Perfume (Parfum) or fragrance is a natural or synthetic cosmetic ingredient added to emit a pleasant aroma or to improve the odor of a cosmetic formula. It is a mixture of substances, not revealed by the manufacturer, which may contain ingredients with allergenic potential, endocrine disruptors, and other possible harmful effects on human health. This study aims to analyze children's cosmetics labels to assess the presence of Perfume.
MethodsThe researchers randomly visited points of sale in Curitiba, the capital of a southern Brazilian state; in order to catalog the largest possible number of children's cosmetics items.
Results398 children's cosmetics were analyzed and found Parfum on 295 (74.1 %) of the labels, including 90.4 and 79,1 % of the shampoos and wet wipes, respectively.
ConclusionExposure of children's skin to fragrances can lead to local side effects such as allergies, but also to systemic effects, and the lack of knowledge of the general population and health professionals about its possible deleterious effects emphasizes the importance of changes in the regulation of cosmetics aiming to reduce the use of this ingredient.
Exposure to chemicals begins prenatally and continues after birth, primarily through personal care products. This exposure can negatively impact children's health due to the immaturity of organs and systems. Children's skin has a thinner stratum corneum and therefore is more susceptible to the penetration of irritants and allergens. Also, children have a higher body surface-to-weight ratio which leads to greater absorption of substances applied to the skin.1
In most countries, regulations on children's cosmetics differ from that of adults. But even so, they are based on current but still limited scientific knowledge about the effect of cosmetic ingredients. After years of using a certain component, it is possible that new evidence can refute its safety and it would be prohibited.
A fragrance is a mixture of substances added to provide a pleasant aroma or mask an unpleasant odor to a formula, and identified in a cosmetic ingredient list simply as “Parfum”.2,3 Its origin can be natural, derived from organic materials, such as essential oils or natural extracts; or synthesized from a mixture of chemical products.1,4 Generally, the exact components of the mixture are not disclosed by the manufacturer, since it's not mandatory by the legislation – it's a “trade secret” supported by marketing strategies, which makes information about the safety of the compounds limited.3 Cosmetic label regulation varies worldwide, and in many countries, including Brazil, there are specific lists of ingredients that must be disclosed separately in the label (e.g. Amyl cinnamal, Coumarin, Eugenol, Geraniol, Benzyl benzoate, Citronellol, Farnesol, d-Limonene, Linalool), whilst others are disclosed only as “Parfum”.3
Since fragrances are frequently used in daily personal care products worldwide, the present work describes its presence on the labels of over-the-counter children's products in the southern Brazilian capital and aims to inform the population and health professionals about its high frequency and potential risks.
MethodsFrom February to December 2021, the researchers randomly visited various points of sale (pharmacies, cosmetics stores and markets) in a range of 15 km from downtown Curitiba, the capital of a southern Brazilian state with a population of 1.7 million, in order to catalog the largest possible number of children's cosmetics items. All cosmetics found on those stores, in each category, were included, as long as infant use was expressly indicated in the label, regardless of price or other characteristics.
The following categories of children's cosmetics were included: liquid soaps, bar soaps, shampoos, conditioners, moisturizers, diaper rash prevention creams, wet wipes, sunscreens and repellents. All evaluated products were indicated for use by children (packages stated “children”, “kids” or “baby”).
The labels were photographed and the lists of ingredients in the INCI nomenclature (International Nomenclature of Cosmetic Ingredients) were extracted to a Microsoft Excel® software spreadsheet. The fragrances were identified in the ingredient list with the term “Parfum/Fragrance”.
As this is not a study with humans, there was no need to submit the study to the ethics committee.
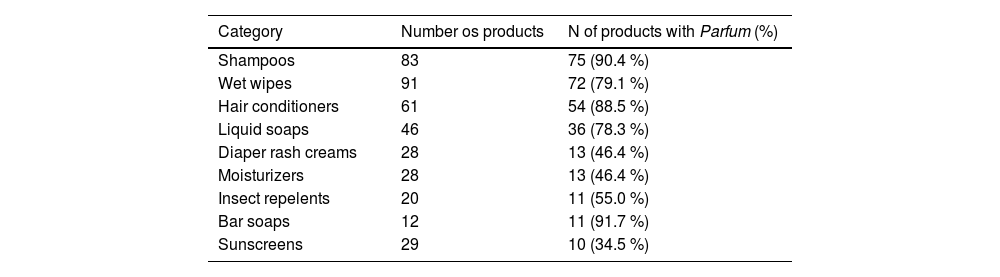
ResultsThree hundred and ninety-eight products were evaluated and 295 (74.1 %) had Parfum/Fragrance on their labels. The total number of products included in each category and the number and percentage of those that had Parfum/Fragrance in their composition are described in Table 1.
Total number of children's cosmetics included in each category and percentage of which had Parfum in their ingredient list.
Shampoos, wet wipes, and conditioners were the products that most had parfum in their composition and are widely used in the pediatric age group.
In 2008, White and McFadden analyzed 38 cosmetic products marketed for babies and found that 84 % (32) of them had fragrance chemicals,5 more than the present results - 74.1 % (295). This may be explained because the authors only included “parfum”, but not other ingredients separately disclosed.
Phthalates, synthetic musks, and “sensitizers” are the most common groups of synthesized fragrance mixtures. In cosmetics, phthalates are used to prevent rapid evaporation of the scent, but they are also ingredients for the plastic industry, adhesives and detergents. Synthetic musks are mainly used for fragrance and fixation in cosmetics and household products, and sensitizers are chemicals with a high probability of causing allergic reactions in humans.2
Besides allergy, other effects have been related to fragrance compounds in various studies, such as endocrine disruption, genital malformations, infertility, precocious puberty, and neurotoxicity and carcinogenicity.2,6,7
Animal data point to a high sensitivity of the development of the hypothalamic-pituitary-gonadal axis to endocrine-disrupting chemicals, which have already been detected in the placenta and amniotic fluid and are associated with impaired fetal development.6 There has been a decrease in the age of onset of puberty in girls and boys over the last 30 years and the exposure to endocrine-disrupting chemicals, in the environment and by cosmetics, may be related.6 Exposure to fragrance compounds may also be related to fetal growth restriction, as shown the study by Zhao et al., 2016, with 220 mother-newborn pairs, including 110 with fetal growth restriction newborns and 110 normal newborns. Maternal urine samples were collected during the third trimester and tested for 5 phthalate ester metabolites. The authors found alterations in placental IGF2 DNA methylation, that could be part of a pathway to adverse fetal growth.8
Besides phthalates, synthetic polycyclic musks may display estrogenic activities in vitro and in vivo effects depending on specific receptors.9 Also, low concentrations of some synthetic musks were found to have anti-progestogenic effects.10
Prenatal exposure to phthalates, measured by phthalate metabolite concentrations in maternal urine, was associated with behavior disorders, like problem-externalization, emotional control, aggression, and depression in children aged 4–9, through a specific questionnaire designed to assess executive cognitive function in children.11 A synthesis of many other studies that point to neurological consequences of fragrance compounds can be found at Pinkas et al., 2017, such as effects in different developmental stages on hippocampal structure, neuronal apoptosis in the hippocampus, and others.2
Ahern et al., 2022, in a Danish study with 1278.685 children, showed that exposure to phthalates from medically prescribed drugs, prenatally or after birth, was positively associated with the incidence of osteosarcoma and lymphoma before age 19 years.12 More studies are needed to clarify which childhood cancer cases could be avoided by reducing or eliminating the phthalate content of medications and other consumer products.12
Lee et al., 2016, in a review of the relationship between cancer progression and the effects of endocrine-disrupting chemicals, found in vitro studies that indicated that several phthalates possess plausible anti-androgenic effects, which correlates with increased breast cancer risk and involvement in cancer metastasis (increased cell proliferation, tumor mobility and invasiveness of tumor cells).13
Natural fragrance compounds, botanical extracts, and essential oils are not risk-free. Phytochemicals such as linalool are potentially allergenic,14,15 and are present, e.g., in lavender essential oil (Lavandula angustifolia), used worldwide. Lavender and tea tree essential oils have been linked to gynecomastia in prepubertal males and premature thelarche in girls.16
White and McFadden, 2008, pointed to the possible relationship of haptens – found in many cosmetic ingredients, including fragrance compounds, with the development of atopic diseases. Haptens are small molecules that can, when combined to atopy-related proteins, bind to receptors and stimulate altered (Th2) immune response.5 Paradoxically, haptens are also present in rinse-off cleansing products and emollients for atopic dermatitis, as shown by Kunkiel et al., 2023.17
Exposure to fragrances can also lead to asthma, contact dermatitis (irritant or allergic), dyschromia, and photosensitivity.1,4,18 Irritant-type dermatitis occurs at the site of product application, such as in the armpits when using perfumed deodorants, or due to fragrances in other personal care products. Allergic-type dermatitis, on the other hand, has urticaria as a common presentation and frequently occurs after exposure to Balsam of Peru (Myroxylon pereirae).4,18 The North American Contact Dermatitis Group estimates that allergic contact dermatitis occurs in 5.3 % to 11.3 % of the general population. In the analysis of the main allergens in patch tests in 2015/2016, three screening tests for fragrances were found relevant: Fragrance-Mix-1, Fragrance-Mix-2, and Myroxylon pereirae.19 Sensitive areas like the perineal region are more susceptible to allergic contact dermatitis by fragrances from wet wipes.20 In the present study, 79.1 % of the 91 wet wipes contained fragrance. Yu et al. in a North American study found the ingredient in 33.3 % of the 63 wet wipes analyzed. This difference can be explained because some products do not include the term “fragrance” on the ingredients, according to each country's legislation, but the individual components of the fragrance, such as linalool or geraniol, make identification difficult.20
In fact, a limitation of this study is that the authors only searched for the words "parfum" and "fragrance" in the cosmetics’ ingredients lists, but individual fragrance components were not included. Therefore, the number of products containing fragrances is also likely to be higher.
Due to the potential health risks posed by fragranced products, in the United States and Europe, initiatives to monitor and regulate fragrances in children's products are growing. Similar initiatives should be adopted worldwide.1 The lack of knowledge of the general population and health professionals, added to the remarkable presence of perfume in the daily routine of families, emphasizes the importance of further studies in this area. Health professionals and Pediatric Societies must demand improvement in regulation processes and ongoing monitoring to ensure the safety of children's skin products on the market.