preterm infants, and the colon in full-term or near full-term infants. The mortality of these newborns submitted to surgery due to NEC was not different according to the gestational age at birth.
Conflicts of interestThe authors declare no conflicts of interest.
Please cite this article as: Feldens L, Souza JC, Fraga JC. There is an association between disease location and gestational age at birth in newborns submitted to surgery due to necrotizing enterocolitis. J Pediatr (Rio J). 2018;94:320–4.