To describe the clinical characteristics of children and adolescents admitted to intensive care with confirmed COVID-19.
MethodProspective, multicenter, observational study, in 19 pediatric intensive care units. Patients aged 1 month to 19 years admitted consecutively (March–May 2020) were included. Demographic, clinical-epidemiological features, treatment, and outcomes were collected. Subgroups were compared according to comorbidities, age < 1 year, and need for invasive mechanical ventilation. A multivariable logistic regression model was used for predictors of severity.
ResultsSeventy-nine patients were included (ten with multisystemic inflammatory syndrome). Median age 4 years; 54% male (multisystemic inflammatory syndrome, 80%); 41% had comorbidities (multisystemic inflammatory syndrome, 20%). Fever (76%), cough (51%), and tachypnea (50%) were common in both groups. Severe symptoms, gastrointestinal symptoms, and higher inflammatory markers were more frequent in multisystemic inflammatory syndrome. Interstitial lung infiltrates were common in both groups, but pleural effusion was more prevalent in the multisystemic inflammatory syndrome group (43% vs. 14%). Invasive mechanical ventilation was used in 18% (median 7.5 days); antibiotics, oseltamivir, and corticosteroids were used in 76%, 43%, and 23%, respectively, but not hydroxychloroquine. The median pediatric intensive care unit length-of-stay was five days; there were two deaths (3%) in the non- multisystemic inflammatory syndrome group. Patients with comorbidities were older and comorbidities were independently associated with the need for invasive mechanical ventilation (OR 5.5; 95% CI, 1.43–21.12; p = 0.01).
ConclusionsIn Brazilian pediatric intensive care units, COVID-19 had low mortality, age less than 1 year was not associated with a worse prognosis, and patients with multisystemic inflammatory syndrome had more severe symptoms, higher inflammatory biomarkers, and a greater predominance of males, but only comorbidities and chronic diseases were independent predictors of severity.
Descrever as características clínicas de crianças e adolescentes internados em unidade de terapia intensiva com COVID-19 confirmada.
MétodoEstudo prospectivo, multicêntrico, observacional, em 19 unidades de terapia intensiva pediátrica. Foram incluídos pacientes entre um mês e 19 anos, admitidos consecutivamente (março a maio de 2020). As características demográficas, clínico-epidemiológicas, o tratamento e os resultados foram coletados. Os subgrupos foram comparados de acordo com as comorbidades, idade <1 ano e necessidade de ventilação mecânica invasiva. Um modelo de regressão logística multivariável foi utilizado para preditores de gravidade.
ResultadosSetenta e nove pacientes foram incluídos (10 com síndrome inflamatória multissistêmica). Mediana de idade, quatro anos; 54% eram do sexo masculino (síndrome inflamatória multissistêmica, 80%); 41% tinham comorbidades (síndrome inflamatória multissistêmica, 20%). Febre (76%), tosse (51%) e taquipneia (50%) foram comuns nos dois grupos. Sintomas graves e gastrointestinais e marcadores inflamatórios mais elevados foram mais frequentes na presença de síndrome inflamatória multissistêmica. Infiltrados intersticiais pulmonares foram comuns em ambos os grupos, mas o derrame pleural foi mais prevalente no grupo com síndrome inflamatória multissistêmica (43% vs. 14%). A ventilação mecânica invasiva foi utilizada em 18% (mediana 7,5 dias); antibióticos, oseltamivir e corticosteroides foram utilizados em 76%, 43% e 23%, respectivamente, mas não a hidroxicloroquina. A mediana do tempo de permanência na unidade de terapia intensiva pediátrica foi de 5 dias; duas mortes ocorreram (3%) no grupo não- síndrome inflamatória multissistêmica. Os pacientes com comorbidades eram mais velhos, e as comorbidades foram independentemente associadas à necessidade de ventilação mecânica invasiva (OR 5,5; IC95%, 1,43-21,12; P 0,01).
ConclusõesNas unidades de terapia intensiva pediátrica brasileiras, a COVID-19 apresentou baixa mortalidade, a idade inferior a um ano não foi associada a um pior prognóstico, os pacientes com síndrome inflamatória multissistêmica apresentaram sintomas mais graves, biomarcadores inflamatórios mais elevados e uma grande predominância no sexo masculino, mas apenas a presença de comorbidades e doenças crônicas foi um preditor independente de gravidade.
Brazil has become the epicenter of infection by the new coronavirus (SARS-CoV-2) in South America and the second country with the highest number of cases and deaths in the world. COVID-19, as this disease is termed by the World Health Organization, still represents a challenge in the pediatric population, although the number and severity of cases are lower when compared to the adult population. Data from several countries show that children and adolescents accounted for less than 2% of symptomatic cases, with hospitalization rates (0.6–20%) and mortality (0–4%) significantly lower than in adults.1–5 Infants and children with previous chronic conditions represent the most vulnerable pediatric population, with greater severity.1,2,4,5 Recently, several countries reported cases of a multisystemic inflammatory syndrome in children (MIS-C) temporarily associated with COVID-19. This syndrome is characterized by prolonged fever, gastrointestinal symptoms, and altered inflammatory markers, associated with signs of organ dysfunction, acute cardiac failure, Kawasaki disease (complete or incomplete), or toxic shock syndrome.6–8
The report of the Latin American experience with COVID-19 in pediatrics is still quite limited, especially concerning children admitted to intensive care settings. This study aimed to describe the clinical characteristics of a cohort of children and adolescents admitted to Brazilian pediatric intensive care units (PICUs).
Materials and methodsStudy design, patient selection, and settingThis was a prospective, multicenter study, conducted in 19 PICUs associated with the Brazilian Research Network in Pediatric Intensive Care (BRnet-PIC). Pediatric patients (1 month–19 years of age) with confirmed COVID-19 were included consecutively between March 1 and May 31, 2020, after obtaining informed consent. The studywas approved by the Research Ethics Committees of all institutions.
Diagnosis and data collectionDiagnosis of COVID-19 was confirmed by reverse transcription-polymerase chain reaction (RT-PCR) assay from oro/nasopharyngeal swabs or tracheal aspirates, and when outside the period of positivity for RT-PCR, using IgM and/or IgG antibodies positive against SARS-CoV-2. Demographic, epidemiological, clinical, and outcome data from patients were collected prospectively, using standardized case report forms (REDCap – Vanderbilt University; TN, United States). MIS-C and non-MIS-C patients were compared according to the presence of comorbidities, age less than 1 year, and the need for invasive mechanical ventilation (IMV).
Data processing and statistical analysisCategorical variables were described as frequencies and percentages, and continuous variables as medians and interquartile ranges (IQRs). Comparisons between groups were made using the chi-squared test or Fisher’s exact test for categorical variables, and the Mann–Whitney test for continuous variables. A multivariable logistic regression model was used to assess the association of the covariates age less than 1 year, sex, race, and presence of comorbidities with more severe forms of the disease, characterized by the need for IMV. A significance level of 5% (two-tailed) and 95% confidence intervals were established. The software R, v. 3.6.1, (R Foundation, Vienna, Austria) was used for statistical analysis.
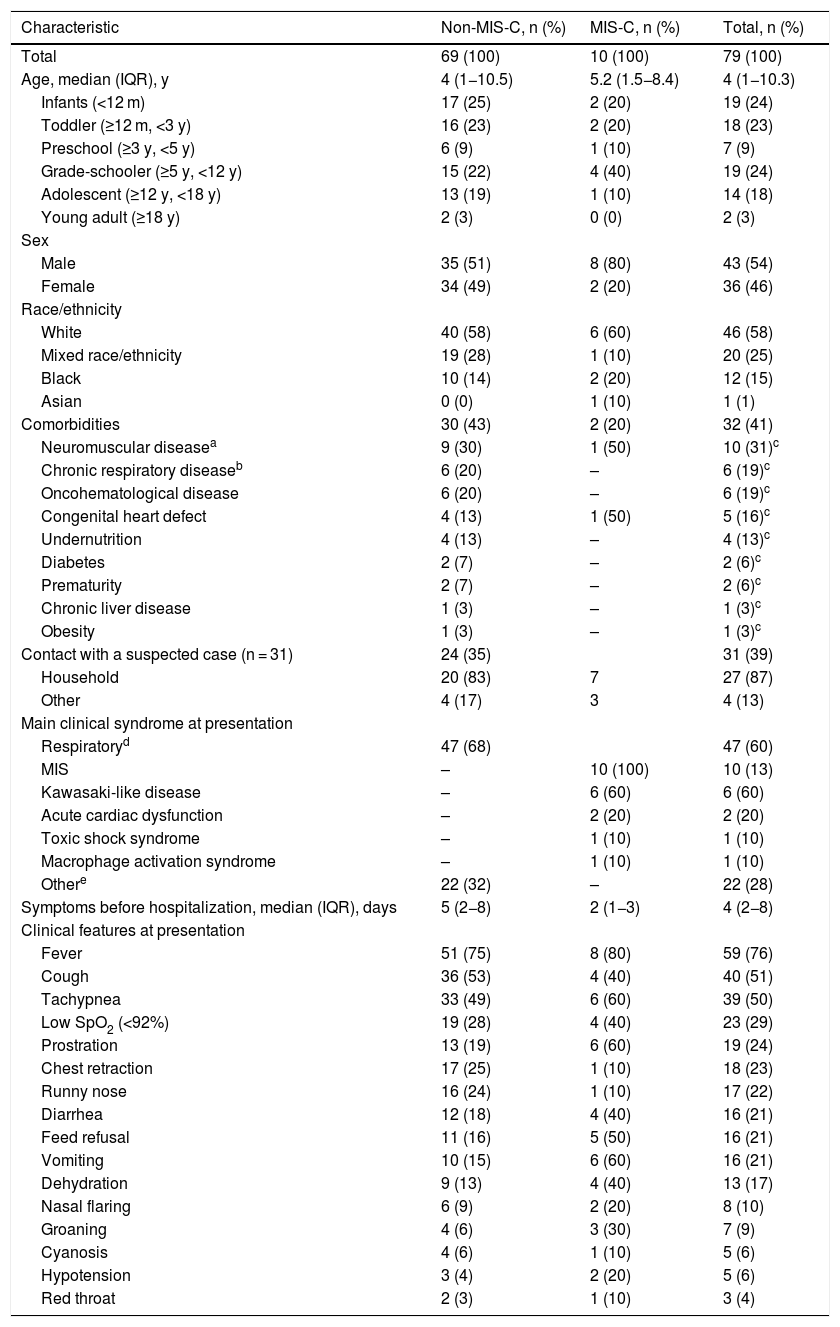
ResultsDemographic and epidemiological featuresSeventy-nine patients were included (ten with MIS-C). Overall, the median age was four years, 54% were male (MIS-C, 80%), 58% were white, and 41% had previous comorbidities (MIS-C, 20%), of which neuromuscular diseases predominated (28%), mainly non-progressive encephalopathy. Other comorbidities, such as chronic respiratory disease, oncohematological disease, congenital heart disease, and undernutrition were also prevalent, representing together about 27% of the total. Thirty-one patients (39%) reported contact with a suspected case (household, 87%; Table 1).
Demographic, epidemiological, and clinical features of pediatric patients with COVID-19.
| Characteristic | Non-MIS-C, n (%) | MIS-C, n (%) | Total, n (%) |
|---|---|---|---|
| Total | 69 (100) | 10 (100) | 79 (100) |
| Age, median (IQR), y | 4 (1−10.5) | 5.2 (1.5−8.4) | 4 (1−10.3) |
| Infants (<12 m) | 17 (25) | 2 (20) | 19 (24) |
| Toddler (≥12 m, <3 y) | 16 (23) | 2 (20) | 18 (23) |
| Preschool (≥3 y, <5 y) | 6 (9) | 1 (10) | 7 (9) |
| Grade-schooler (≥5 y, <12 y) | 15 (22) | 4 (40) | 19 (24) |
| Adolescent (≥12 y, <18 y) | 13 (19) | 1 (10) | 14 (18) |
| Young adult (≥18 y) | 2 (3) | 0 (0) | 2 (3) |
| Sex | |||
| Male | 35 (51) | 8 (80) | 43 (54) |
| Female | 34 (49) | 2 (20) | 36 (46) |
| Race/ethnicity | |||
| White | 40 (58) | 6 (60) | 46 (58) |
| Mixed race/ethnicity | 19 (28) | 1 (10) | 20 (25) |
| Black | 10 (14) | 2 (20) | 12 (15) |
| Asian | 0 (0) | 1 (10) | 1 (1) |
| Comorbidities | 30 (43) | 2 (20) | 32 (41) |
| Neuromuscular diseasea | 9 (30) | 1 (50) | 10 (31)c |
| Chronic respiratory diseaseb | 6 (20) | – | 6 (19)c |
| Oncohematological disease | 6 (20) | – | 6 (19)c |
| Congenital heart defect | 4 (13) | 1 (50) | 5 (16)c |
| Undernutrition | 4 (13) | – | 4 (13)c |
| Diabetes | 2 (7) | – | 2 (6)c |
| Prematurity | 2 (7) | – | 2 (6)c |
| Chronic liver disease | 1 (3) | – | 1 (3)c |
| Obesity | 1 (3) | – | 1 (3)c |
| Contact with a suspected case (n = 31) | 24 (35) | 31 (39) | |
| Household | 20 (83) | 7 | 27 (87) |
| Other | 4 (17) | 3 | 4 (13) |
| Main clinical syndrome at presentation | |||
| Respiratoryd | 47 (68) | 47 (60) | |
| MIS | – | 10 (100) | 10 (13) |
| Kawasaki-like disease | – | 6 (60) | 6 (60) |
| Acute cardiac dysfunction | – | 2 (20) | 2 (20) |
| Toxic shock syndrome | – | 1 (10) | 1 (10) |
| Macrophage activation syndrome | – | 1 (10) | 1 (10) |
| Othere | 22 (32) | – | 22 (28) |
| Symptoms before hospitalization, median (IQR), days | 5 (2−8) | 2 (1−3) | 4 (2−8) |
| Clinical features at presentation | |||
| Fever | 51 (75) | 8 (80) | 59 (76) |
| Cough | 36 (53) | 4 (40) | 40 (51) |
| Tachypnea | 33 (49) | 6 (60) | 39 (50) |
| Low SpO2 (<92%) | 19 (28) | 4 (40) | 23 (29) |
| Prostration | 13 (19) | 6 (60) | 19 (24) |
| Chest retraction | 17 (25) | 1 (10) | 18 (23) |
| Runny nose | 16 (24) | 1 (10) | 17 (22) |
| Diarrhea | 12 (18) | 4 (40) | 16 (21) |
| Feed refusal | 11 (16) | 5 (50) | 16 (21) |
| Vomiting | 10 (15) | 6 (60) | 16 (21) |
| Dehydration | 9 (13) | 4 (40) | 13 (17) |
| Nasal flaring | 6 (9) | 2 (20) | 8 (10) |
| Groaning | 4 (6) | 3 (30) | 7 (9) |
| Cyanosis | 4 (6) | 1 (10) | 5 (6) |
| Hypotension | 3 (4) | 2 (20) | 5 (6) |
| Red throat | 2 (3) | 1 (10) | 3 (4) |
COVID-19, coronavirus disease 2019; IQR, interquartile range; MIS-C, multisystemic inflammatory syndrome in children; PICU, pediatric intensive care unit; SpO2, pulse oximeter oxygen saturation.
Upon admission, 47 patients (60%) had respiratory symptoms, most of them pneumonia or bronchiolitis (70%), and ten (13%) had MIS-C, 60% a Kawasaki-like disease. The other 22 (28%) had mostly gastrointestinal and neurological symptoms (Table 1). The median time of symptoms before hospitalization was five days in the non-MIS-C group and two days in the MIS-C group. The most common signs and symptoms at presentation were fever (76%), cough (51%), and tachypnea (50%). However, other findings were also prevalent (Table 1). In the MIS-C group, severe symptoms such as tachypnea (60%), low SpO2 (40%), prostration (60%), groaning (30%), hypotension (20%), nasal flaring (20%), cyanosis (10%), and gastrointestinal symptoms (40–60%) were more frequent than in the non-MIS-C group (Table 1).
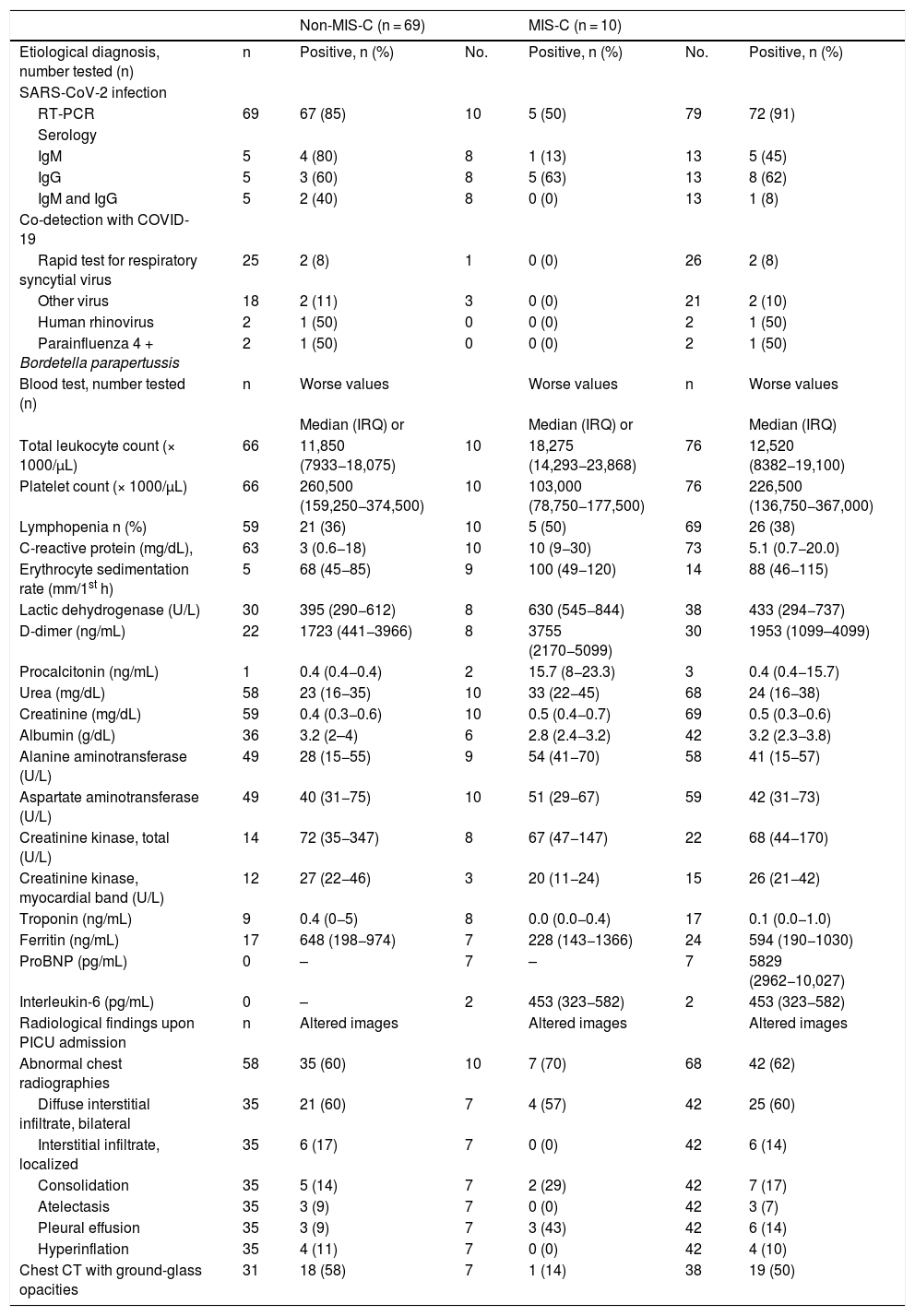
Diagnosis confirmation; laboratory and radiological findingsThe diagnosis was confirmed by RT-PCR in 72 patients (all 79 tested, 91% positive). The others confirmed the diagnosis by detection of IgM and/or IgG antibodies (five in the MIS-C group; Table 2). Co-detection with other viruses occurred in about 15% tested (two respiratory syncytial virus, one human rhinovirus, and one association of parainfluenza 4 with Bordetella pertussis; Table 2).
Diagnosis confirmation, laboratory and radiological findings of pediatric patients with COVID-19.
| Non-MIS-C (n = 69) | MIS-C (n = 10) | |||||
|---|---|---|---|---|---|---|
| Etiological diagnosis, number tested (n) | n | Positive, n (%) | No. | Positive, n (%) | No. | Positive, n (%) |
| SARS-CoV-2 infection | ||||||
| RT-PCR | 69 | 67 (85) | 10 | 5 (50) | 79 | 72 (91) |
| Serology | ||||||
| IgM | 5 | 4 (80) | 8 | 1 (13) | 13 | 5 (45) |
| IgG | 5 | 3 (60) | 8 | 5 (63) | 13 | 8 (62) |
| IgM and IgG | 5 | 2 (40) | 8 | 0 (0) | 13 | 1 (8) |
| Co-detection with COVID-19 | ||||||
| Rapid test for respiratory syncytial virus | 25 | 2 (8) | 1 | 0 (0) | 26 | 2 (8) |
| Other virus | 18 | 2 (11) | 3 | 0 (0) | 21 | 2 (10) |
| Human rhinovirus | 2 | 1 (50) | 0 | 0 (0) | 2 | 1 (50) |
| Parainfluenza 4 + Bordetella parapertussis | 2 | 1 (50) | 0 | 0 (0) | 2 | 1 (50) |
| Blood test, number tested (n) | n | Worse values | Worse values | n | Worse values | |
| Median (IRQ) or | Median (IRQ) or | Median (IRQ) | ||||
| Total leukocyte count (× 1000/µL) | 66 | 11,850 (7933−18,075) | 10 | 18,275 (14,293−23,868) | 76 | 12,520 (8382−19,100) |
| Platelet count (× 1000/µL) | 66 | 260,500 (159,250−374,500) | 10 | 103,000 (78,750−177,500) | 76 | 226,500 (136,750−367,000) |
| Lymphopenia n (%) | 59 | 21 (36) | 10 | 5 (50) | 69 | 26 (38) |
| C-reactive protein (mg/dL), | 63 | 3 (0.6−18) | 10 | 10 (9−30) | 73 | 5.1 (0.7−20.0) |
| Erythrocyte sedimentation rate (mm/1st h) | 5 | 68 (45−85) | 9 | 100 (49−120) | 14 | 88 (46−115) |
| Lactic dehydrogenase (U/L) | 30 | 395 (290−612) | 8 | 630 (545−844) | 38 | 433 (294−737) |
| D-dimer (ng/mL) | 22 | 1723 (441−3966) | 8 | 3755 (2170−5099) | 30 | 1953 (1099–4099) |
| Procalcitonin (ng/mL) | 1 | 0.4 (0.4−0.4) | 2 | 15.7 (8−23.3) | 3 | 0.4 (0.4−15.7) |
| Urea (mg/dL) | 58 | 23 (16−35) | 10 | 33 (22−45) | 68 | 24 (16−38) |
| Creatinine (mg/dL) | 59 | 0.4 (0.3−0.6) | 10 | 0.5 (0.4−0.7) | 69 | 0.5 (0.3−0.6) |
| Albumin (g/dL) | 36 | 3.2 (2–4) | 6 | 2.8 (2.4−3.2) | 42 | 3.2 (2.3−3.8) |
| Alanine aminotransferase (U/L) | 49 | 28 (15−55) | 9 | 54 (41−70) | 58 | 41 (15−57) |
| Aspartate aminotransferase (U/L) | 49 | 40 (31−75) | 10 | 51 (29−67) | 59 | 42 (31−73) |
| Creatinine kinase, total (U/L) | 14 | 72 (35−347) | 8 | 67 (47−147) | 22 | 68 (44−170) |
| Creatinine kinase, myocardial band (U/L) | 12 | 27 (22−46) | 3 | 20 (11−24) | 15 | 26 (21−42) |
| Troponin (ng/mL) | 9 | 0.4 (0−5) | 8 | 0.0 (0.0−0.4) | 17 | 0.1 (0.0−1.0) |
| Ferritin (ng/mL) | 17 | 648 (198−974) | 7 | 228 (143−1366) | 24 | 594 (190−1030) |
| ProBNP (pg/mL) | 0 | – | 7 | – | 7 | 5829 (2962−10,027) |
| Interleukin-6 (pg/mL) | 0 | – | 2 | 453 (323−582) | 2 | 453 (323−582) |
| Radiological findings upon PICU admission | n | Altered images | Altered images | Altered images | ||
| Abnormal chest radiographies | 58 | 35 (60) | 10 | 7 (70) | 68 | 42 (62) |
| Diffuse interstitial infiltrate, bilateral | 35 | 21 (60) | 7 | 4 (57) | 42 | 25 (60) |
| Interstitial infiltrate, localized | 35 | 6 (17) | 7 | 0 (0) | 42 | 6 (14) |
| Consolidation | 35 | 5 (14) | 7 | 2 (29) | 42 | 7 (17) |
| Atelectasis | 35 | 3 (9) | 7 | 0 (0) | 42 | 3 (7) |
| Pleural effusion | 35 | 3 (9) | 7 | 3 (43) | 42 | 6 (14) |
| Hyperinflation | 35 | 4 (11) | 7 | 0 (0) | 42 | 4 (10) |
| Chest CT with ground-glass opacities | 31 | 18 (58) | 7 | 1 (14) | 38 | 19 (50) |
COVID-19, coronavirus disease 2019; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; RT-PCR, reverse transcription polymerase chain reaction; IgM, immunoglobulin M; IgG, immunoglobulin G; IQR, interquartile range; proBNP, pro-B type natriuretic peptide; PICU, pediatric intensive care unit, CT, computed tomography.
Lymphopenia was present in 36% of patients in the non-MIS-C group (MIS-C, 50%). C-reactive protein (CRP) was 3 mg/dL or greater in 50% of 63 patients in the non-MIS-C group, compared to 10 mg/dL or greater in 50% of all ten patients in the MIS-C group. Erythrocyte sedimentation rate (ESR), lactic dehydrogenase (LDH), and D-dimer were tested in five, 30, and 22 patients, respectively, in the non-MIS-C group, and in almost all patients in the MIS-C group. They were increased in at least 75% of all patients, but much higher in the MIS-C group. In contrast, renal and hepatic functions were tested in most patients and were preserved in at least 75% of them in both groups. Ferritin and troponin were tested in only 24 and 17 patients, respectively, most of them in the MIS-C group. Ferritin was elevated in at least 50% of patients, and was higher in the non-MIS-C group. Troponin was elevated in at least 50% of patients in the non-MIS-C group, but not in the MIS-C group. Of the other cardiac injury markers, creatinine kinase (CK) was abnormal in five of 22 patients (408–2345 U/L), and the myocardial creatinine kinase band (CK–MB) was elevated in eight out of 15 patients (25 and 89 U/L). Only one of these patients was in the MIS-C group (toxic shock syndrome, CK 1389 U/L, CK–MB 28 U/L). The pro-b-type natriuretic peptide (proBNP) was increased in six of seven (86%) patients tested, all in the MIS-C group, with a median value of 5829 (range, 222−16,996 pg/mL). Procalcitonin was investigated in only three patients and was elevated in all (one in the non-MIS-C group, 0.4 ng/mL; two in the MIS-C group: an acute cardiac dysfunction, 0.37 ng/mL, and a toxic shock syndrome, 31 ng/mL). Finally, interleukin-6 was measured in only two patients in the MIS-C group, both with very high values: a patient with acute cardiac dysfunction (711 pg/mL) and a patient with toxic shock syndrome (194 pg/mL; Table 2).
Chest radiography was abnormal in 60–70% of patients in both groups, predominantly bilateral diffuse interstitial infiltrate (57–60%). Remarkably, pleural effusion was present in seven patients (43%) in the MIS-C group and in only three patients (9%) in the non-MIS-C group. Thirty-eight patients underwent computed tomography (CT) of the chest and ground-glass opacities were present in 58% in the non-MIS-C group and in only 14% in the MIS-C group (Table 2).
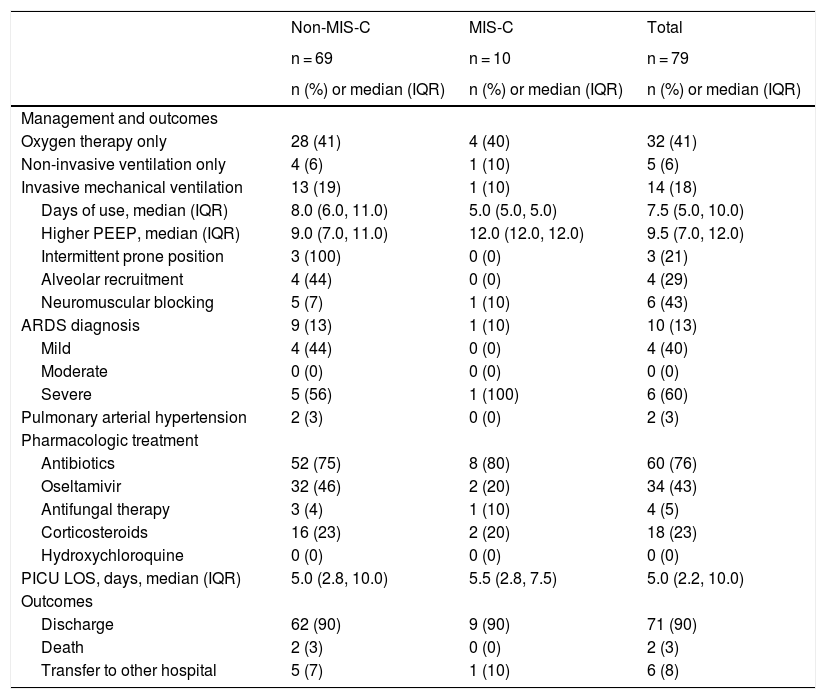
Management and clinical outcomesFifty-one patients (65%) needed some type of ventilatory support: 32 (41%) used only oxygen therapy, five (6%) needed only noninvasive ventilation (NIV), and 14 (18%) needed IMV. In those requiring IMV, ten (71%) had acute respiratory distress syndrome (ARDS), of whom six were classified as severe, requiring neuromuscular blocking (n = 6), alveolar recruitment maneuver (n = 4), and intermittent prone position (n = 3). Two of these patients developed pulmonary arterial hypertension. The median duration of IMV was 7.5 days (IQR 5–10) and the median highest positive end-expiratory pressure (PEEP) was 9.5 cmH2O (IQR 7–12). One of those severe cases of ARDS was in the MIS-C group (Table 3).
Management and clinical outcomes of pediatric patients with COVID-19.
| Non-MIS-C | MIS-C | Total | |
|---|---|---|---|
| n = 69 | n = 10 | n = 79 | |
| n (%) or median (IQR) | n (%) or median (IQR) | n (%) or median (IQR) | |
| Management and outcomes | |||
| Oxygen therapy only | 28 (41) | 4 (40) | 32 (41) |
| Non-invasive ventilation only | 4 (6) | 1 (10) | 5 (6) |
| Invasive mechanical ventilation | 13 (19) | 1 (10) | 14 (18) |
| Days of use, median (IQR) | 8.0 (6.0, 11.0) | 5.0 (5.0, 5.0) | 7.5 (5.0, 10.0) |
| Higher PEEP, median (IQR) | 9.0 (7.0, 11.0) | 12.0 (12.0, 12.0) | 9.5 (7.0, 12.0) |
| Intermittent prone position | 3 (100) | 0 (0) | 3 (21) |
| Alveolar recruitment | 4 (44) | 0 (0) | 4 (29) |
| Neuromuscular blocking | 5 (7) | 1 (10) | 6 (43) |
| ARDS diagnosis | 9 (13) | 1 (10) | 10 (13) |
| Mild | 4 (44) | 0 (0) | 4 (40) |
| Moderate | 0 (0) | 0 (0) | 0 (0) |
| Severe | 5 (56) | 1 (100) | 6 (60) |
| Pulmonary arterial hypertension | 2 (3) | 0 (0) | 2 (3) |
| Pharmacologic treatment | |||
| Antibiotics | 52 (75) | 8 (80) | 60 (76) |
| Oseltamivir | 32 (46) | 2 (20) | 34 (43) |
| Antifungal therapy | 3 (4) | 1 (10) | 4 (5) |
| Corticosteroids | 16 (23) | 2 (20) | 18 (23) |
| Hydroxychloroquine | 0 (0) | 0 (0) | 0 (0) |
| PICU LOS, days, median (IQR) | 5.0 (2.8, 10.0) | 5.5 (2.8, 7.5) | 5.0 (2.2, 10.0) |
| Outcomes | |||
| Discharge | 62 (90) | 9 (90) | 71 (90) |
| Death | 2 (3) | 0 (0) | 2 (3) |
| Transfer to other hospital | 5 (7) | 1 (10) | 6 (8) |
ARDS, acute respiratory distress syndrome; IQR, interquartile range; LOS, length of stay; PEEP, positive end expiratory pressure; PICU, pediatric intensive care unit.
Antibiotics were used in 76% of all patients, oseltamivir in 43%, and corticosteroids in 23%. No patient used hydroxychloroquine (Table 3). The median PICU length of stay (LOS) was five days (IQR 2.2–10), and most patients were discharged (90%). The only two deaths (3% mortality) occurred in the non-MIS group, both patients with severe comorbidities and chronic use of corticosteroids (Table 3). One was a 14-year-old girl with chronic liver disease and the other a 1-year-old girl with chronic lung disease and short bowel syndrome.
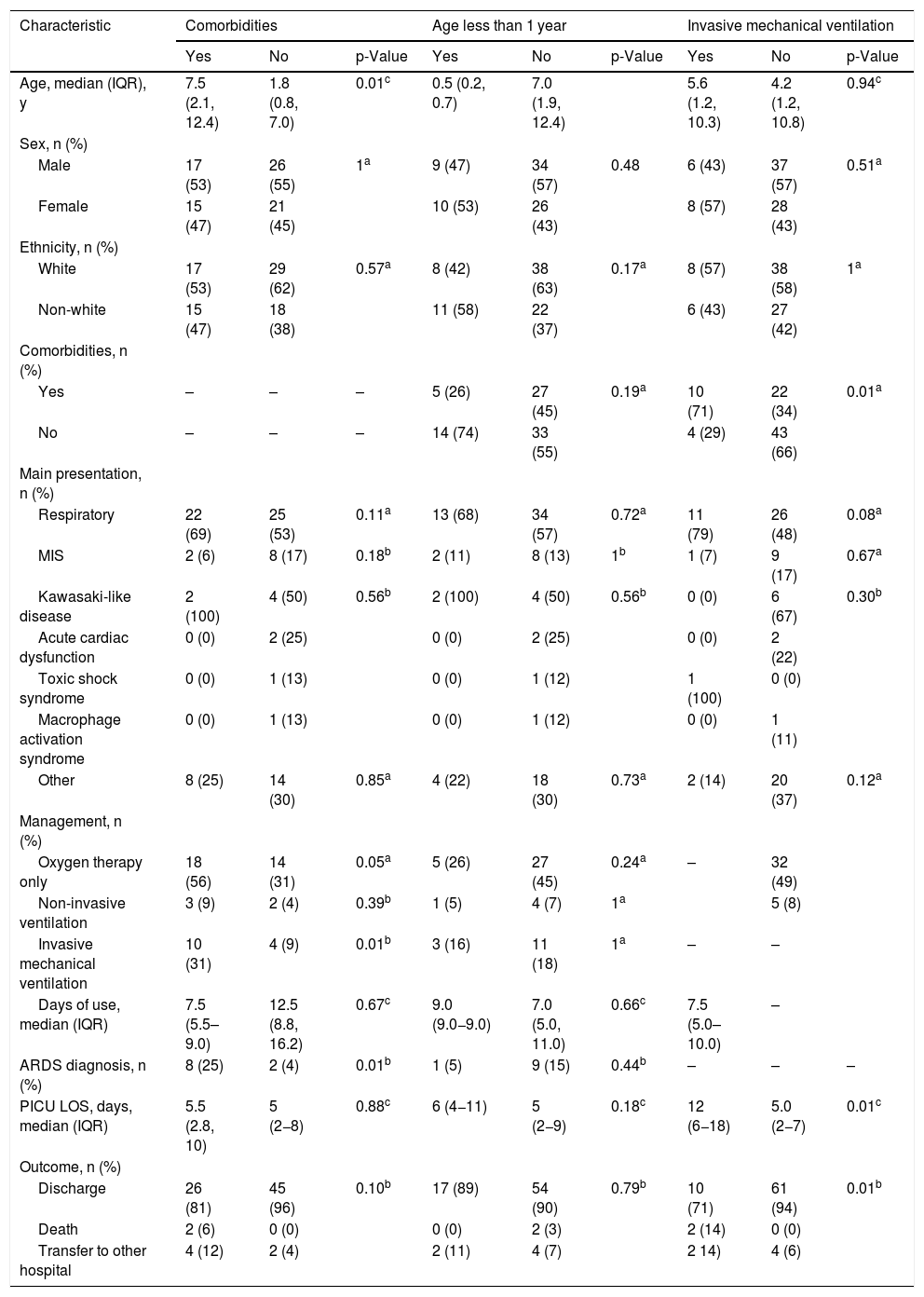
Specific subgroupsThe subgroup of patients with comorbidities was significantly older (median age: 7.5 vs. 1.8 years, p = 0.01), had a greater need for oxygen therapy (56% vs. 31%, p = 0.05) and IMV (31% vs. 9%, p = 0.01), and more frequent ARDS diagnosis (25% vs. 4%, p = 0.01), but there were no differences regarding the duration of respiratory support and PICU LOS. In patients who required IMV, the PICU LOS was significantly longer (12.0 vs. 5.0 days, p = 0.01). Age less than 1 year did not determine a different clinical presentation. In patients with MIS-C, no significant differences were observed among patients with comorbidities, age less than 1 year, and the need for IMV (Table 4a). These three specific groups were also compared, considering only patients who did not have MIS-C, but the results were quite similar (Tables S1 and S2, supplementary material).
Demographics and clinical features of pediatric patients with COVID-19 according to the presence of comorbidities, age less than 1 year, and the need for invasive mechanical ventilation, in all patients (n = 79).
| Characteristic | Comorbidities | Age less than 1 year | Invasive mechanical ventilation | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Yes | No | p-Value | Yes | No | p-Value | Yes | No | p-Value | |
| Age, median (IQR), y | 7.5 (2.1, 12.4) | 1.8 (0.8, 7.0) | 0.01c | 0.5 (0.2, 0.7) | 7.0 (1.9, 12.4) | 5.6 (1.2, 10.3) | 4.2 (1.2, 10.8) | 0.94c | |
| Sex, n (%) | |||||||||
| Male | 17 (53) | 26 (55) | 1a | 9 (47) | 34 (57) | 0.48 | 6 (43) | 37 (57) | 0.51a |
| Female | 15 (47) | 21 (45) | 10 (53) | 26 (43) | 8 (57) | 28 (43) | |||
| Ethnicity, n (%) | |||||||||
| White | 17 (53) | 29 (62) | 0.57a | 8 (42) | 38 (63) | 0.17a | 8 (57) | 38 (58) | 1a |
| Non-white | 15 (47) | 18 (38) | 11 (58) | 22 (37) | 6 (43) | 27 (42) | |||
| Comorbidities, n (%) | |||||||||
| Yes | – | – | – | 5 (26) | 27 (45) | 0.19a | 10 (71) | 22 (34) | 0.01a |
| No | – | – | – | 14 (74) | 33 (55) | 4 (29) | 43 (66) | ||
| Main presentation, n (%) | |||||||||
| Respiratory | 22 (69) | 25 (53) | 0.11a | 13 (68) | 34 (57) | 0.72a | 11 (79) | 26 (48) | 0.08a |
| MIS | 2 (6) | 8 (17) | 0.18b | 2 (11) | 8 (13) | 1b | 1 (7) | 9 (17) | 0.67a |
| Kawasaki-like disease | 2 (100) | 4 (50) | 0.56b | 2 (100) | 4 (50) | 0.56b | 0 (0) | 6 (67) | 0.30b |
| Acute cardiac dysfunction | 0 (0) | 2 (25) | 0 (0) | 2 (25) | 0 (0) | 2 (22) | |||
| Toxic shock syndrome | 0 (0) | 1 (13) | 0 (0) | 1 (12) | 1 (100) | 0 (0) | |||
| Macrophage activation syndrome | 0 (0) | 1 (13) | 0 (0) | 1 (12) | 0 (0) | 1 (11) | |||
| Other | 8 (25) | 14 (30) | 0.85a | 4 (22) | 18 (30) | 0.73a | 2 (14) | 20 (37) | 0.12a |
| Management, n (%) | |||||||||
| Oxygen therapy only | 18 (56) | 14 (31) | 0.05a | 5 (26) | 27 (45) | 0.24a | – | 32 (49) | |
| Non-invasive ventilation | 3 (9) | 2 (4) | 0.39b | 1 (5) | 4 (7) | 1a | 5 (8) | ||
| Invasive mechanical ventilation | 10 (31) | 4 (9) | 0.01b | 3 (16) | 11 (18) | 1a | – | – | |
| Days of use, median (IQR) | 7.5 (5.5–9.0) | 12.5 (8.8, 16.2) | 0.67c | 9.0 (9.0−9.0) | 7.0 (5.0, 11.0) | 0.66c | 7.5 (5.0–10.0) | – | |
| ARDS diagnosis, n (%) | 8 (25) | 2 (4) | 0.01b | 1 (5) | 9 (15) | 0.44b | – | – | – |
| PICU LOS, days, median (IQR) | 5.5 (2.8, 10) | 5 (2−8) | 0.88c | 6 (4−11) | 5 (2−9) | 0.18c | 12 (6−18) | 5.0 (2−7) | 0.01c |
| Outcome, n (%) | |||||||||
| Discharge | 26 (81) | 45 (96) | 0.10b | 17 (89) | 54 (90) | 0.79b | 10 (71) | 61 (94) | 0.01b |
| Death | 2 (6) | 0 (0) | 0 (0) | 2 (3) | 2 (14) | 0 (0) | |||
| Transfer to other hospital | 4 (12) | 2 (4) | 2 (11) | 4 (7) | 2 14) | 4 (6) | |||
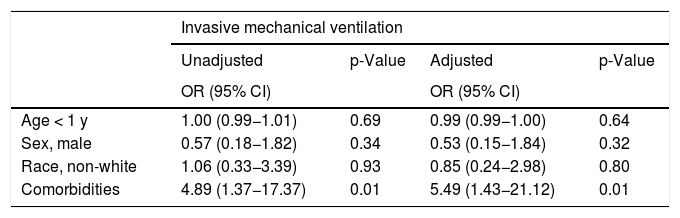
The multivariate analysis showed that only the presence of comorbidities was significantly associated with severity, represented by the need for IMV (adjusted OR, 5.5; 95% CI, 1.43–21.12; p = 0.01) (Table 4b).
Unadjusted and adjustedd odds ratios and 95% confidence intervals for pediatric patients with COVID-19, according to the need for invasive mechanical ventilation (n = 79).
| Invasive mechanical ventilation | ||||
|---|---|---|---|---|
| Unadjusted | p-Value | Adjusted | p-Value | |
| OR (95% CI) | OR (95% CI) | |||
| Age < 1 y | 1.00 (0.99−1.01) | 0.69 | 0.99 (0.99−1.00) | 0.64 |
| Sex, male | 0.57 (0.18−1.82) | 0.34 | 0.53 (0.15−1.84) | 0.32 |
| Race, non-white | 1.06 (0.33−3.39) | 0.93 | 0.85 (0.24−2.98) | 0.80 |
| Comorbidities | 4.89 (1.37−17.37) | 0.01 | 5.49 (1.43−21.12) | 0.01 |
IQR, interquartile range; MIS, multisystemic inflammatory syndrome; ARDS, acute respiratory distress syndrome; PICU, pediatric intensive care unit; LOS, length of stay; OR, odds ratio; CI, confidence interval; PICU, pediatric intensive care unit.
To the best of the authors’ knowledge, this is the first prospective, multicenter study to report characteristics and outcomes of children with COVID-19 admitted to PICUs in Brazil, the current epicenter of the disease in Latin America and the second in the world, after the United States. Although most children and adolescents have a benign course of the disease, it has been shown that some patients can develop severe acute clinical conditions, especially those with previous comorbidities, and later on, also present MIS-C.
In the present cohort, although the median age was 4 years and a quarter were infants, 44% were school-age children, adolescents, and young adults, a higher percentage than is generally observed in Brazilian PICUs for this age group, usually around 25%.9 This is similar to that reported by a large European study10 and by North American PICUs, which reported an even higher percentage of admissions in this age group (about 70%).2,11 The present study did not observe a large difference between the median ages of the MIS-C and non-MIS-C groups, although the number of patients with MIS-C was small. As for sex, there were no differences in the non-MIS-C group, while in patients with MIS-C there was a male predominance of 4:1, which is different from what was reported by other authors.12,13 There was a small predominance of white patients (58–60%), which contrasts with the predominance of the Brazilian population, which is 56% non-white. However, approximately half of the patients were from private hospitals, accessible only to the middle- and upper-income population, where white patients are the majority.
Comorbidities have been described as an important risk factor for more severe cases of COVID-19 in children, representing between 50–80% of PICU admissions.11,14,15 Although the present study had a slightly lower percentage of these patients (41%), they were significantly older and had a significantly more severe presentation (more ARDS, more ventilatory support, more IMV). The logistic regression model showed that comorbidities were independently associated with the need for IMV, with an adjusted odds ratio of 5.5 [95% CI, 1.4–21.1). Neuromuscular disease, chronic respiratory disease, and oncohematologic disease were the most prevalent comorbidities in this cohort, which is slightly different from others reports in pediatric critical care.5,11,14,15
Only about 40% of patients had a previous history of contact with a suspected case, mostly at home, which is similar to other reports.4 Fever was the predominant symptom, followed by several respiratory and gastrointestinal signs and symptoms, which did not differ from what has been reported in other studies.5,10,14,16 There was a higher prevalence of gastrointestinal symptoms in the MIS-C group, such as diarrhea and vomiting with dehydration, which was also observed by other authors.12,13 The interval between the onset of symptoms and hospitalization was shorter in the MIS-C group (median, 2 vs. 5 days). This may be explained by the most severe cases in this group, some of them presenting with signs of shock and cardiorespiratory failure. Although the main clinical syndrome at hospitalization was generally of respiratory or gastrointestinal origin, there were ten patients (13%) admitted to the PICU because of MIS-C. This is a new phenomenon related to COVID-19 in children and it is expected that many of these patients need monitoring in the PICU, as well as by other pediatric specialties.6
The majority of patients had their diagnosis confirmed by RT-PCR (91%), but a greater proportion of negative results in the MIS-C group was observed (50% vs. 15%). This is compatible with previous data, showing that MIS-C is a late manifestation associated with COVID-19, outside the positivity window for RT-PCR. In this group, the association with a SARS-CoV-2 infection was made in half of the patients by a positive serology, mainly IgG. However, in the non-MIS-C group, positive IgM was detected in 80% of the few cases in which it was measured. Co-detection with other viruses was observed in 15% of the patients in which it was investigated (only in non-MIS-C group), which is similar to other COVID-19 studies,5,10 and in other severe respiratory virus infections in children.17
Lymphopenia was observed in 38% of patients (50% in the MIS-C group), which has also been described in other pediatric COVID-19 studies.5,14,15,18 Although some adult studies have associated lymphopenia with a worse prognosis,19,20 this is still not clear in pediatrics. Inflammatory markers – such as ESR, CRP, LDH, D-dimer, procalcitonin, and ferritin – were elevated in most tested patients, but mainly in the MIS-C group, which is in accordance with the diagnostic criteria for this syndrome.21–23 In the present cohort, troponin, CK, and CK–MB were measured in less than 20% of patients; elevated levels were found in at least 50% of them. These cardiac injury biomarkers have also been reported as abnormal in other studies, especially in patients with some form of cardiac failure.12–14 Another known sensitive marker for heart failure detection, proBNP, was elevated in all seven patients in which it was measured in the MIS-C group, as has also been reported in other studies.6,24 The present study also measured interleukin-6 (IL-6) in two patients in the MIS-C group, both with severe cardiac dysfunction and shock; very high levels were found. Elevated levels have previously been described in critically ill pediatric patients with COVID-19.5,8,12
Another finding consistent with cardiac dysfunction in patients in the MIS-C group was pleural effusion, detected by chest radiography, a feature much less frequent in the non-MIS-C group. The other radiological findings of diffuse bilateral interstitial infiltrates and ground-glass opacities on chest X-rays and on CT were present in most patients, which is consistent with previous reports.10,18,25
As for the management of patients, the majority needed some kind of respiratory support, most of them only oxygen therapy, but about 20% needed IMV (median 7.5 days), which is within the reported range of use (18–50%) described in other studies.5,10,11,14 Of these, 71% developed ARDS, mainly severe, requiring neuromuscular blocking, high PEEP, alveolar recruitment maneuver, and prone position in some cases. Although it was not possible to confirm bacterial infections, antibiotic therapy was used in three-quarters of patients and oseltamivir in almost half, which can be explained by the national guidelines for the treatment of SARS, which indicates the initial use of empirical oseltamivir.26 Hydroxychloroquine was not prescribed, although some studies have reported its use in 7%–47% of patients.5,10,11,14 Perhaps this difference can be explained by the fact that the cases in Europe and the United States started weeks or months before those in Brazil, when a more compassionate use of drugs could be explained and also fewer studies were available.
Demographic and clinical characteristics were compared according to the presence of comorbidities, age below 1 year, and the need for IMV. Although the numbers in these subgroups were small, significant differences were found, determined by the presence of comorbidities. These patients were older and needed more respiratory support, with more cases of ARDS. The only two deaths also occurred in this group. The presence of comorbidities independently increased the chance of IMV, but the factors associated with a worse prognosis need more investigation. Obesity, which is reported as the worse prognostic factor in children with COVID-19,5,11 was not frequent in the present cohort, where the main significant comorbidities were neuromuscular diseases, chronic respiratory diseases, and cancer. Age less than 1 year, sex, and race were not associated with more severe cases in the present study, although infants had a worse clinical course in China and the United States.1,2 As MIS-C is a poorly understood disease that appears to occur in the subacute phase of SARS-CoV-2 infection, the same subgroup comparisons and the assessment of predictors of severity were made in only the non-MIS-C group, but the results did not differ. As also shown in all pediatric studies on COVID-19, the vast majority of the present patients progressed well and were discharged, with a mortality rate of just 3%.
The present study has some limitations. As it involved only Brazilian patients, this may limit the generalization of the results. In addition, some details about treatment are lacking, such as the reason for the high percentage of use of antibiotics or corticosteroids. Not all patients had inflammatory markers measured, so they could not be compared depending on the severity of the disease. Despite these limitations, it is believed that this study may contribute to a better understanding of COVID-19, as it describes the first large series of patients admitted to PICUs in the Southern Hemisphere.
In conclusion, to the best of the authors’ knowledge, this is the first study on COVID-19 in PICU patients in Brazil. It was shown that the characteristics of this disease in tropical and subtropical locations are similar to other countries. In this cohort, lethality was low, and chronic diseases and other comorbidities played an important role in the development of severe forms of the disease. Unlike other studies, the age less than 1 year was not associated with a worse prognosis. Patients with MIS had more severe symptoms, higher inflammatory biomarkers, and a greater predominance of males.
FundingThis study was supported by grants from the following Brazilian research promotion agencies:
- •
Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (National Council for Scientific and Technological Development), process No. 401597/2020-2.
- •
Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) (Carlos Chagas Filho Foundation for Research Support of the State of Rio de Janeiro), process No. E-26/010.000160/2020, grant No. 2020/0996.
The authors declare no conflicts of interests.
The authors thank the following collaborating authors, for their work and support for this study:
• Ana Carolina Cabral P Scarlato - Hospital Rios D’Or;
• Rodrigo Moulin Silva - Hospital Pedro Ernesto (UERJ);
• Camila Furtado Guedes Pinto - Hospital Pedro Ernesto (UERJ);
• Mariana Barros Genuíno de Oliveira - Instituto D’Or de Pesquisa e Ensino (IDOR);
• Jaqueline Rodrigues Robaina - Instituto D’Or de Pesquisa e Ensino (IDOR).
Please cite this article as: Prata-Barbosa A, Lima-Setta F, Santos GR, Lanziotti VS, Castro RE, Souza DC, et al. Pediatric patients with COVID-19 admitted to intensive care units in Brazil: a prospective multicenter study. J Pediatr (Rio J). 2020;96:582–92.
Study conducted at Brazilian Research Network in Pediatric Intensive Care (BRnet-PIC), Brazil.