To evaluate lung functional capacity (FC) for physical exercise in children and adolescents with sickle cell disease (SCD) through the six-minute walk test (6MWT).
MethodA cross-sectional prospective study was performed to evaluate the FC of 46 patients with SCD through the 6MWT. The following parameters were assessed: heart rate (HR), respiratory rate (RR), peripheral pulse oxygen saturation (SpO2), peak expiratory flow (PEF), blood pressure (systolic and diastolic), dyspnea, and leg fatigue (modified Borg scale) at rest, in the end of the test, and ten minutes after the 6MWT. The total distance walked was also recorded. For statistical analysis, the parametric variables were analyzed using the paired Student's t-test, analysis of variance (ANOVA), and Bonferroni multiple comparisons, with a significance level set at p ≤ 0.05.
ResultsThe 46 patients were aged age 9.15±3.06 years, presented baseline Hb of 9.49±1.67g/dL, and walked 480.89±68.70 m. SCD diagnosis was as follows: group 1- HbSS (n=20)/HbSβ0-thalassemia (n=3) and group 2 - HbSC (n=20)/HbSβ+-thalassemia (n=3). Regarding total distance walked, patients in group 1 walked a shorter distance than patients in group 2 (459.39±57.19 vs. 502.39±73.60 m; p=0.032). There was no statistical difference regarding PEF in the three moments of evaluation. The SpO2 in ambient air and SpO2 with O2 differed between groups 1 and 2 (p<0.001 vs. p=0.002), as well as the RR (p=0.001).
ConclusionThese patients showed a lower FC for exercise than that predicted for the age range in the literature. Patients diagnosed with HbSS/Sβ0-thalassemia had a lower performance in the test than those with HbSC/Sβ+-thalassemia regarding total distance walked, RR, and SpO2 after the 6MWT.
Avaliar a capacidade funcional pulmonar (CF) para o exercício físico de crianças e adolescentes com doença falciforme (DF) pelo teste da caminhada de seis minutos (TC6′).
MétodosEstudo transversal prospectivo avaliando a CF pelo TC6′ de 46 pacientes com DF. Foram avaliados: frequência cardíaca (FC), frequência respiratória (FR), saturação de pulso de oxigênio (SpO2), pico de fluxo expiratório (PFE), pressão arterial (PA) sistólica e diastólica, dispneia e cansaço em membros inferiores (escala de Borg modificada) em repouso, ao término e 10 minutos após o TC6′ e a distância percorrida. Análise estatística: test t-Student pareado, análise de variância e comparações múltiplas de Bonferroni, significância p ≤ 0,05.
ResultadosDos 46 pacientes, a média±dp da idade foi 9,15±3,06 anos, hemoglobina basal 9,4±1,67g/dL e distância percorrida 480,89±68,70 m. Diagnóstico da DF: Grupo 1– HbSS (n=20)/HbSβ0-talassemia (n=3); e Grupo 2 – HbSC (n=20)/ HbSβ+-talassemia (n=3). O Grupo 1 apresentou menor distância percorrida do que o Grupo 2 (459,39±57,19 vs 502,39±73,60m; p=0,032). Não houve diferença estatística em relação ao PFE. A SpO2 em ar ambiente e a SpO2 com O2 (1 L/min) após o teste foi maior no Grupo 2 (p<0,001 e p=0,002, respectivamente). A FR foi maior no Grupo 1 ao final do TC6′ (p<0,001).
ConclusãoEsta amostra apresentou CF para o exercício abaixo do predito para a faixa etária na literatura. Os pacientes com HbSS/Sβ0-talassemia apresentaram desempenho inferior na distância percorrida, FR e SpO2 após o TC6′, comparativamente aos pacientes com HbSC/Sβ+-talassemia.
Sickle cell anemia (HbSS) is the most common monogenic hereditary disease in Brazil, with an estimated prevalence of heterozygotes for HbS ranging from 2% to 8% in the general population.1 HbSS, the most severe form of sickle cell disease (SCD) is a hemoglobinopathy resulting from the single amino acid substitution of a glutamic acid for a valine at the sixth position of the beta globin chain, on chromosome 11, giving rise to hemoglobin S (HbS).2 This alteration in hemoglobin is responsible for the anomalous form of erythrocytes, leading to hemolytic anemia, endothelial vasculopathy, and vaso-occlusive phenomena, followed by tissue ischemia and necrosis, with subsequent organ dysfunction, which are responsible for the high mortality of SCD.1,2 SCD occurs when HbS combines with another hemoglobinopathy, such as C, D, β-thalassemia, or another HbS.3
The lung is a major target organ of acute and chronic complications in SCD; acute chest syndrome (ACS) is the second most frequent cause of hospitalization in this population, with high rates of morbidity and mortality.4–6 It is an acute complication usually triggered by a clinical picture of infection. It can be defined by a combination of signs and symptoms, which include dyspnea, chest pain, fever, cough, and a new pulmonary infiltrate.7 The proliferative vasculopathy that occurs in sickle cell disease is the main cause of the chronic pulmonary alterations that occur in these patients.8
The chronic alterations and recurrent episodes of ACS decrease the functional capacity (FC) in patients with SCD. MacLean et al.,9 when assessing lung function in children with SCD through spirometry, observed a restrictive pulmonary pattern and a progressive reduction in lung volume. Another prospective study,10 with patients aged 10 to 26 years, found alterations in pulmonary function, with a predominance of mixed or combined pattern.
Thus, the evaluation of FC should be part of outpatient monitoring of these patients. However, studies assessing and addressing the FC in children and adolescents with SCD are limited.11 A simple and effective method to evaluate the FC is to apply the six-minute walk Ttest (6MWT), which provides information about functional status, oxygen consumption, exercise tolerance, and patient survival according to test performance.12,13 The 6MWT assesses the individual's submaximal effort, similar to the effort made in some daily life activities, representing their FC to exercise.12
Although the 6MWT has not been widely studied in patients with hemoglobinopathies, especially in pediatric patients, preliminary data suggest that there may be a good correlation among this test, maximum oxygen uptake, and severity of pulmonary hypertension in adults with SCD.14 There are no published articles in the literature evaluating the FC in children and adolescents with SCD; thus this study aimed to evaluate the FC to exercise in children and adolescents with SCD using the 6MWT.
MethodsThis was a prospective cross-sectional study evaluating 46 children and adolescents with SCD aged between 6 and 18 years, distributed according to HbSS genotype, HbSβ0-thalassemia, HbSC, and HbSβ+-thalassemia. The children who were attended to at the pediatric hematology outpatient clinic from April of 2009 to July of 2010 and met the inclusion criteria were evaluated.
The FC of these patients was evaluated through the 6MWT, following the guidelines of the American Thoracic Society.12 The study was approved by the Ethics Committee in Research of the Escola Paulista de Medicina/Universidade Federal de São Paulo (EPM/UNIFESP). Exclusion criteria were patients with hemodynamic instability or neurological or orthopedic diseases that limited exercise performance.
Basal hemoglobin (Hb) and history of hospitalizations for pulmonary complications were collected from the patients’ medical records. The assessment of body mass (in kg) and height (in m) was performed in a standardized manner, using an anthropometric digital scale (Filizola®). The classification of nutritional status was performed using Z-score distribution for weight/age, height/age and body mass index (BMI). The WHO AnthroPlus 2007 software of the World Health Organization (WHO) was used to obtain the BMI and Z score values.15
The 6MWT was performed in a 10-meter long corridor, outdoors, with markings at every meter and indicators on the ground indicating the beginning and end of the trajectory. It was decided to perform the test in a 10-meter corridor due to the promising results in the “shuttle walk test”, which also uses this distance.16–20
Patients received theoretical and practical training regarding the objectives of the 6MWT and how they should perform it. Each patient underwent two tests (with a 30-minute interval between them); the test with best performance was considered if the difference between tests was less than 10% In case of a difference higher than 10%, a new test would be performed. No patient had to perform more than two tests.
The 6MWT was administered by two trained physical therapists. At the beginning of the test, the patient was at rest for 15 to 20minutes and had fasted for at least two hours. At the end of six minutes, the patient stopped where he/she was, and one of the therapists approached him/her with a chair and assessment equipment in order to record the total distance walked. The number of turns was counted manually, and a stopwatch (Casio®) was used to mark the time.
Water was offered to the patients before and after the 6MWT. All patients used oxygen during the test (nasal cannula 1 L/min), regardless of SpO2 on ambient air, in order to prevent hypoxemia during exercise, due to the risk of a vaso-occlusive crisis caused by SCD.
All variables were assessed at rest, at the end of the test, and at 10minutes after it. The following parameters were measured: systolic (SBP) and diastolic (DBP) blood pressure with a BD® sphygmomanometer; respiratory rate (RR), SpO2, and heart rate (HR) with a portable pulse oximeter (Dixtal®). The peak expiratory flow (PEF) was measured using an Assess Peak Flow Meter® device; three measurements were taken and the highest was considered.21 Dyspnea and lower-limb fatigue were assessed by the modified Borg scale.21 All variables were measured with the patient in the sitting position, except for PEF, which was measured in the standing position.
To analyze the behavior of SCD during the 6MWT, the sample was divided into two groups according to the disease genotype. Group 1 consisted of patients with HbSS/HbSβ0-thalassemia and group 2 consisted of patients with HbSC/ HbSβ+-thalassemia, as patients in group 1 tend to have a more severe clinical course than those in group 2.22,23
Statistical analysisParametric variables were analyzed by paired Student's t-test and continuous measurements by ANOVA for repeated measures. Multiple Bonferroni comparisons were made between the two evaluation moments, and values were considered statistically significant when p ≤ 0.05. Results are shown as mean±standard deviation (SD), median (minimum-maximum), frequency, and percentage. The statistical analysis was performed using the Statistical Package for Social Sciences (SPSS) release 15.0.
ResultsThis study evaluated 46 patients with SCD with the following demographic characteristics, expressed as mean±standard deviation (SD): age 9.15±3.06 years; basal Hb 9.49±1.67g/dL; weight 30.98±11.30kg; height 1.34±0.16 m; BMI 16.63±2.40kg/m2; percentile weight/age 49.29±26.57; height/age 42.83±25.92; and BMI/age 46.86±24.70. The Z-scores for all anthropometric indices were: weight/age 0.04±1.03; height/age 0.22±0.95; and BMI/age 0.20±1.26. The total distance walked during the 6MWT was 480.89±68.70 m.
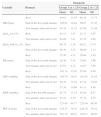
Patients in group 1 and group 2 were similar regarding age, gender, and anthropometric variables; group 1 had lower mean Hb values and higher frequency of pneumonia and/or ACS than group 2 (Table 1). The distance walked by patients in group 1 was significantly shorter than that of patients in group 2 (459.39±57.19 vs. 502.39±73.60 m, p=0.032 - Table 1).
Demographic and clinical data and distance walked in the six-minute walk test by the HbSS/Sβ0-thalassemia and HbSC/Sβ+-thalassemia groups.
| HbSS / Sβ0-thalassemia group 1 | HbSC / Sβ+-thalassemia group 2 | p | |
|---|---|---|---|
| Demographic data | |||
| Age (years) | 9.21±3.45 | 9.08±2.69 | 0.887 |
| Gender (%) | 56.53 (male) | 52.17 (male) | 0.767a |
| Weight (Kg) | 30.85±13.31 | 31.11±9.16 | 0.939 |
| Height (m) | 1.34±0.18 | 1.34±0.14 | 0.956 |
| Body mass index | 16.37±2.67 | 16.89±2.13 | 0.472 |
| Hb (mean g/dL) | 8.26±1.33 | 10.70±0.90 | <0.001 |
| Hospitalizations due to pulmonary complications | 0.001b | ||
| PNM % (n) | 47.82 (11) | 34.78 (8) | |
| ACS % (n) | 26.08 (6) | 0.0 | |
| ACS and PNM % (n) | 13.05 (3) | 8.70 (2) | |
| No complications % (n) | 13.05 (3) | 56.52 (13) | |
| Distance walked (m) | 459.30±57.19 | 502.39±73.6 | 0.032 |
Tests, Student's t-test; ACS, acute chest syndrome; n, number; m, meters; PNM, pneumonias.
Analyzing the 46 patients, it was observed that the cardiorespiratory variables (HR and RR) differed only at the end of the 6MWT compared to the other moments in time (p<0.001). SBP and DBP decreased significantly ten minutes after the end of the 6MWT, when compared to measurements at the end of the test (p=0.003, p<0.001) (Table 2).
Respiratory and cardiorespiratory variables of children and adolescents with sickle cell disease submitted to the six-minute walk test.
| Variables | Rest | End of test (sixth minute) | Ten minutes after end of the test | p |
|---|---|---|---|---|
| HR (bpm) | 91.93±13.0 | 102.76±20.3a | 91.17±12.1 | <0.001 |
| SpO2 c/ O2 (%) | 97.46±1.9 | 97.20±1.9 | - | 0.183 |
| SpO2 AA (%) | 95.15±3.3 | - | 95.72±3.0 | 0.053 |
| RR (rpm) | 21.67±4.4 | 26.17±5.0a | 21.26±4.2 | <0.001 |
| SBP (mmHg) | 99.37±11.6 | 103.22±15.4 | 97.96±11.6a | 0.003 |
| DBP (mmHg) | 59.26±10.0 | 62.35±10.2 | 56.46±7.9a | <0.001 |
bpm, beats per minute; DBP, diastolic blood pressure; HR, heart rate; mmHg, millimeters of mercury; rpm, respirations per minute; RR, respiratory rate; SBP, systolic blood pressure; SpO2 AA, pulse oxygen saturation with ambient air; SpO2 w/ O2, pulse oxygen saturation with oxygen (1 L/min).
aTest: analysis of variance with repeated measures, with two factors.
There was no statistical difference regarding PEF (L/min) between the three moments in time, as well as in the evaluation of lower-limb stress and fatigue assessed by the Borg scale (Table 3).
Modified Borg scale and peak expiratory flow of children and adolescents with sickle cell disease submitted to the six-minute walk test.
| Variables | Rest | End of the test (Sixth minute) | Ten minutes after end of the test | p |
|---|---|---|---|---|
| Borg Da | 0 (0-3) | 0 (0-9) | 0 (0-4) | - |
| Borg LLLLa | 0 (0-5) | 1 (0-10) | 0 (0-9) | - |
| PEF (L/min) | 223.04±65.92 | 231.63±76.65 | 226.63±67.06 | 0.074 |
Borg D, Borg of dyspnea; Borg LLLL, Borg of lower limbs; L/min, liters per minute; PEF, peak expiratory flow.
Test: analysis of variance with repeated measures, with two factors.
Table 4 presents the description of the variables measured during the 6MWT (at rest, end of the test and ten minutes after the 6MWT) in groups 1 and 2. When performing multiple comparisons of mean differences between the moments when cardiorespiratory and respiratory variables were measured in Groups 1 and 2, it was observed (means of measurements/standard error; p) at rest vs. end of the 6MWT that there was an increase in HR (-10.83/2.50; p < 0.001), RR (-4.50/0.56; p < 0.001), SBP (-3.85 / 1.50, p=0.052) and DBP (-3.09/1.48, p=0.119); restvs. ten minutes after the end of the 6MWT, a decrease in HR (0.76/2.93, p=1), RR (0.41/0.70, p=1), SBP (1.41/2.03, p=1) and DBP (2.80/1.76, p=0.343); end of the 6MWTvs. ten minutes after the end of the 6MWT, a decrease in HR (11.59/2.50; p < 0.001), RR (4.91/0.56, p < 0.001), SBP (5.26/1.59, p=0.004) and DBP (5.89/1.48, p < 0.001). When comparing restvs. ten minutes after the end of the 6MWT, there was an increase in SpO2 AA (-3.65/0.53; p < 0.001). When comparing Groups 1 and 2 at restvs. six minutes after the end of the 6MWT there was an increase in SpO2 with O2 (-1.57/0.47, p=0.002).
Description of variables at the moments (at rest, end of the test, and ten minutes after the test) in the HbSS/HbSβ0 -thalassemia and HbSC/HbS β+ -thalassemia groups.
| Diagnosis | |||||
|---|---|---|---|---|---|
| Variable | Moment | Group 1 (n=23) | Group 2 (n=23) | ||
| Mean | SD | Mean | SD | ||
| Rest | 94.61 | 11.93 | 89.26 | 13.73 | |
| HR (bpm) | End of the test (sixth minute) | 105.65 | 19.44 | 99.87 | 21.25 |
| Ten minutes after end of test | 94.39 | 12.13 | 87.96 | 11.48 | |
| SpO2 AA (%) | Rest | 93.13 | 3.55 | 97.17 | 1.07 |
| Ten minutes after end of test | 94.09 | 3.41 | 97.35 | 0.88 | |
| SpO2 with O2 (%) | Rest | 96.70 | 2.30 | 98.22 | 0.74 |
| End of the test (sixth minute) | 96.39 | 2.25 | 98.00 | 1.13 | |
| Rest | 23.30 | 4.33 | 20.04 | 3.84 | |
| RR (rpm) | End of the test (sixth minute) | 28.30 | 5.34 | 24.04 | 3.66 |
| Ten minutes after end of test | 22.65 | 4.32 | 19.87 | 3.66 | |
| Rest | 101.74 | 13.02 | 97.00 | 9.80 | |
| SBP (mmHg) | End of the test (sixth minute) | 104.78 | 18.55 | 101.65 | 11.65 |
| Ten minutes after end of test | 98.70 | 12.54 | 97.22 | 10.85 | |
| Rest | 57.39 | 10.96 | 61.13 | 8.86 | |
| DBP (mmHg) | End of the test (6th minute) | 61.74 | 11.14 | 62.96 | 9.27 |
| Ten minutes after end of test | 55.22 | 9.59 | 57.70 | 5.75 | |
| Rest | 223.04 | 64.77 | 223.04 | 68.50 | |
| PEF (L/min) | End of the test (sixth minute) | 234.57 | 78.78 | 228.70 | 76.12 |
| Ten minutes after end of test | 223.70 | 66.83 | 229.57 | 68.65 | |
bpm, beats per minute; DBP, diastolic blood pressure; HR, heart rate; L/min, liters/minute; mmHg, millimeters of mercury; RR, respiratory rate; rpm, respirations per minute; SBP, systolic blood pressure; PEF, peak expiratory flow; SpO2 AA, pulse oxygen saturation with ambient air; SpO2 w/ O2, pulse oxygen saturation with oxygen (1 L/min).
This study demonstrated that 65.21% of patients had a history of hospitalizations for pulmonary complications (ACS and/or pneumonia). People with a history of recurrent ACS have lower peak maximum oxygen consumption (VO2) and greater possibility of chronic lung injury, which may have an important impact on FC for exercise.11 Pulmonary complications were similar to those reported in literature: 86.95% of patients in group 1 had a history of hospitalization for pulmonary complications (pneumonia and/or ACS), whereas in group 2 the incidence was 43.48%. Clinical manifestations of SCD, in general, are more severe in patients with HbSS and HbSβ0-thalassemia, and ACS is more frequent in this group.6,23
The total distance walked in the 6MWT by healthy children is influenced by age, height, and weight: it is inversely proportional to weight and directly proportional to age and height.24 Although the nutritional status of the patients were within the mean normal values, differing from other studies in SCD where patients tend to be malnourished,25,26 the distance walked during the 6MWT was shorter than that predicted for age and height,27–29 meaning that factors other than weight and height, such as hemoglobin value and the occurrence of ACS, may be related to these findings.
When comparing the total distance walked, patients in group 2 walked a significantly longer distance than those in group 1. These results may be related to the fact that patients with HbSS and Sβ0-thalassemia have a clinically more severe form of the disease, with lower basal Hb values and greater number of hospitalizations for pulmonary complications, which may have also caused a significant difference in the comparison of the two groups regarding SpO2 and RR during the 6MWT.
Liem et al.11 evaluated FC in 30 children and adolescents with SCD undergoing exercise testing, and found FC limitation ranging from moderate to severe. FC for exercise was significantly lower in children with a history of recurrent ACS. and was related to the level of basal Hb (9.5±1.6g/dL), similar to values found in this study (9.49±1.67g/dL - Table 1).
Most children and adolescents adapt to the increased cardiac demand caused by chronic anemia, and generally have a 50% to 75% reduced exercise capacity.30 Chronic anemia can lead to lower tissue oxygenation, mainly during exercise, in which the use of oxygen is increased to meet the demand for energy. In SCD, some compensatory mechanisms prevent this from occurring, such as increased heart rate during exercise, increased systolic volume, and decreased peripheral vascular resistance, reducing the resistance of sickle erythrocytes in capillary transit and oxygen supply to the tissues, thus allowing close to normal saturation in the mixed venous blood.31,32 This could explain the present results of a significant increase in HR at the end of the 6MWT without significant changes in SpO2.
The study by Campbell et al.32 concluded that the exercise-induced SpO2 decrease was more related to the degree of anemia and hemolysis than to previous history of ACS and severe pain. Lammers et al.33 demonstrated that SpO2 varies little during the 6MWT in healthy children, whereas there is an increase in HR and RR. In the present study, a significant increase in HR, RR and SpO2 with O2 was observed at the end of the 6MWT.
SBP in patients with SCD is described as normal and DBP as decreased, resulting in a lower mean arterial pressure.34 The response observed in this study, a decrease in SBP and DBP ten minutes after the 6MWT, may be related to the decrease in HR and consequent decrease in cardiac output, maintaining vasodilation for a period of time after the exercise, which was also described in other studies.35,36
A subjective method of dyspnea and fatigue assessment is through the Borg scale.21 The Borg scale can be used as an aid to the 6MWT, allowing for the evaluation of the degree of respiratory distress according to the patient's perception through subjective indices.12 In this study, there were no significant changes in respiratory distress of patients during the 6MWT, which may have been due to the oxygen supplied to patients (nasal cannula 1L/min) during the 6MWT.
When evaluating PEF, there was no statistical difference among the three moments in time (Table 3). Lower PEF in asthmatic children is related to disease symptoms and the risk of exacerbation.37 In the present sample of children with SCD, there was no difference between groups 1 and 2 when PEF was evaluated at rest.
Regarding the variables analyzed during the test, it was observed that SpO2 in ambient air (AA SpO2) and SpO2 with 1 L/min of O2 were on average significantly higher in group 1 than in group 2. Significant differences were also found for RR, and higher values were observed in group 1 regardless of the time of evaluation. These findings may be related to the fact that pulmonary complications were more frequent in group 1.
This is a novel study; however, it has the limitation of using a 10-meter corridor to perform the 6MWT, whereas the ATS recommends a 30-meter corridor.12 Despite the recommendation of the ATS, some studies38–40 applied different lengths for the 6MWT. Sciurba et al.40 stated that the length of the track is not the most important feature of the 6MWT. The standardization of the shuttle walk test recommends the use of a 10-meter corridor,19 and there is a reference value for the distance traveled (544.28±131.13 meters) for Brazilian children (mean age±SD: 11.28±1.85 years).20 This study sample showed low predicted values for the distance walked in the 10-meter corridor, when compared to healthy Brazilian children.
It can be concluded that this sample of children and adolescents with SCD had FC for exercise below that predicted for the age range when compared to healthy children. Patients diagnosed with HbSS/Sβ0-thalassemia had a worse performance regarding total distance walked, HR, and SpO2 after 6MWT when compared to patients with HbSC/Sβ+-thalassemia. It is suggested that other studies should be conducted with similar designs for evaluation of FC in patients of this age group with SCD, as they show alterations in FC, which are related to the disease prognosis and quality of life.
Conflicts of interestThe authors declare no conflicts of interest.
Please cite this article as: Hostyn SV, de Carvalho WB, Johnston C, Braga JA. Evaluation of functional capacity for exercise in children and adolescents with sickle-cell disease through the six-minute walk test. J Pediatr (Rio J). 2013;89:588–594.