In previous studies, smaller renal volumes were reported in prematurely born infants, however, these renal volumes were not corrected for body surface area, the main determinant of renal size. Given the rapid growth of the renal cortex after premature birth, the authors hypothesized that corrected volumes would not differ from healthy controls.
MethodsAmbispective cohort study with prospective follow-up of prematurely born babies in a large specialized center and retrospectively recruited healthy control group. Children were assessed for renal length and renal volumes at age 5 by three independent ultrasonographers. Detailed anthropometry, blood pressure and renal function were also obtained. Age independent z-scores were calculated for all parameters and compared using descriptive statistics.
ResultsEighty-nine premature study participants (median 32 weeks gestational age) and 33 healthy controls (median 38 weeks gestational age) were studied. Study participants did not differ in age, sex, Afro-Colombian descent, height, blood pressure, serum creatinine, or new Schwartz eGFR. Premature study participants had a significantly lower weight (17.65 ± 2.93 kg) than controls (19.05 ± 2.81 kg, p = 0.0072) and lower body surface area. The right renal volumes were significantly smaller (39.4 vs 43.4 mL), but after correction for body surface area, the renal volume and renal length z-scores were identical for both kidneys (mean right kidney -0.707 vs -0.507; mean left kidney -0.498 vs -0.524, respectively).
ConclusionRenal volumes need to be corrected to body surface area. After correction for body surface area, 5-year-old healthy and prematurely born children have comparable renal volumes.
Many metazoan organs are comprised of highly branched tubular structures, for instance, the lungs and the kidney.1 They are developed through a process called branching morphogenesis. Branching organs have a finite endowment of organoids at a given developmental age, and as for the kidneys, this process is largely completed by 36 weeks of gestation in humans.2 Genetic factors such as Adamts18 metalloprotease and many others as well as environmental factors modify nephron endowment.3 Prematurity is known to affect nephron endowment. It is believed that premature delivery results in an interruption or at least an alteration of the normal nephrogenesis which would normally continue to the 36th week of gestation.4 A recent study suggests that some nephrogenesis can still occur after premature delivery, but the resulting glomeruli are far fewer than normal and abnormal.5 A key feature of prematurity is the low birth weight (LBW). Research in England and Wales showed the impact of LBW on mortality in later childhood and adolescence.6 In Colombia, 1 of 10 newborns have a preterm birth. LBW accounts for 7.7% of all newborns of which 50% are preterm newborns.7 As such, LBW is a key feature of preterm birth. Low birth weight is defined as a birth weight < 2,500 g.
Unfortunately, there is currently no reliable methodology to measure nephron endowment. Serum creatinine, the most widely used biomarker of glomerular function, has many pitfalls and is affected by maternal renal function for at least one week.8 Abitbol et al. suggested using renal volume instead.9 It is known that after premature delivery, renal cortex volume and thus total renal volume increases rapidly.10 This is believed to occur due to glomerular hyperfiltration as a consequence of nephron underdosing.11 On the other hand, it has been shown that renal volumes are decreased later in life when compared to healthy age-matched controls, at ages 7 and 1112 and 20. Unfortunately, these studies did not correct renal volume for the body surface area.13 In a recent paper on the renal volume of prematurely born infants, the difference in the renal volumes no longer was significant after correcting for body surface area.14 As this fact would invalidate the previously reported lower renal volumes as a consequence of prematurity, the authors analyzed the question in a prospective cohort study of prematurely born infants and healthy controls. The authors hypothesized that renal volumes would not be different between prematurely and healthy born infants.
MethodsSetting and study populationCali is a large city of 2.3 million inhabitants and the capital of the Valle del Cauca Department in Southwestern Colombia. Cali's natality rate was 12 per 1000 habitants in 2017 with an infant mortality rate of 9 per 1000 life newborns, and 11% LBW.15
Kangaroo Mother House Alfa is a comprehensive program of medical care for preterm children in Cali. This program follows children during their first year of life. Mother baby dyads are referred after hospital discharge and receive a comprehensive follow-up which is led by nurses. Fundación Valle del Lili is a tertiary care university hospital with a large catchment area covering all of Southwestern Colombia. The pediatric nephrology program is mid-size with approximately 10 renal transplants and 3000 clinic visits per annum.
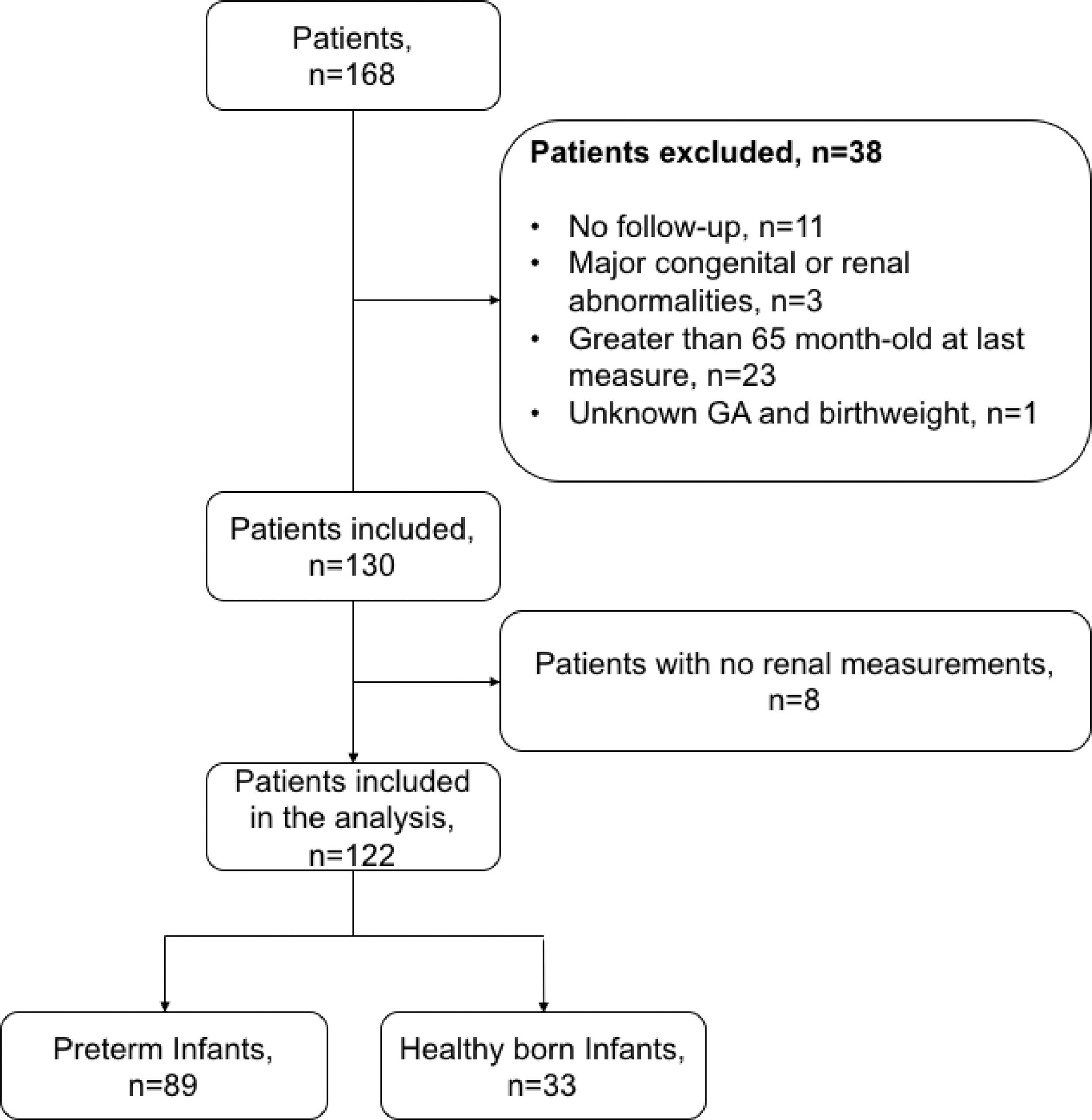
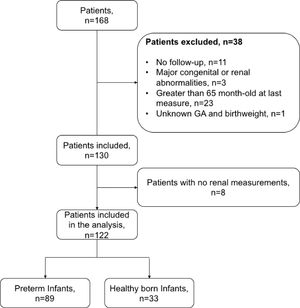
Study design, inclusion criteria and data collectionThis is an ambispective (retrospective and prospective) study. The exposed cohort was five-year-old children with a gestational age <36 weeks who were included in the kangaroo program when they were infants less than 2 months of age. The authors excluded children with major congenital or renal abnormalities and/or loss of contact. The enrolled children had their first visit to the kangaroo program between 2006 and 2012. Follow-up was collected at 5 years of age (±3 months). The authors obtained a non-exposed (NE) cohort (healthy children with birth weight greater than 2500 grams and gestational age >36 weeks) from different schools around the city. This study is in accordance with the Declaration of Helsinki and was approved by the Fundación Valle del Lili ethics board committee. Informed written consent was obtained from parents in each case.
Exposure variablesWeight at birth and gestational age were recorded from the clinical chart of the child, as well as when he/she entered the Kangaroo program. The estimated gestational age (GA) was determined from an ultrasound (US) during the first 20 weeks of gestation. If that was not available, the GA was calculated from the most recent US prenatal ultrasound. Finally, if no prenatal ultrasound was performed, the GA was estimated using the last menstrual period and neonatal examination.16 Both weights at birth and gestational age were captured in the Kangaroo program administrative database, with other demographical and clinical variables.
Outcome variablesThe main outcome variables were renal ultrasound dimensions at 5 years of age. Each child had one renal US study for renal dimensions (with a General Electric, LOGIQ* E9) using an age-appropriate 9–12 mHz curved array transducer. The US was done by three independent radiologists with the subject lying in the supine position and scanned in the para-coronal view with the transducer positioned to obtain the longest kidney dimension. Then a transverse image was obtained at a 90° angle to the longest axis where the width (transverse dimension) and thickness (anteroposterior) dimensions of the kidney were measured. Each radiologist obtained 3 measurements of all three dimensions for a total of 27 measurements of each kidney per participant. Kidney volume was calculated using the ellipsoid formula: [length*width*thickness*π]/6.17 Simultaneously, 3 repeated measurements for weight, height, and blood pressure were performed and serum creatinine was measured by phlebotomy through an experienced nurse.
Height was measured without shoes on a wall-mounted stadiometer and weight was measured without heavy clothes using either a digital or balance-beam scale. Body mass index (BMI) was calculated as weight (kg)/height (m2). Age- and gender-independent height and weight z-scores were calculated as previously described, using the Ped(z) app with the WHO reference intervals.18
Blood pressure was measured with the subject sitting quietly after 5 min, with back supported, feet on the floor, and right arm supported at heart level. With an appropriate cuff size and a standard clinical sphygmomanometer, a stethoscope was placed over the brachial artery pulse, proximal and medial to the cubital fossa, and below the bottom edge of the cuff per standard technique. The authors used the fourth task force blood pressure thresholds to calculate age- and gender-independent blood pressure z-scores.19,20
Blood samples were collected in anticoagulant and lithium heparin tubes. Creatinine was isotope dilution-mass spectrometry traceable and measured using a calorimetric modified Jaffe method using alkaline picrate.21 Estimated glomerular filtration rate (eGFR) was calculated from the average of the three height measurements and serum creatinine with the new modified Schwartz formula.22 The body surface area was calculated using the Mosteller formula.23
Statistical analysisFor analyses, children were classified as follows: the non-exposed group defined as gestational age (GA) 38–41 weeks, and five-year weight > 5th percentile using the WHO growth charts,18 and the exposed group consisted of the other children born at < 36 weeks of gestation. According to the renal volume results in the study by Kandasamy et al.,24 the difference in the mean renal volumes between term and preterm infants was 3.6 mL. To achieve a 95% bilateral confidence interval, statistical power of 80%, with a sample size ratio of 3 (prematurely versus full-term born group), the sample size required for the full-term group was 27 and 89 for the premature group. Anthropometric z-scores were calculated using the app Ped(z) based on WHO growth charts.18 Renal volume z-scores were calculated using the same app based on the body surface area nomograms based on Scholbach and Weitzel.13 The authors also calculated the total kidney volumes and the total kidney volumes per body surface area (TKV/m2).25
Calculations were performed on the average of all repeated measurements. Continuous variables were analyzed for normal distribution using the D'Agostino-Pearson normality test.26 Simple descriptive statistics were used wherever possible. Categorical values were analyzed using Fisher's exact test, for continuous variables the Student's t-test for normally distributed parameters was used, and the Mann-Whitney test otherwise. All calculations were performed using GraphPad Prism version 5.0f for Mac, registered to GF.
ResultsDemographics and clinical variablesFig. 1 shows the flow and distribution of the study participants included. Thirty-three study participants comprised the control group whereas 89 study participants comprised the premature group. Table 1 summarizes the anthropometric and renal measurements of the two participant groups.
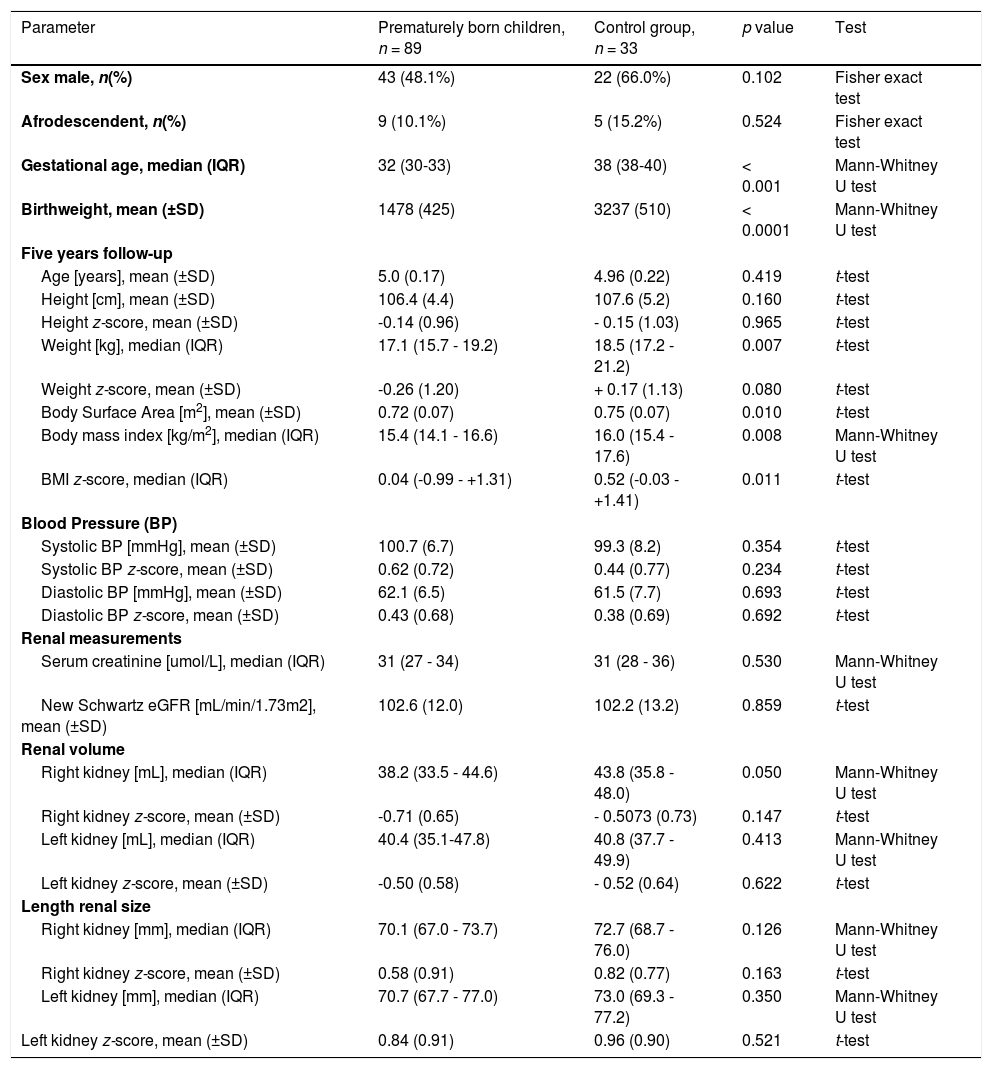
Prematurely born children and control group demographics and renal volume measurements.
The control group (n = 33) had a mean birth weight of 3237 g, whereas the premature group had a significantly lower mean birth weight of 1478 g (p < 0.0001). The median gestational age was 32 weeks in the premature group and 38 weeks in the control group (p < 0.001). There were no significant statistical differences with regards to sex or Afro-Colombian origin.
Anthropometry differed among both groups. While study participants had an identical age and height, the weight of the prematurely born children was significantly lower. The mean weight was 17.65 kg in the premature cohort and 19.05 kg in the control cohort (p = 0.0072). There was a trend towards a lower weight z-score (-0.2570 in the premature group and +0.1697 in the control group, p = 0.0402, one-sided t-test). Subsequently, the body surface area was significantly lower in the premature group.
There was no difference in the systolic and diastolic blood pressure z-scores. The average systolic blood pressure in the premature group was 100.7(±6.7) mm Hg, not significantly different from the control group (99.3(±8.2) mmHg (p = 0.3539). The average diastolic blood pressure in the premature group was 62.1(±6.5) mmHg, not significantly different from the control group (61.5(±7.7) mmHg (p = 0.6933). The average systolic blood pressure z-score in the premature group was +0.62(±0.72), not significantly different from the control group (0.44(±0.77) (p = 0.2336). The average diastolic blood pressure z-score in the premature group was +0.43(±0.68), not significantly different from the control group (0.38(±0.69) (p = 0.6921).
Renal function measured by biomarkers was not different among groups. Mean serum creatinine and Schwartz eGFR were similar among both groups, with a mean eGFR of 102.6 mL/min/1.73 m2 in the premature group and 102.2 mL/min/1.73 m2 in the control group.
The kidney volume of the right kidney was significantly smaller in the premature group (mean 39.4 mL) as compared to the control group (43.37 mL, p = 0.0306). However, after correcting the renal volume to body surface area and expressing the measurements as z-scores, there was no significant difference between groups. The left renal volume and the left renal volume z-score did not differ among groups (Table 1). The authors also found no differences between groups for the right or left kidney length or the right or left kidney length z-score (Table 1).
The authors also calculated the total renal volumes and the total renal volumes per body surface area (TKV/m2). The mean TKV/m2 of the preterm group was 112.5 ± 18.8 mL, not significantly different from the control group (114.5 ± 19.1 mL, p = 0.6034, unpaired t-test). There was also no significant correlation between the renal volume z-score and the TKV/m2.
The authors then calculated the correlation between the renal volume z-scores and the gestational age. Interestingly, neither the right nor left renal volume z-score correlated significantly with the gestational age (Pearson r = 0.1056 and 0.006 for the right and left renal volumes, respectively). There was also no difference in kidney volume when discriminating by sex (Pearson r = 0.1097 and -0.0759 for the right and left renal volumes, respectively).
DiscussionIn this study, the authors found normal renal length z-scores and renal volume z-scores in both the prematurely born children and the controls. Only the raw right renal volume was significantly lower in the premature group. After correcting for body surface area, this was no longer significant. The authors also found no correlation of renal length or renal volume z-scores and gestational age, suggesting that, through hyperfiltration, renal volume can be normalized by 5 years of age even in extremely premature babies. The authors also found no difference in markers of renal function. However, the children born prematurely had a significantly lower weight and a trend towards a lower weight z-score.
These findings are in keeping with the recent paper by Rakow et al.14 While the raw right kidney volumes were smaller in the premature group, similar to the previous reports,12 the authors did not find this for the left kidney volumes. As outlined in the introduction, both of these studies did not correct for body surface area. It is conceivable that the differences that were previously reported would disappear similar to the findings of Rakow and the present study's findings. Moreover, the renal function parameters were also not different, similar to that of Miklaszewska et al.27 Renal volumes correlate tightly with the body surface area, the relationship between renal volumes and body surface area never changes during childhood.13 Renal volume z-scores are independent of age in childhood.13 The renal volume is adjusted through a complex system of hormones by increasing the diameter of the individual glomeruli to the body surface area, thereby making the kidney cortex grow without increasing the number of nephrons. Nonetheless, Crump et al. showed in a large national cohort study a strong inverse association between gestational age at birth and risk of chronic kidney disease from childhood into mid-adulthood. After adjustment for other perinatal and maternal factors, those born preterm or extremely preterm had nearly twofold and threefold higher risks, respectively.28 It would have been ideal to use the renal volume z-score as a surrogate for the identification of risk. However, the present study's data would suggest that renal volumes, especially when corrected for body surface area, are not useful biomarkers for the identification of prematurely born babies at risk for CKD later in life, at least not yet at the age of five years. The significantly lower weight of the prematurely born children has previously been described.29 This explains the lower body surface area found in the present study's participants, which subsequently mitigates any differences between groups with regards to kidney volumes. The data presented here are in keeping with the recent description of a rapid increase of renal cortex volumes in prematurely born children by Li et al.10
The present study has some limitations. The proportion of extremely premature babies was low. Only 8 study participants were under 28 weeks of gestation. The authors also did not have cystatin C measurements, which are a better tool for assessing renal function in children. The original cohort shrunk because of refusal to participate, which introduces an unknown bias. The follow-up of 5 years may be insufficient to demonstrate differences in renal volumes. The authors also faced challenges recruiting the control group due to the need for a blood test. The fact that the control group comprises only 1/3 of the prematurely born children group may have introduced bias. Moreover, the authors used German reference intervals for the calculation of the age-independent z-scores. Data from Argentina may be more representative and have been published by Bianchi et al., however, these data do not permit the calculation of age-independent z-scores.30 Nonetheless, the study comprises a sizeable cohort of longitudinally study participants that compares favorably to the published literature. The repeated measures of all kidney dimensions by three independent radiologists, each in triplicates, form a strength.
Taken together, the present study's data suggest that renal volumes of 5-year old prematurely born children do not differ from healthy controls, especially after adjusting for body surface area. The authors propose to always calculate renal volume z-scores, which are easily facilitated by the Ped(z) app. The measurement of renal volume z-scores at age 5 may not identify low nephron endowment in prematurely born children and may therefore not be a useful tool to identify study participants at risk of CKD later in life.
FundingThis study was generously supported by the International Society of Nephrology with $10,000 US and Dr. Wendy Hoy from Australia with $10,000 US through the International Society of Nephrology (ISN) Sister Renal Center Program: Boston Children´s Hospital USA – Fundación Valle del Líli Cali Colombia.
First of all, the authors thank our study participants and their caregivers. We thank the Lilibeth Caberto Kidney Clinical Research Unit, University of Western Ontario, in London Ontario, for the generous assistance with research space and infrastructure. The authors thank the Centro de investigaciones clinicas of Fundación Valle del Lili for the strong support given during the entire research process. Dra. Laura Torres-Canchala was generously supported by the International Society of Nephrology for a two-month research sabbatical at the University of Western Ontario.