To evaluate outcomes of oral food challenge (OFC) test to assess tolerance in infants with non-IgE-mediated cow's milk allergy (CMA) with gastrointestinal manifestations and explore clinical data predictive of these outcomes.
MethodsSingle-center retrospective study including infants (age < 12 months) who were referred for CMA between 2000 and 2018 and underwent OFC on follow-up. A univariate logistic regression test was performed to evaluate variables associated with the outcomes of the follow-up OFC test.
ResultsEighty-two patients were included, 50% were male. Eighteen patients had a positive OFC test (22%). Most patients had presented with hematochezia (77%). The median age of symptom onset was 30 days. Two-thirds of the patients were on appropriate infant formula (extensively hydrolyzed or amino acid-based formula), exclusively or in association with breastfeeding. The median time on an elimination diet before the OFC test was 8 months (Q1 6 – Q3 11 months). All cases with positive follow-up OFC tests (n = 18) had been exposed to cow's milk-based formula before the first clinical manifestation of CMA. Five out of eight cases with Food Protein-Induced Enterocolitis Syndrome (FPIES) had positive OFC tests. Exposure to cow's milk-based formula before diagnosis, a history of other food allergies, hematochezia and diarrhea were predictors of a positive OFC test.
ConclusionsIn infants with non-IgE-mediated CMPA with gastrointestinal manifestations, the use of cow's milk-based formula, a history of other food allergies, and hematochezia and diarrhea upon initial presentation were associated factors for the later achievement of tolerance.
Non-IgE-mediated cow's milk allergy (CMA) is a common food allergy during childhood, typically presenting in infancy,1,2 more often with gastrointestinal manifestations.3 Symptoms vary and are non-specific, which is a problem for recognition by pediatricians and pediatric gastroenterologists who deal with this condition.3 There are different presentations of non-IgE-mediated CMA, including food protein-induced enteropathy, enterocolitis, or proctocolitis. Allergic proctocolitis is the most common presentation, with mild rectal bleeding in an infant who is typically otherwise well and does present with faltering growth. In contrast, Food Protein-Induced Enterocolitis Syndrome (FPIES) is more rare and severe – acute FPIES typically presents in infants ages 4 to 12 months, 1 to 4 h after the ingestion of the protein, with profuse vomiting, pallor, and lethargy, and it can progress to dehydration and shock, while chronic FPIES has been described with persistent vomiting, diarrhea, and/or failure to thrive.4–7
The majority of non-IgE-mediated cow's milk protein allergies are transient and resolve in early childhood. The achievement of tolerance varies according to the form of presentation: allergic proctocolitis is usually associated with the earliest development of tolerance, generally before the first year of life. Meanwhile, FPIES tend to resolve later and the time to achieve tolerance in patients varies according to the food involved. In patients with FPIES induced by cow's milk protein, tolerance is typically acquired around 3 years of age. It has been described in the literature that up to 20% of exclusively breastfed infants have spontaneous resolution of the allergy without the mother being on an exclusion diet, however, factors associated with the acquisition of tolerance are rarely described.4,8
The oral food challenge (OFC) test is the gold standard for confirmation of the diagnosis of non-IgE-mediated allergies, and also on the follow-up to assess for the development of tolerance.5,8,9 To confirm the diagnosis of non-IgE-mediated CMA, it is recommended to exclude cow's milk protein (CMP) for 2-4 weeks before performing the OFC test.10 In patients who present with mild to moderate symptoms, the test can be performed at home.6,10 In cases with severe manifestations, such as suspected FPIES, the OFC test should be performed in a hospital environment.6,10,11 To evaluate the achievement of tolerance, the test is performed after 6 months of exclusion diet or at 12 months of age (whichever is earlier).4,8
There is extensive medical literature on the prevention, diagnosis and treatment of non-IgE-mediated cow's milk protein allergies, and it is known that the acquisition of tolerance occurs in most cases around 12 months. However, there is little data regarding factors that interfere with the development of tolerance in children with CMA. The aims of this study were to assess outcomes of the oral food challenge (OFC) test to assess the development of tolerance in infants with non-IgE-mediated cow's milk allergy (CMA) with gastrointestinal manifestations and to explore clinical data associated with these outcomes.
MethodsThis single-center retrospective study was conducted with infants (age < 12 months) with a diagnosis of CMA referred to the outpatient clinic of the pediatric gastroenterology unit of a university-affiliated hospital in Brazil, from 2000 to 2018. Inclusion criteria were (all three must have been fulfilled): 1) having a clinic diagnosis of non-IgE mediated CMA with gastrointestinal manifestations in the first year of life; 2) being on a CMP-free diet for at least 8 weeks; and 3) having undergone OFC test on follow-up to assess the development of tolerance. Exclusion criteria aimed to omit infants receiving inappropriate restricted diets if they had been continuously exposed to CMP and/or breasted infants whose mothers had not been on a strict CMP-free diet.
Data were collected on age at symptoms onset, gender, gestational age, comorbidities and associated allergic diseases, clinical presentation, CMP-free diet, and outcomes of OFC test. According to the symptoms, the cases were classified as mild-to-moderate vs. severe.10
The OFC test was performed in three different ways, individualizing each case:
Home protocolFor infants who were exclusively breastfed, with the breastfeeding mother being on an appropriate CMP elimination diet, the OFC test was performed with home reintroduction of CMP.11 The mother received education on how to progressively and safely reintroduce dairy foods, with careful observation of signs and symptoms. Contact information to reach the team was provided and patients were followed up closely in the outpatient clinic.
Hospital protocolIf the infant had been on appropriate formula for CMA, the OFC test was performed in a hospital environment, under the supervision of a specialized medical team, following the most up-to-date OFC test protocol at the time, most recently similar to the one described by Vandenplas et al.11: Initially, gauze was soaked with an intact cow's milk protein-based formula and gently applied to the patient's lips. If they remained asymptomatic after 15 min, a formula containing intact cow's milk protein was administered orally and the dose was gradually increased (0.5, 1, 3, 10, 30, 50 to 100 mL) every 30 min. Patients remained under observation for two hours after the last dose of formula was given.
FPIES protocolIn cases of FPIES, the OFC test protocol was also performed in a hospital environment, carried out with a different protocol than the one previously described, following the most up-to-date OFC test protocol for FPIES at the time, most recently similar to the one published by Nowak-Węgrzyn et al.12: all patients had peripheral venous access inserted before the test was initiated, and the dose of cow's milk protein varied from 0.06 to 0.6 g of protein/kg (average of 0.3 g/kg), divided in 3 equal doses, given every 30 min. A limit of a maximum of 3g in each step or a maximum of 10g in the entire test was followed. All patients remained under observation for at least 4 h after the last dose of formula was given.
In all cases, after the end of the test, if no symptoms were identified by the end of the observation period, the patient was discharged home. A follow-up telephone call always took place in the next day to assess for late symptoms, and patients had a follow-up appointment after one week.
The test was considered:
- -
positive in the presence of immediate signs and symptoms (the same as the patient initially presented), persistence or increase in the intensity of subjective symptoms, or if late symptoms were reported by caregivers.
- -
negative: in the absence of symptoms.
The patients were stratified according to the outcome of the follow-up OFC test: positive vs. negative (development of tolerance). A univariate logistic regression analysis was performed to evaluate variables associated with the outcomes of the follow-up OFC test. For all analyses, a p-value < 0.05 was considered statistically significant. The 95% confidence intervals are presented. The data were analyzed using IBM Statistic SPSS (IBM SPSS Statistics for Windows. NY, EUA).
The study was conducted according to the Declaration of Helsinki, and ethical approval was obtained from the Ethics Committee of the University of Campinas (Approval number 2.986.745)
ResultsInitially, a total of 205 medical records were analyzed, but only 82 patients were enrolled according to established inclusion and exclusion criteria. Of these, 41 were male (50%), 10% had atopic conditions (asthma, rhinitis and atopic dermatitis), and the same percentage had allergies to foods other than CMA. Fifty-three percent had a history of prematurity.
The median age at the first clinical manifestation was 30 days (Q1 25 – Q3 60 days), and by the time of the follow-up OFC test, these patients had been on a CMP elimination diet over a median time of 8 months (Q1 6 – Q3 11 months). The clinical characteristics of infants referred with a diagnosis of cow's milk allergy and who underwent OFC test, stratified according to the outcome of OFC test are shown in Table 1.
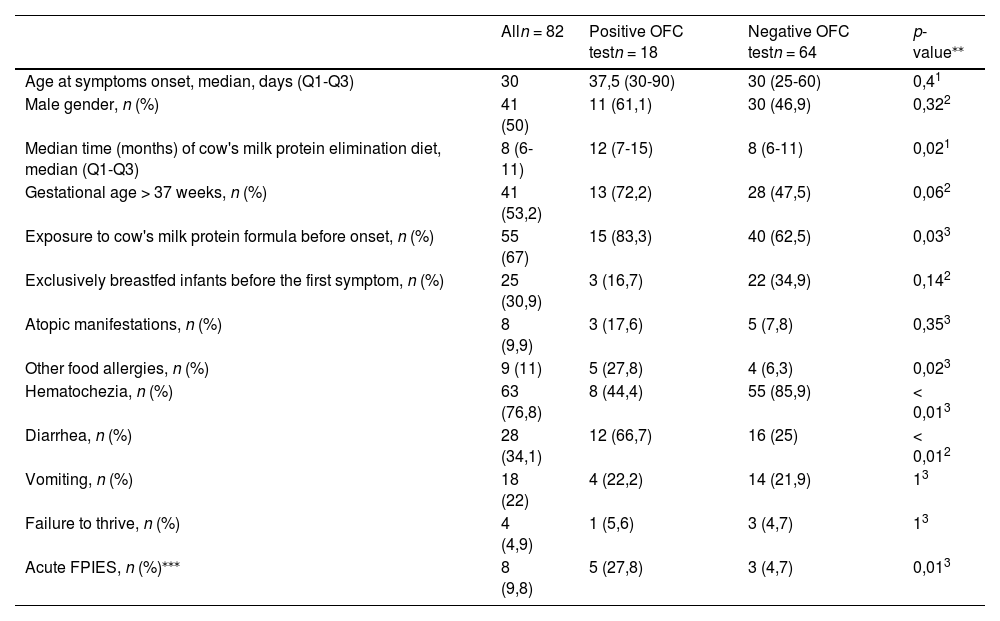
Clinical characteristics of infants referred with a diagnosis of cow's milk allergy and who underwent oral food challenge (OFC) test, stratified according to outcome of OFC test.
| Alln = 82 | Positive OFC testn = 18 | Negative OFC testn = 64 | p-value⁎⁎ | |
|---|---|---|---|---|
| Age at symptoms onset, median, days (Q1-Q3) | 30 | 37,5 (30-90) | 30 (25-60) | 0,41 |
| Male gender, n (%) | 41 (50) | 11 (61,1) | 30 (46,9) | 0,322 |
| Median time (months) of cow's milk protein elimination diet, median (Q1-Q3) | 8 (6-11) | 12 (7-15) | 8 (6-11) | 0,021 |
| Gestational age > 37 weeks, n (%) | 41 (53,2) | 13 (72,2) | 28 (47,5) | 0,062 |
| Exposure to cow's milk protein formula before onset, n (%) | 55 (67) | 15 (83,3) | 40 (62,5) | 0,033 |
| Exclusively breastfed infants before the first symptom, n (%) | 25 (30,9) | 3 (16,7) | 22 (34,9) | 0,142 |
| Atopic manifestations, n (%) | 8 (9,9) | 3 (17,6) | 5 (7,8) | 0,353 |
| Other food allergies, n (%) | 9 (11) | 5 (27,8) | 4 (6,3) | 0,023 |
| Hematochezia, n (%) | 63 (76,8) | 8 (44,4) | 55 (85,9) | < 0,013 |
| Diarrhea, n (%) | 28 (34,1) | 12 (66,7) | 16 (25) | < 0,012 |
| Vomiting, n (%) | 18 (22) | 4 (22,2) | 14 (21,9) | 13 |
| Failure to thrive, n (%) | 4 (4,9) | 1 (5,6) | 3 (4,7) | 13 |
| Acute FPIES, n (%)⁎⁎⁎ | 8 (9,8) | 5 (27,8) | 3 (4,7) | 0,013 |
*Statistics presented: n (%); Median (Q1-Q3).
At the time of referral, 28% of the infants were fed with an extensively hydrolyzed formula, 24% with an amino acid-based formula, 16% were exclusively breastfed with their mothers on a CMP-restricted diet, 12% were using soy-based formula and 20% were using non-recommended beverages, such as juices, soy-based beverages or milk from other mammals.
Of the 82 follow-up OFC tests, 22% were positive. The univariate logistic regression evaluation is presented in Table 2. All patients who were on exclusive breastfeeding with their mothers on a restricted diet (13/82) had negative OFC tests. Patients who were on exclusive breastfeeding (with the mother on a CMA-restricted diet) achieved tolerance at a median age of 6 months (Q1 6.6 - Q3 9), while infants who were not exclusively breastfed achieved tolerance later, at a median age of 10.5 months (Q1 4.5 - Q3 10).
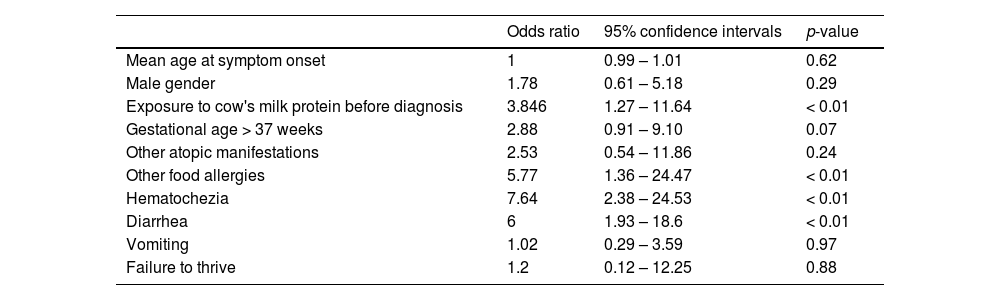
Univariate logistic regression analysis for clinical variables associated with the failure to achieve tolerance to cow's milk protein.
It is evident that patients who had a higher percentage of positive oral food challenge (OFC) tests were fed with cow's milk protein (CMP) based formula before the first clinical manifestation of non-IgE mediated cow's milk allergy (CMA), compared to patients who had a negative OFC test. Additionally, all patients whose symptoms began while exclusively breastfeeding had a negative follow-up OFC test.
Among the OFC tests carried out with infants who were exclusively breastfed, none of the tests were positive. For this group, the median time of maternal restricted CMP diet was 6 months.
DiscussionThere is scarce data in the literature on factors that influence the achievement of tolerance in CMA, especially in non-IgE-mediated cases.13,14 It is known that compared to IgE-mediated CMA, non-IgE-mediated cases are generally milder and resolve earlier, as most manifest as allergic proctocolitis.13 In IgE-mediated CMA, some factors such as the severity of the family history of atopic diseases, later onset of symptoms, presence of other food allergies and/or other allergic diseases have been associated with cases of persistent CMA.15
In our study, 78% of children referred to a tertiary care center with a diagnosis of non-IgE-mediated CMA had already acquired tolerance (negative OFC tests). Exclusive breastfeeding (EBF) seems to help to induce tolerance for CMA: most patients with positive OFC had received CMP-based formula before the first manifestation of CMA and patients who were on exclusive breastfeeding with their mother on an exclusion diet achieved tolerance about 3 months earlier than those who were not.
There is some contradictory data in the literature on how dietary factors may influence the occurrence of food allergies, but it seems that EBF has a protective effect against allergies.14,16,17 Some studies evaluating this benefit have shown a reduction in the incidence of CMA in infants up to 18 months and atopic dermatitis up to three years of age.18 Meanwhile, the early use of CMP-based formulas has been associated with intestinal dysbiosis, which is a risk factor for food allergy.18,19 Furthermore, it has been hypothesized that breast milk contains anti-allergenic properties and antibodies that induce tolerance to food antigens.19
The median age at the first clinical manifestation reported in our study was 30 days. In the study by Senocak et al. the median age of the onset of symptoms was 60 days, slightly lower than the authors found in the present study, but both are in agreement with previous literature published – in the first six months of life).20 The authors hypothesize that the higher age reported in the present study results from the referral of premature infants after discharge from the neonatal intensive care unit at the present study's own center. In keeping with our findings, Kaya et al. reported hematochezia and diarrhea as the main manifestations of non-IgE-mediated CMA.21
In the present study, the occurrence of hematochezia and diarrhea as symptoms were associated with the persistence of CMA, while manifestations of vomiting, dehydration, malnutrition, and others (crying, irritability and regurgitation) were not. In a study carried out by Topal et al., children with proctocolitis and abdominal discomfort had an earlier development of tolerance compared to children who had IgE-mediated symptoms. The present study only evaluated children with non-IgE-mediated gastrointestinal symptoms, which justifies the difference in results.13
One-third of the positive OFC had a diagnosis of FPIES. In FPIES it is mandatory to perform the OFC test in a hospital environment, as the authors described.12,22 Among the patients with FPIES, two-thirds had a positive OFC test, and all the patients were exposed to the CMP-based formula before the first manifestation. These findings are in agreement with previously published studies – the emergence of FPIES in patients who are exclusively breastfed is rare, with few cases reported in the literature.23,24
There was no association between prematurity and the positivity of the OFC test. It has been reported that no significant difference between the occurrences of cow's milk reactions in the first year of life among premature infants compared to term infants, with preterm infants having the onset of symptoms at the same age as term children, as well as a similar presentation and outcome.20,25,26
The authors did not find an association between atopic diseases (asthma, allergic rhinitis, and atopic dermatitis) and the positivity of the OFC test. This association is well seen in cases of IgE-mediated allergies (Topal et al). However, the prevalence of other food allergies was higher among patients who did not develop tolerance, similar to the study by Topal et al., in which the presence of food allergies was considered an independent factor for the persistence of food allergy.18,27–30
Continued breastfeeding after the diagnosis of CMPA has been demonstrated to be beneficial. Unfortunately, the authors report a high rate of weaning infants who were on EBF prior to the first manifestations of non-IgE-Mediated CMPA with gastrointestinal manifestations. Breastfeeding not only prevents CMPA, but it is also associated with milder manifestations, shorter time to develop tolerance and lower costs with formulas. Continued breastfeeding should be even more stimulated in the context of CMPA.27
The authors also observed that of the 25 infants who presented symptoms while on EBF, only 13 were still EBF at the time of the OFC test – it is possible that the diagnosis of CMPA and the CMP-elimination diet may have interfered with the maternal ability to continue to breastfeed. Interestingly, none of the patients who remained breastfed had a positive OFC test.
Despite the limitations of being a single-center retrospective study, small sample, and the diagnosis of CMPA being clinical (possible selection bias), the present conclusions are that in cases of non-IgE-mediated CMPA with gastrointestinal manifestations in infants, exposure to cow's milk-based formula, a history of other food allergies, hematochezia and diarrhea were associated factors for the later achievement of tolerance. Maintaining breastfeeding should be the focus in the management of CMPA.
Funding sourceNo funding was secured for this study.
Contributors statementAll authors approved the final version as submitted and agreed to be accountable for all aspects of the work.