To estimate asthma prevalence, severity, and associated factors in adolescents who live in a low relative humidity environment.
MethodsIn this cross-sectional study, adolescents aged 13–14 years from the city of Petrolina located in the Brazilian semiarid region answered the International Study of Asthma and Allergies in Childhood (ISAAC) questionnaire. The possible explanatory variables of the study were gender, family income, mother's education, smokers in the household, parental history of asthma, personal history of allergic rhinitis or atopic dermatitis, and physical activity level. Poisson regression analysis was used to assess the association between asthma and the explanatory variables.
ResultsA total of 1591 adolescents participated in the study, of whom 49.7% were male. The prevalence of active asthma, severe asthma, and physician-diagnosed asthma were 14.0%, 10.4%, and 17.8%, respectively. Adolescents with asthma missed more school days than their peers (33 vs. 22 days/year; p<0.03). Associated factors that remained significant after adjustment were history of asthma in parents (PR=2.65, p<0.001) and personal diagnosis of allergic rhinitis (PR=1.96, p<0.001) and/or atopic dermatitis (PR=2.18, p<0.001).
ConclusionAsthma prevalence in this low-humidity environment was lower, but more severe than those reported in other Brazilian cities. The dry climate might hamper disease control and this may have contributed to the higher school absenteeism observed. The association of asthma with allergic rhinitis and atopic dermatitis as well as a history of asthma in parents suggests that atopy is an important risk factor for asthma in this population.
Estimar a prevalência, a gravidade e os fatores associados à asma em adolescentes que vivem em uma região de baixa umidade relativa do ar.
MétodosEstudo transversal em adolescentes de 13 e 14 anos do semiárido brasileiro. Os participantes responderam ao questionário International Study of Asthma and Allergies in Childhood (ISAAC). As variáveis explanatórias do estudo foram sexo, renda familiar, escolaridade da mãe, fumante na residência, antecedente de asma nos genitores, antecedentes de rinite alérgica, dermatite atópica e nível de atividade física. A análise de regressão de Poisson foi utilizada para avaliar a associação entre a asma e as variáveis explanatórias.
ResultadosParticiparam da pesquisa 1.591 adolescentes, sendo 49,7% do sexo masculino. As prevalências para asma em atividade, asma grave e diagnóstico médico de asma foram de 14,0%, 10,4% e 17,8%, respectivamente. Adolescentes asmáticos faltaram mais as aulas do que seus pares (33 vs 22 aulas/ano; p<0,03). Fatores associados que permaneceram significantes após ajuste foram antecedentes de asma nos genitores (RP=2,65, p<0,001), rinite alérgica (RP=1,96, p<0,001) e/ou dermatite atópica (RP=2,18, p<0,001).
ConclusãoNeste ambiente de baixa umidade foram observadas menor prevalência, mas maior gravidade da asma que aquelas relatadas em outras cidades brasileiras. O clima seco talvez possa dificultar o controle da doença e isso pode ter contribuído para o maior absenteísmo escolar nos doentes. A associação entre rinite alérgica, dermatite atópica e antecedentes de asma nos genitores sugerem que a atopia é importante fator de risco para a asma nesta população.
Asthma is a common chronic inflammatory airway disease in childhood, clinically characterized by recurrent episodes of wheezing, breathlessness, and coughing.1 It is quite prevalent worldwide and climatic factors, especially humidity, can have an important role in disease development, considering the low prevalence found in desert regions when compared to regions where humidity and rainfall are higher, as in tropical regions.2–7
This characteristic may be related to the proliferation of house dust mites, which cannot survive in low-humidity environments and are a major determinant of allergic sensitization.4–10 In this case, in dry climate regions, other agents rather than mites can be responsible for triggering asthma; they include other airborne allergens such as pollen, air pollution, small particles present in soils, and animal epithelia, in addition to the use of humidifiers in homes.4,8,9
In Brazil there is still scarce research on asthma and risk factors in different regions of the country, especially regarding the different ecosystems.2,11 This fact makes it difficult to plan and carry out public policies aimed at prevention, as the existing studies were mainly performed in larger municipalities, especially in capital cities, most of which are located on the coast or in subtropical regions in the South/Southeast states.2,11
Thus, the aim of the study was to evaluate the prevalence, severity, and factors associated with asthma in adolescents aged 13–14 years residing in low-humidity regions, with hot and dry climate. The study was carried out in a municipality whose climate is classified as semiarid, characterized by scarce and irregular rainfall and high temperatures.
MethodsThis was a cross-sectional, population-based study carried out with students aged 13 and 14 years, enrolled in state public schools in 2014 in the municipality of Petrolina (state of Pernambuco) located in northeastern Brazil, which has a population of approximately 294,000 inhabitants,12 occupying an area corresponding to 4561.872km2. This study was approved by the Ethics Committee on Human Research of Universidade de Pernambuco (protocol: 459.304).
Data were collected through questionnaires applied at schools and information was recorded in forms consisting of pre-coded questions, containing questions on age, gender, allergic diseases, physical activity level (PAL), maternal education, family income, presence of a smoker in the household, and history of asthma in the parents. The questionnaires filled out by the adolescents were reviewed at the end of the day for identification and opportune correction of completion errors; incomplete, questionable, or inconsistent data were subsequently verified through new visits.
The ISAAC questionnaire, translated and validated for Brazil, was used for the evaluation of allergic diseases.2 This questionnaire has three modules (asthma, rhinitis, and eczema) with up to eight questions each, and is self-applicable and easy to understand. The authors considered for classification of asthma prevalence or active asthma those who answered “yes” to the question “In the last 12 months, have you had wheezing?”. For the diagnosis of asthma, the authors considered those who answered “yes” to the question “Have you ever had asthma in your life?” and for the diagnosis of severe asthma, those adolescents classified as having active asthma who reported chest wheezing that was strong to the point of affecting speech. This questionnaire does not contain questions regarding the use of medication. Those adolescents who answered “yes” to the following questions were considered as having rhinitis and atopic dermatitis, respectively: “In the last 12 months, have you had a problem with sneezing, rhinorrhea (runny nose), and nasal obstruction, when you did not have the flu or a cold?” and “In the last 12 months, have you had skin spots (eczema)?”.
To assess the impact of asthma on the adolescents’ daily activities, absenteeism and overall school performance were evaluated, as well as activity in the physical education classes. School absenteeism was evaluated by observing the total number of missed school days in each discipline through the school attendance record related to the year prior to the study. School performance was assessed using the average of the grades obtained during the year prior to the study in compulsory subjects (mathematics, Portuguese, science, social sciences, and arts education), common to all schools. The maternal level of schooling was characterized as 0–9 years, 10–13 years, and greater than 14 years of schooling, whereas family income was characterized as up to two minimum wages and higher than two minimum wages.
The level of physical activity was assessed using the International Physical Activity Questionnaire (IPAQ – short version) validated for Brazil,13 which classifies the individual as very active, active, irregularly active, or sedentary, according to the frequency, duration, and intensity of weekly physical activities. Individuals were considered active when they met the following criteria: (a) vigorous physical activity on ≥3 days/week, with a duration of at least 20min/session; (b) moderate physical activity or walking on ≥5 days/week, with duration of at least 30min/session; (c) any physical activity whose frequency was ≥5 days/week, with duration of at least 150min/week. Subjects who did not meet the abovementioned criteria were classified as inactive. Written questionnaires were distributed in classrooms and filled out by the adolescents themselves, under the supervision of the previously trained researchers.
Sample size and statistical analysisThe WinPepi program (PEPI-for-Windows)14 was used to quantify the sample size according to the following criteria: estimated population of 31,555 students; confidence interval of 95%; sampling error of five percentage points; estimated prevalence of 50%, as there were more than two variables of interest; sample design effect set at three times the minimum size of the sample; and sample loss of 20%, therefore totaling a minimum sample size of 1425 students.
The sample selection procedure followed a sequence of steps in an attempt to obtain a representative sample of students from state schools regarding the distribution according to school size. In order to assist sample planning, schools were organized into three categories: small (up to 200 students), medium-sized (201–499 students) and large (more than 500 students), and a stratification criterion was applied, considering proportionality by size.
After all steps, a total number of 18 schools and 57 classes was obtained, representing 42% of state schools in Petrolina. Considering a minimum of 25 students per classroom, 1425 students were expected to be evaluated. As some grades had more than 25 students per classroom, a total of 1591 students were evaluated. The schools were chosen by drawing lots after randomization carried out using the WinPepi program.14
Data were processed and analyzed using SPSS, version 20.0 and Stata 7.0 (StataCorp. 2015. Stata Statistical Software: Release 7. College Station, USA). Initially, data were entered into the statistical package SPSS using double entry and inconsistencies were compared. The Kolmogorov–Smirnov test was applied to test the normality assumption. The comparison between the medians was performed using the Mann–Whitney test for non-parametric variables. To assess the differences between the proportions of asthma and associated factors, the chi-squared test and chi-squared test with Yates's correction were used when appropriate. All conclusions were based on a significance level of 5%.
Prevalence ratios (PR) for asthma were calculated for each exposure variable by Poisson regression. The variables with p-value<0.20 in the bivariate analysis were selected for the multivariate regression model, aimed to assess the impact of the explanatory variables. The results were expressed as adjusted prevalence ratios (PR) with their respective 95% confidence intervals (95% CI) and p-values<0.05 were considered statistically significant.
ResultsA total of 1591 adolescents aged 13–14 years participated in the study, of whom 791 (49.7%) were male and 800 (50.3%) female; 67.2% of their families earned up to two minimum wages and only 17.8% of the mothers had college/university level of schooling.
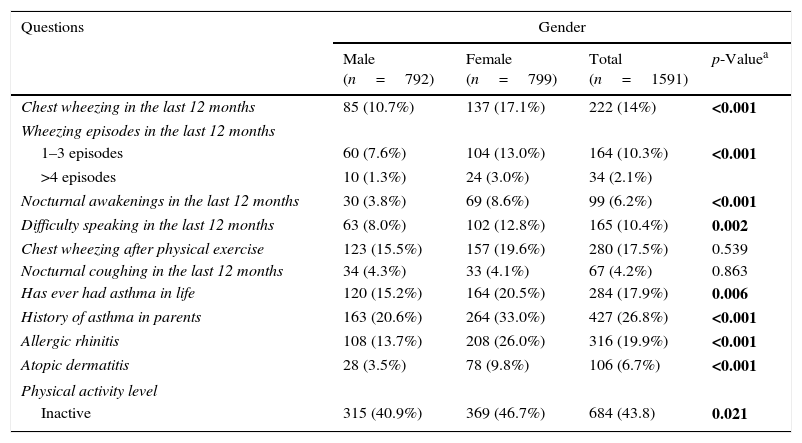
The distribution of adolescents by gender and factors related to allergic diseases shows that the prevalence of active asthma, severe asthma, and physician-diagnosed asthma were 14.0%, 10.4%, and 17.8%, respectively, and that female adolescents had a higher prevalence of asthma, allergic rhinitis, and dermatitis (Table 1).
Distribution of students by gender and factors related to allergic diseases.
| Questions | Gender | |||
|---|---|---|---|---|
| Male (n=792) | Female (n=799) | Total (n=1591) | p-Valuea | |
| Chest wheezing in the last 12 months | 85 (10.7%) | 137 (17.1%) | 222 (14%) | <0.001 |
| Wheezing episodes in the last 12 months | ||||
| 1–3 episodes | 60 (7.6%) | 104 (13.0%) | 164 (10.3%) | <0.001 |
| >4 episodes | 10 (1.3%) | 24 (3.0%) | 34 (2.1%) | |
| Nocturnal awakenings in the last 12 months | 30 (3.8%) | 69 (8.6%) | 99 (6.2%) | <0.001 |
| Difficulty speaking in the last 12 months | 63 (8.0%) | 102 (12.8%) | 165 (10.4%) | 0.002 |
| Chest wheezing after physical exercise | 123 (15.5%) | 157 (19.6%) | 280 (17.5%) | 0.539 |
| Nocturnal coughing in the last 12 months | 34 (4.3%) | 33 (4.1%) | 67 (4.2%) | 0.863 |
| Has ever had asthma in life | 120 (15.2%) | 164 (20.5%) | 284 (17.9%) | 0.006 |
| History of asthma in parents | 163 (20.6%) | 264 (33.0%) | 427 (26.8%) | <0.001 |
| Allergic rhinitis | 108 (13.7%) | 208 (26.0%) | 316 (19.9%) | <0.001 |
| Atopic dermatitis | 28 (3.5%) | 78 (9.8%) | 106 (6.7%) | <0.001 |
| Physical activity level | ||||
| Inactive | 315 (40.9%) | 369 (46.7%) | 684 (43.8) | 0.021 |
Values are expressed in absolute numbers and percentages between parentheses. Bold values indicate statistical difference.
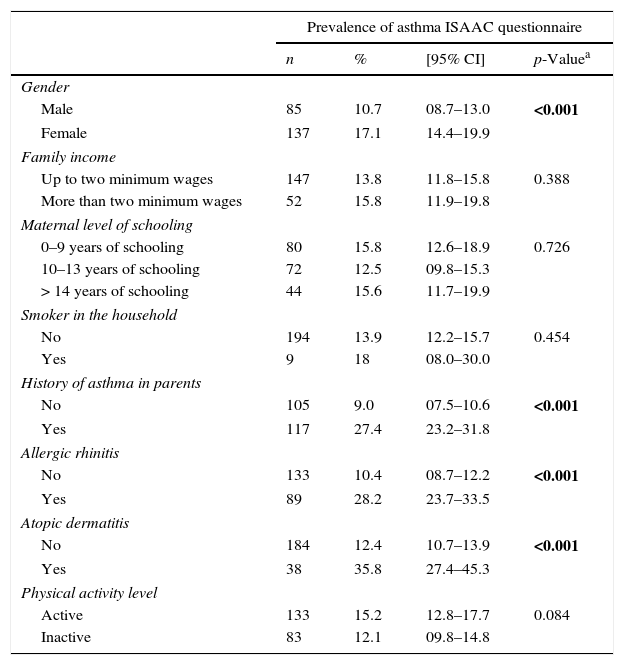
The bivariate analysis of the active asthma prevalence and possible intervening factors (Table 2) showed the importance of the association with allergic rhinitis and atopic dermatitis, as well as the history of asthma in the parents (28.2% vs. 10.4%, 35.8% vs. 12.4%, and 27.3% vs. 8.3%, respectively, all with p<0.001).
Prevalence of asthma in adolescents according to gender, family income, maternal level of education, smoking status, history of asthma, allergic rhinitis, atopic dermatitis, and level of physical activity (total number of assessed adolescents: 1591).
| Prevalence of asthma ISAAC questionnaire | ||||
|---|---|---|---|---|
| n | % | [95% CI] | p-Valuea | |
| Gender | ||||
| Male | 85 | 10.7 | 08.7–13.0 | <0.001 |
| Female | 137 | 17.1 | 14.4–19.9 | |
| Family income | ||||
| Up to two minimum wages | 147 | 13.8 | 11.8–15.8 | 0.388 |
| More than two minimum wages | 52 | 15.8 | 11.9–19.8 | |
| Maternal level of schooling | ||||
| 0–9 years of schooling | 80 | 15.8 | 12.6–18.9 | 0.726 |
| 10–13 years of schooling | 72 | 12.5 | 09.8–15.3 | |
| > 14 years of schooling | 44 | 15.6 | 11.7–19.9 | |
| Smoker in the household | ||||
| No | 194 | 13.9 | 12.2–15.7 | 0.454 |
| Yes | 9 | 18 | 08.0–30.0 | |
| History of asthma in parents | ||||
| No | 105 | 9.0 | 07.5–10.6 | <0.001 |
| Yes | 117 | 27.4 | 23.2–31.8 | |
| Allergic rhinitis | ||||
| No | 133 | 10.4 | 08.7–12.2 | <0.001 |
| Yes | 89 | 28.2 | 23.7–33.5 | |
| Atopic dermatitis | ||||
| No | 184 | 12.4 | 10.7–13.9 | <0.001 |
| Yes | 38 | 35.8 | 27.4–45.3 | |
| Physical activity level | ||||
| Active | 133 | 15.2 | 12.8–17.7 | 0.084 |
| Inactive | 83 | 12.1 | 09.8–14.8 | |
CI, confidence interval. Bold values indicate statistical difference.
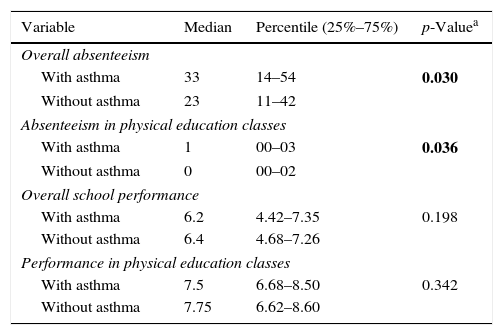
Adolescents with asthma missed more school days, including physical education classes, than their non-asthmatic peers (p<0.05). However, lower academic performance was not detected among students with asthma (Table 3).
Overall absenteeism, absenteeism in physical education classes, overall school performance, and performance in physical education classes by the students.
| Variable | Median | Percentile (25%–75%) | p-Valuea |
|---|---|---|---|
| Overall absenteeism | |||
| With asthma | 33 | 14–54 | 0.030 |
| Without asthma | 23 | 11–42 | |
| Absenteeism in physical education classes | |||
| With asthma | 1 | 00–03 | 0.036 |
| Without asthma | 0 | 00–02 | |
| Overall school performance | |||
| With asthma | 6.2 | 4.42–7.35 | 0.198 |
| Without asthma | 6.4 | 4.68–7.26 | |
| Performance in physical education classes | |||
| With asthma | 7.5 | 6.68–8.50 | 0.342 |
| Without asthma | 7.75 | 6.62–8.60 | |
Bold values indicate statistical difference.
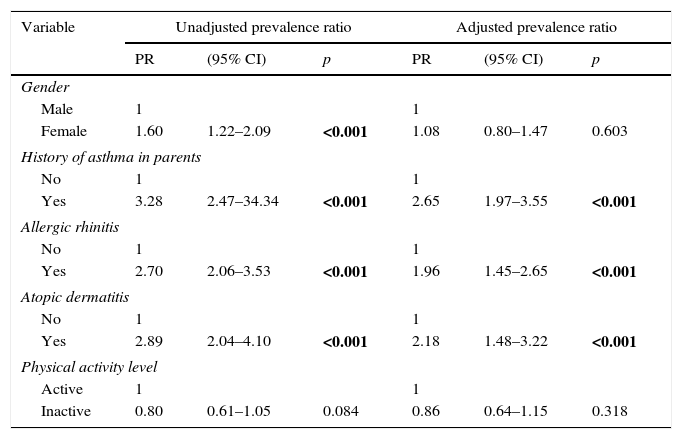
At the Poisson regression analysis, which included the female gender and a lower level of physical activity, it was observed that the only variables that remained in the final model were allergic rhinitis and atopic dermatitis, as well as a history of asthma in the parents (Table 4).
Poisson regression for asthma prevalence by gender, history of asthma in parents, allergic rhinitis, atopic dermatitis, and level of physical activity.
| Variable | Unadjusted prevalence ratio | Adjusted prevalence ratio | ||||
|---|---|---|---|---|---|---|
| PR | (95% CI) | p | PR | (95% CI) | p | |
| Gender | ||||||
| Male | 1 | 1 | ||||
| Female | 1.60 | 1.22–2.09 | <0.001 | 1.08 | 0.80–1.47 | 0.603 |
| History of asthma in parents | ||||||
| No | 1 | 1 | ||||
| Yes | 3.28 | 2.47–34.34 | <0.001 | 2.65 | 1.97–3.55 | <0.001 |
| Allergic rhinitis | ||||||
| No | 1 | 1 | ||||
| Yes | 2.70 | 2.06–3.53 | <0.001 | 1.96 | 1.45–2.65 | <0.001 |
| Atopic dermatitis | ||||||
| No | 1 | 1 | ||||
| Yes | 2.89 | 2.04–4.10 | <0.001 | 2.18 | 1.48–3.22 | <0.001 |
| Physical activity level | ||||||
| Active | 1 | 1 | ||||
| Inactive | 0.80 | 0.61–1.05 | 0.084 | 0.86 | 0.64–1.15 | 0.318 |
CI, confidence interval; PR, prevalence ratio. Bold values indicate statistical difference.
The prevalence of asthma, severe asthma, and physician-diagnosed asthma in this study in the Brazilian semiarid region were 14.0%, 10.4%, and 17.8%, respectively. The factors associated with asthma were allergic rhinitis and atopic dermatitis, as well as a history of asthma in the parents; additionally, adolescents with asthma missed more school days than their peers without asthma.
Climatic differences can have a significant influence on asthma prevalence, which is demonstrated by the observed higher prevalence in regions with higher relative humidity and rainfall and lower prevalence in regions with hot and dry climates.2–6 When comparing these findings with studies carried out in other Brazilian locations,2,11 a lower prevalence of asthma was observed, possibly related to the specific climatic characteristics of the studied region.2–6 The dry climate hinders the survival of dust mites, the main household allergens, which require hydration obtained from the humidity of the air to survive and, thus, environments with relative humidity below 55% are lethal to them.6,9
However, as in this study, asthma is still detected in these regions, and one possible explanation is the presence of other allergens, such as pollen and spores of plants,4 or contact with animal epithelia and substances found in the soil that can be found suspended in the air, which, when processed into smaller particles, can be transported by winds over long distances.8 There have been reports of allergic sensitization in areas close to pistachio plantations in the desert in Iran4 and species of fungi have been described as the agent with the highest association with asthma in semiarid climates in the desert.15–17 Allergic respiratory diseases caused by plants may also exist, which have not been yet identified, in this area in northeastern Brazil, as one of the characteristics of this research area consists of irrigated fruit crops with large plantation fields.
Another interesting fact is that although asthma is less prevalent in this locality, the asthmatic adolescents reported a higher degree of disease severity, which appears to directly affect the lives of these individuals. Rabe et al. 18 describe a negative effect of asthma with considerable loss of work and school days in the most severe cases, in addition to the fact that current control around the world is far from the objectives established in the recommendations of international guidelines.1
In fact, the higher degree of school absenteeism found in the present study (33 vs. 22 school days/year; p<0.03) seems to be related to the severity of asthma, as the majority (74%) of adolescents with active asthma reported symptoms that were consistent with severe disease.8,18,19 This fact indicates a possible lack of identification and control of the disease and, moreover, it is possible that in the hot, dry climate, patients have a more severe disease phenotype. Models showing asthma triggering after exercise with patients breathing dry air suggest that bronchospasms may be more severe under these conditions.21,22
Even with a lower prevalence of asthma than the national average (14% vs. 19%),2 the disease in this location seems to have a more severe presentation (10.4%) when compared to the rest of Brazil (4.7%)2 and the world (6.9%).11 Studies in desert regions of Saudi Arabia5,23 have shown a greater number of visits to the emergency room in individuals with asthma when compared to individuals living in European cities (62% vs. 24%),24 indicating an association with greater disease severity in these areas, which may be related to the availability of care, as well as to the climatic characteristics of the region.
Data from the Informatics Department of the Brazilian Unified Health System (Departamento de Informática do Sistema Único de Saúde [DATASUS]) show that the number of hospital admissions for asthma during the seven-year period in Petrolina is higher than in municipalities located on the coast and in the hinterlands of the state of Pernambuco with a similar population size. The standardized hospitalization rate per 100,000 inhabitants was 509.4 in Petrolina (semiarid region), 259.58 in Caruaru (hinterlands), and 180.53 in Paulista (coastal region).12 This is an alarming scenario for asthma in this region, which seems to be effectively more severe and predominantly allergic, as it is strongly associated with other characteristics of atopy, such as allergic rhinitis and atopic dermatitis.
In this study, after adjustment of variables, the association of asthma with allergic rhinitis (prevalence ratio=1.96) was lower than that found in other Brazilian studies (reported values between 2.19 and 4.36)25; nevertheless, together with atopic dermatitis, it was still an important factor for the diagnosis of asthma in this population. These findings suggest that even in a dry and hot climate, atopy appears to be present as a major factor. An additional fact, besides this association with other allergic diseases, is the report of a history of asthma in the parents as a consistent factor associated with the prevalence of disease, which contributed to a prevalence ratio of 2.65. This may suggest the need to alert adults with asthma to identify signs of this disease in their children and contribute, together with the health care staff, to early and adequate care.19,20,26
It is important to consider some limitations of this study. The use of questionnaires can result in some recall and/or interpretation bias, and there are temporal gaps that exist between multicentric ISAAC trials in Brazil.27,28 Studies carried out almost ten years ago in the same state, using similar methodology, but in places with very different climatic conditions, showed differences in prevalence between the more humid region of the coast (19.1%), the intermediate region of the Brazilian hinterlands (17.9%), and the current study, carried out in the semiarid region (14%).2
Additionally, variables not included in the study – such as eating habits, atopic sensitization, and psychological factors – can influence the occurrence of asthma.26,29,30 The fact that the ISAAC questionnaire does not contain questions about treatment does not allow for making inferences about a possible association between the prevalence of asthma or even observed severe asthma and the use of medications, even though they have been prescribed, might not have been objectively used. However, the results described herein can help to understand the prevalence of asthma and its severity in hot, dry climates.
In conclusion, in the municipality of Petrolina, located in the semiarid region of Northeast Brazil, the prevalence of asthma was lower than those reported in other Brazilian cities in studies that used similar methodology, despite calling attention to the high proportion of reports of more severe disease symptoms.2,27,28 Considering the responses to the ISAAC questionnaire, it can be inferred that asthma, at least for the physicians, is not a neglected disease, as the frequency of physician-diagnosed asthma is higher (17.9%) to that of wheezing in the last year (14%); however, this is no guarantee that patients will be treated appropriately. The low air humidity and lack of clinical control are possible explanations for this finding. These data should be used to guide health policies under similar conditions, and further studies should be conducted to assess whether the improvement in medical care will reduce the number of hospitalizations and visits to emergency rooms due to asthma that were found in this study.
Conflicts of interestThe authors declare no conflicts of interest.
The authors would like to thank Professor Dirceu Solé for his invaluable contributions in reviewing the text.
Please cite this article as: Correia Junior MA, Sarinho ES, Rizzo JA, Sarinho SW. Lower prevalence and greater severity of asthma in hot and dry climate. J Pediatr (Rio J). 2017;93:148–55.