To estimate the prevalence and factors associated with overweight/obesity development in adolescents with early diagnosed phenylketonuria treated exclusively by diet.
MethodologyIn this cross-sectional study anthropometric measurements, serum phenylalanine levels, and 10 metabolites associated with lipid and carbohydrate metabolism were analyzed in 101 adolescents aged 10–20 years. Adolescents were categorized into overweight/obesity and eutrophic/low body mass index groups. These patients were compared using Student's t-test, Pearson's chi-square test, Wald's chi-square test for multivariate analysis. Further, to verify whether the prevalence of overweight/obesity found in the study population was similar to that in the general population, the authors compared the nutritional status of 46 patients aged 13–17 years with that of healthy students of the same age from the National School Health Survey using the chi-square test for adherence. The significance threshold was p < 0.5.
ResultsThe prevalence of overweight/obesity in adolescents was 27.7%. There was no difference in prevalence between sexes. Older age was a protective factor and Increased Homeostasis Model Assessment Insulin Resistance index and high phenylalanine and low-density lipoprotein cholesterol levels were predictive factors for overweight/obesity. The equality hypothesis was not rejected in the comparison of nutritional states of 46 patients aged 13–17 years and healthy students of the same age.
ConclusionThe prevalence of overweight/obesity in phenylketonuria adolescents was similar to what is found in healthy adolescents.
Phenylketonuria (PKU) is an autosomal recessive disorder caused by deficient or absence of activity of the hepatic phenylalanine (Phe) hydroxylase enzyme that converts Phe to tyrosine. The accumulation of toxic metabolites derived from the metabolism of Phe causes severe damage to multiple tissues and organs, which is manifested by several signs and symptoms, mainly neurological in nature, resulting in intellectual disability, the most severe manifestation.
Treatment for PKU should begin immediately after its diagnosis is established by neonatal screening and should be continued throughout life. Patients receiving treatment at a late stage may experience severe consequences of elevated blood Phe levels, culminating in irreversible neurological damage and growth retardation. Early diagnosis and treatment of PKU can prevent or reduce damage to the patients, thus decreasing the incidences of severe intellectual disability and growth retardation. However, problems associated with neurocognition, psychosocial aspects, growth, quality of life, global nutrition, bone metabolism, and prevention of maternal PKU persist in patients.1
Although new therapeutic alternatives for PKU treatments are available, dietary treatment continues to play a key role in PKU management.2 The recommended diet is based on reduced ingestion of natural proteins and maintenance of carbohydrates and fats. Thus, since the 1980s and 1990s, several researchers have suggested that the nature of food contributes to the increased occurrence of overweight and obesity in patients with PKU.3-5
However, the role of dietary treatment in the development of overweight/obesity and metabolic syndrome in patients with PKU is controversial. Thus far, there is no consensus on this issue.1,4,6-12
The increasing prevalence of overweight/obesity worldwide9,13,14 is an obstacle to addressing the controversy. The Brazilian National School Health Survey (PeNSE, 2015)15 studied the frequency of overweight/obesity in all Brazilian states in a sample of 102,301 students aged 13–15 years and 10,926 students aged 16–17 years, with a maximum sampling error of 3% and a 95% confidence interval. The prevalence rate of overweight/obesity in that survey was 23.7%.
However, the authors could not find studies involving only adolescents with PKU during the authors’ literature review. Therefore, this study aimed to determine the prevalence of overweight/obesity and study factors associated with its development in adolescents with PKU who have been exclusively undergoing dietary treatment. These adolescents had been treated and monitored since the establishment of PKU diagnosis on newborn screening at the only PKU treatment center in the state of Minas Gerais, the second-most populous state in Brazil. At the time of the study, Minas Gerais had approximately 21 million inhabitants. The incidence of PKU in the state was 1:20,000. From January 1994 to February 2020, 6,345,222 newborns had been screened, and 376 patients with PKU were being followed up.16
Material and methodsThis was a cross-sectional study involving adolescents with PKU who were regularly monitored since their early diagnosis by a multidisciplinary team at the Special Genetics Service Phenylketonuria Outpatient Clinic at the Clinical Hospital, Minas Gerais, Brazil, and the Center for Newborn Screening and Genetics Diagnosis of the Faculty of Medicine Federal University of Minas Gerais (NUPAD/FM/UFMG). These institutions are responsible for newborn screening, diagnosis, treatment, and monitoring of PKU in the state of Minas Gerais.
All patients with PKU aged 10–20 years who underwent neonatal screening tests and had a Phe level of > 600 µmol/L before 21 days of life in Minas Gerais were eligible to participate in the study. Patients with late diagnosis and patients diagnosed with tetrahydrobiopterin (BH4) deficiency were excluded accordingly. Patients with Phe levels between 240 and 600 µmol/L at diagnosis were followed systematically without dietetic treatment until they completed 6 years, and then the follow-up was terminated for boys, but the girls were advised to come back when they were 12 years old to evaluate the risk of maternal PKU. These girls were also excluded accordingly. During the study period, from January 2017 to December 2018, 128 adolescents aged between 10 and 20 years were precociously diagnosed with PKU. Of these adolescents, three adolescents were excluded—one was a drug user, one was a pregnant adolescent, and one discontinued treatment. Seven patients refused to undergo the necessary procedures to participate in the research. Seventeen patients had changed their address and, hence, could not be reached. Finally, a total of 101 adolescents participated in the study.
All patients underwent weight, height, and waist circumference measurements. The nutritional status of patients aged >18 was assessed by calculating their body mass index (BMI) and classifying it according to the World Health Organization (WHO) criteria.17 Patients aged ≤18 years were classified according to the curves established by the WHO:18 overweight, Z-score >1 and ≤2; obesity, Z-score >2; eutrophic, Z-score ≥-2 and ≤1; and low BMI, Z-score <-2. Waist circumference was classified according to the criteria established by Freedman et al.19 The patients were divided into two groups according to the nutritional status—the overweight/obesity group and the eutrophic/low BMI group.
Food consumption was assessed using the quantitative Food Frequency Questionnaire (FFQ), validated by Colucci et al.20 and adapted by Alves et al.21 for patients with PKU since there was no validated food questionnaire for this population. The Dietpro software version 5i was used for FFQ calculations based on the Brazilian Food Composition Table22 for the consumption of total calories, carbohydrates, proteins, and lipids. The intake of Phe was evaluated using the North American Table.23 Blood samples were collected to determine the levels of total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), triglycerides, ultra-sensitive C-reactive protein (CRP), alkaline phosphatase (AP), fasting glucose, and basal insulin. The Homeostasis Model Assessment Insulin Resistance (HOMA-IR) index was calculated using the mathematical model proposed by Matthews et al.24 and the cutoff point (≤ 3.16) proposed by Keskin et al.25 was chosen.
Disease control was assessed using the mean serum Phe levels measured during adolescence and was considered inadequate when the levels were outside the reference range (≥ 120 and ≤ 700 µmol/L) for the age groups studied.26 A total of 4,447 Phe measurements were performed during adolescence (mean 44 measurements/patient, median 35 measurements/patient).
Further, to verify whether the prevalence of overweight/obesity found in the study population was similar to that in the general population, the authors compared the nutritional status of 46 patients aged 13–17 years with that of healthy students of the same age from the PeNSE (2015).15 This survey studied the frequency of overweight/obesity in all Brazilian states using a sample of 102,301 students aged 13–15 years and 10,926 students aged 16–17 years; the maximum sampling error was 3% with a 95% confidence interval. The prevalence rate of overweight/obesity in the survey was 23.7%.
Data are expressed as absolute numbers, percentages, and means ± standard deviations. For the univariate analysis, Student's t-test was used for continuous variables and Pearson's chi-square test was used for categorical ones. Multivariate analysis using logistic regression was performed for variables with a p-value of <0.40 in univariate analysis. To compare the nutritional status of patients aged 13–17 years with that of healthy students of the same age as the PeNSE (2015),15 the authors used the chi-square test for adherence. The level of significance was set at p < 0.05.
A consent form to participate in this research was signed by the adolescent and his/her legal guardian. The study was approved by the Research Ethics Committee of the Federal University of Minas Gerais (COEP/UFMG) and supported by NUPAD/FM/UFMG.
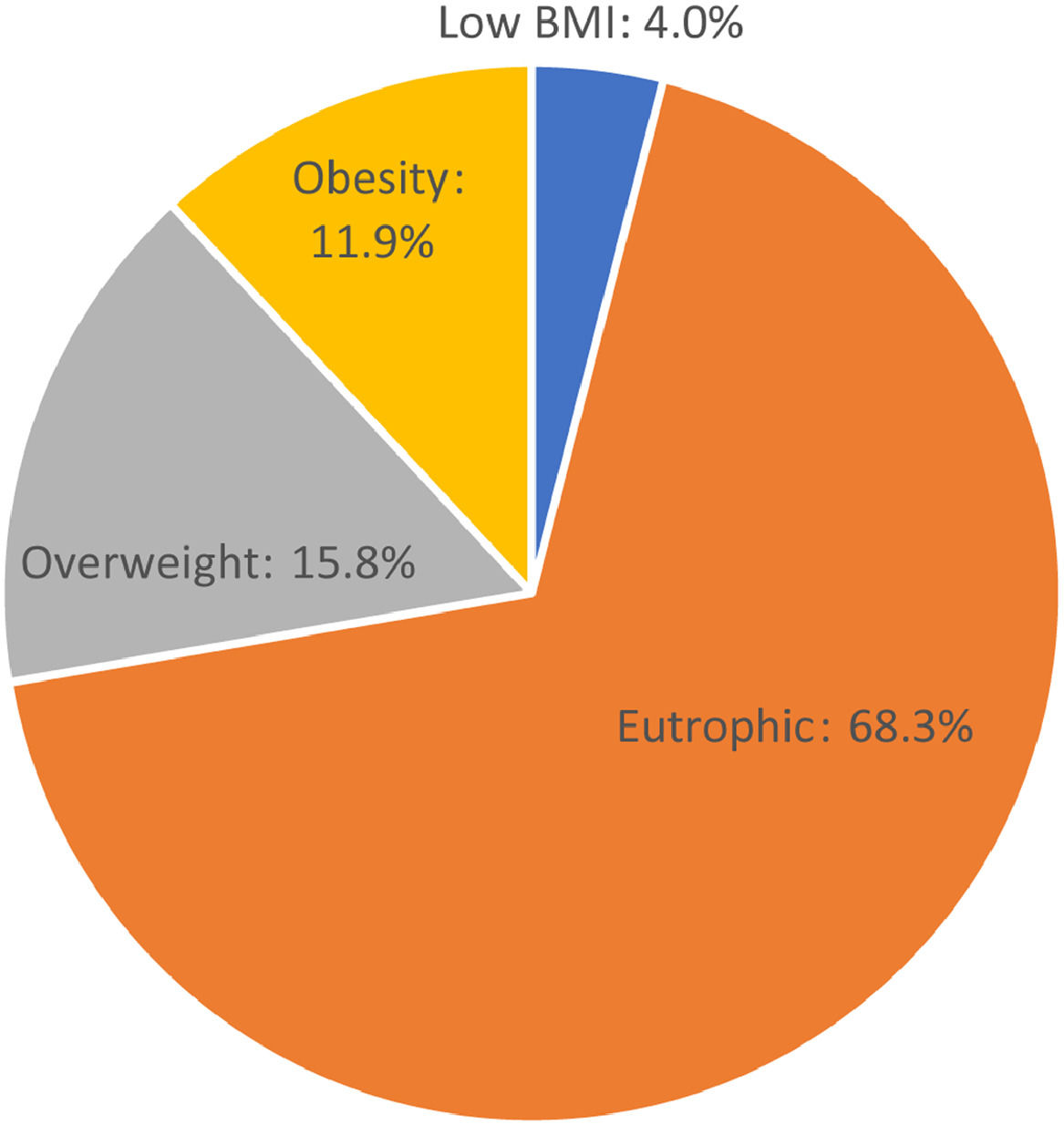
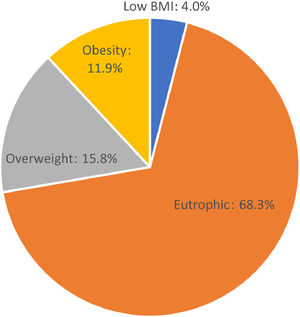
ResultsOf the 101 adolescents, 55 (54.5%) were male and 46 were female. Seventy-three (72.3%; 41 male and 32 female) adolescents were eutrophic or had low BMI. Twenty-eight (27.7%; 14 male and 14 female) adolescents were overweight or had obesity (Fig. 1). There was no significant difference regarding the presence of overweight/obesity between the sexes (p = 0.58). Of the adolescents analyzed, 57 (56.4%) had classic PKU and 44 (43.6%) had mild PKU. There was no significant difference in the nutritional status between adolescents with mild and classic PKU (p = 0.929). The results of the comparative analysis of the nutritional status and the studied variables are shown in Table 1.
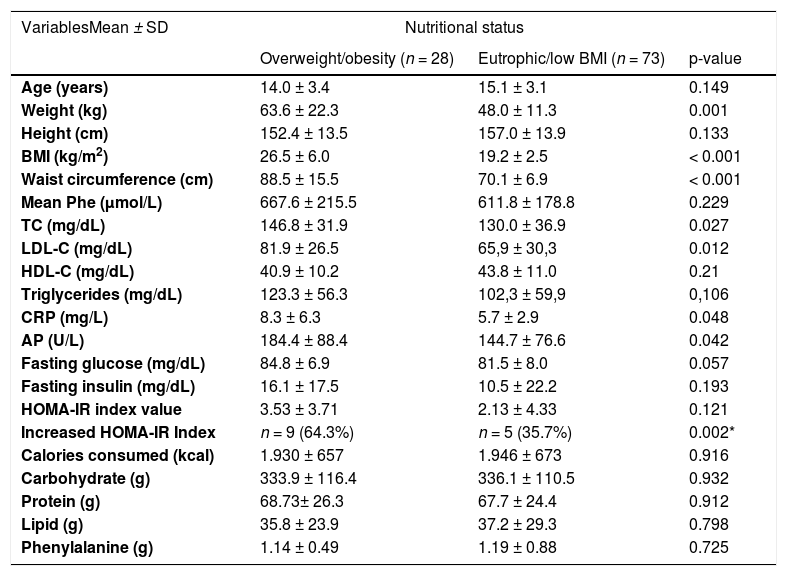
- Association between nutritional status and anthropometric variables, biochemical test results, and food consumption.
p, Student's t-test for independent samples; p*, Pearson's chi-square test
BMI, Body mass index; TC, total cholesterol; LDL-C, low-density lipoprotein cholesterol; HOMA-IR, Homeostasis Model Assessment Insulin Resistance; HDL-C, high-density lipoprotein cholesterol; CRP, C-reactive protein; AP, alkaline phosphatase
Univariate analysis showed significant differences in BMI (p = 0.001); waist circumference (p < 0.001); levels of TC (p = 0.027), LDL-C (p = 0.012), CRP (p = 0.048), AP (p = 0.042); and HOMA-IR index (p = 0.002) between the overweight/obesity and eutrophic/low BMI groups by age.
There was no difference in nutrient intake between the groups. The consumption rates of carbohydrates, proteins, and lipids in the overweight/obesity group were 69.2%, 14.2%, and 16.6%, respectively, while those in the eutrophic/low BMI group were 69%, 13.8%, and 17.2%, respectively.
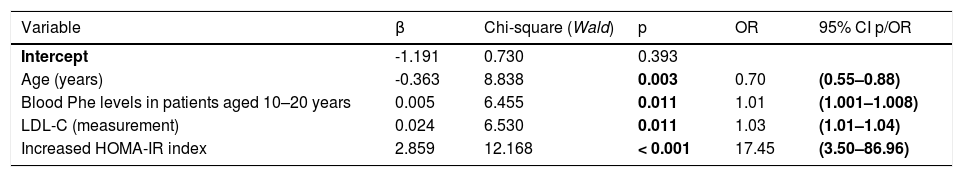
Table 2 shows the final logistic regression model, which indicated that high serum Phe and LDL cholesterol levels and HOMA-IR index were predictive factors for overweight/obesity and that older age was a protective factor for overweight/obesity.
- Final logistic regression analysis model.
Database: 95 children (overweight/obesity, 25 children; eutrophic/low BMI, 70 children). Four children had no information on the HOMA-IR index, and two were considered as outlier cases with regard to the HOMA-IR index (sensitivity = 48.0%, specificity = 88.6%, false-positive rate = 40.0%, false-negative rate = 17.3%, correct results rate = 77.9%).
LDL-C, low-density lipoprotein cholesterol; HOMA-IR, Homeostasis Model Assessment Insulin Resistance
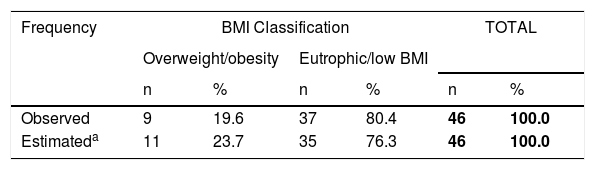
Table 3 shows the comparison of the prevalence of obesity/overweight in patients with PKU aged 13–17 years and in healthy students of the same age from the PeNSE.15 The equality hypothesis was not rejected (p = 0.488).
- Chi-square test for adherence between the observed and estimated results of the Brazilian National School Health Survey (PeNSE, 2015) 15 with regard to the prevalence of overweight/obesity.
| Frequency | BMI Classification | TOTAL | ||||
|---|---|---|---|---|---|---|
| Overweight/obesity | Eutrophic/low BMI | |||||
| n | % | n | % | n | % | |
| Observed | 9 | 19.6 | 37 | 80.4 | 46 | 100.0 |
| Estimateda | 11 | 23.7 | 35 | 76.3 | 46 | 100.0 |
Database: 46 patients.
Chi-square test for adherence: p = 0.488.
The percentages were calculated in relation to the total of the cases and percentages.
Values extracted from reference article “The Brazilian National School Health Survey (PeNSE, 2015)” 15, in which the numbers were calculated based on the prevalence of overweight/obesity of 23.7%.
Two aspects distinguish this study. First, it was conducted at a single center in a populous state. Second, the study exclusively enrolled adolescents. Since this study was performed at a single reference center, there was greater socio-cultural and environmental homogeneity in the studied sample and decreased the chances of investigation bias compared to those in multicenter studies4,7,8,11.
By studying only adolescents, the study allows a better assessment of overweight/obesity at this stage of life, which plays an important role in its prevention in adulthood and should be considered in their follow-up.
There were several published studies on overweight/obesity in only children.2,3,5,12 Some studies also involved children and/or adolescents and adults.4,6,8,10,27,28 The other studies only included adults.7,11 However, studies involving only adolescents with overweight/obesity were not found in the literature.
Another aspect to be considered was the sample size. Due to the low prevalence of the disease, most studies on this topic were multicenter studies, among which most were retrospective ones.4,7,8,11 Only three retrospective multicenter studies had a larger sample size than the present study; all these studies involved children who participated in the Collaborative Study of Children Treated for Phenylketonuria in the USA.5,29,30 Only one single-center study had a larger sample than the present study. However, the study included children, adolescents, and adults with Phe levels between 360 and 600 µmol/L at diagnosis.6 Hence, the sample size of this study was considered suitable to study the research question.
The large number of Phe measurements performed showed an adequate patient follow-up and increased the credibility of the study results.
The assessment of overweight/obesity in PKU presents several difficulties due to the recent increase in the prevalence of obesity worldwide in all age groups and because of the different factors involved in this increment.9,13,14 A similar phenomenon has been observed in Brazil, and it is difficult to define whether the increase in the prevalence of overweight/obesity in the present study is a result of factors intrinsic to PKU, such as diet and its control; biochemical determinants; or other factors affecting society as a whole. In the present study, mild or classic PKU did not influence the nutritional status of patients.
Moreover, neither the percentage of the nutrients consumed nor the total caloric consumption was significantly different between the groups. Genetic determinants and distinct lifestyles, which were outside the scope of this study, may be related to weight differences.
The prevalence rate of overweight/obesity (27.7%) in this study was lower than that reported by Walkowiak et al.12 and Couce et al.8 However, it was higher than the prevalence rate of 22% in a population of patients with PKU in Southern Brazil, reported by Mazzola et al.;27 however, they had evaluated a smaller sample and a wider age group (27 patients aged 5–27 years). The significance of these differences could not be established due to the difference in methodologies and characteristics of the samples.
The comparison of the prevalence of overweight/obesity among patients aged 13–17 years old and healthy students of the same age of PeNSE (2015)15 did not rule out the equality hypothesis. It suggests that the prevalence of overweight/obesity among adolescents with PKU reflects the prevalence of overweight/obesity among healthy Brazilian schoolchildren and does not seem to be due to the disease or its treatment.
Other studies have found no significant differences in the prevalence of overweight and obesity among patients with PKU and the general population.28,31 Belanger-Quintana and Martínez-Pardo6 reported a tendency toward obesity only in the most severe phenotypes; however, most other studies have not reported differences among phenotypes.
Several studies have reported a higher prevalence of overweight/obesity in women with PKU than in men with PKU,4,5,11,12,29 a result not observed in this study. Although the absolute percentage of overweight/obesity in female patients was higher than that in male patients, this difference was not significant.
The final logistic regression model was not robust. The 95% confidence intervals for age as a protective factor and for increased HOMA-IR index as a predictive factor for overweight/obesity were adequate; nevertheless, the lower values of 95% CI for blood Phe and LDL-C levels were very close to unity. The sensitivity of the final model was 48%, which allowed a false-positive rate of 40%, while the specificity was 88.6%, allowing a false-negative rate of 17.3%. However, as a whole, it allowed a correct results rate of 77.9%.
Older age as a protective factor for overweight/obesity is in agreement with the findings of the PeNSE (2015),15 which reported a significantly higher prevalence of overweight/obesity among adolescents aged 13–15 years than in those aged 16–17 years. This suggests that the patterns of overweight/obesity among adolescents with PKU reflect the behavior of overweight/obesity in Brazilian adolescents.
The association between increased HOMA-IR index and overweight/obesity in hyperphenylalaninemia (HPA) has already been explored in the literature. Couce et al.8 reported that higher HOMA-IR index values were associated with BMI, waist circumference, and age in patients with HPA and BH4 deficiency than in controls. This was attributed to the high caloric intake of carbohydrates. Kanufre et al.10 studied 29 overweight and 29 eutrophic patients with PKU aged 4–15 years and reported significantly higher HOMA-IR index values in overweight/obesity patients than in eutrophic patients. Rocha et al.28 reported no differences in HOMA-IR index values between patients with PKU and controls; these values increased in both groups when in the presence of central obesity.
In the present study, although the serum levels of TC, HDL-C, and LDL-C were within the normal ranges in both groups, LDL-C levels were significantly higher in the overweight/obesity group than in the eutrophic/low BMI group. In contrast, a study by Schulpis et al.,32 involving 44 patients with good diet control and 42 with poor adherence to the diet, reported higher LDL-C serum levels in patients with well-controlled PKU than in patients with uncontrolled PKU and in controls.
Higher Phe levels, as a predictive factor for overweight/obesity, although weakly associated, possibly reflected worse adherence to the treatment. It has reinforced the well-established concept that patients with PKU need lifelong dietary control. Other studies have reported that overweight/obesity is associated with high Phe levels, non-adherence to treatment, and poor metabolic control.12,28
ConclusionThis study reinforced the concept that the prevalence of overweight/obesity in adolescents with PKU is similar to that in other adolescents and that it is not associated with the disease or dietary treatment. The results also suggested that older age was a protective factor for overweight/obesity and that increased HOMA-IR index and high serum Phe and LDL-C levels were predictive factors for overweight/obesity.
FundingCenter for Newborn Screening and Genetics Diagnosis of Faculty of Medicine, Federal University of Minas Gerais (NUPAD/FM/UFMG), Belo Horizonte, Brazil provided financial support for this research.
The authors would like to thank the NUPAD/FM/UFMG for the logistical and financial support for conducting the research. The authors also thank the adolescents, as well as their parents or guardians, who voluntarily participated in the research.