Pediatric acute liver failure (PALF) is a heterogeneous, rare, and severe condition, which outcome is survival due to liver spontaneous recovery or death. The patients who do not recover may be allocated to liver transplantation, which is the standard treatment. This study aimed to build a prognostic model to support the clinical decision to indicate liver transplantation for patients with PALF in a Brazilian center.
MethodsThe authors retrospectively analyzed the clinical variables of 120 patients in the liver transplantation program of the 'Children's Institute of the University of São Paulo, Brazil. The authors conducted a univariate analysis of variables associated with survival in PALF. Logistic multivariate analysis was performed to find a prognostic model for the outcome of patients with pediatric acute liver failure.
ResultsRisk factors were analyzed using univariate analysis. Two prognostic models were built using multiple logistic regression, which resulted in 2 models: model 1(INR/ALT) and model 2 (INR/Total bilirubin). Both models showed a high sensitivity (97.9%/96.9%), good positive predictive value (89.5%/90.4%), and accuracy (88.4%/88.5%), respectively. The receiver operating characteristic was calculated for both models, and the area under the curve was 0.87 for model 1 and 0.88 for model 2. The Hosmer-Lemeshow test showed that model 1 was good.
ConclusionThe authors built a prognostic model for PALF using INR and ALT that can contribute to the clinical decision to allocate patients to liver transplantation.
Pediatric acute liver failure (PALF) is a dynamic and heterogeneous clinical condition manifested by an abrupt onset of a liver-based coagulopathy and biochemical evidence of hepatocellular injury resulting from rapid deterioration in liver cell function.1,2 In children, the current classification is based on the definition established by the Pediatric Acute Liver Failure (PALF) Study Group,3 including the following criteria: a) Acute liver injury (raised transaminases); b) International Normalized Ratio (INR) ≥ 2.0 regardless of hepatic encephalopathy (HE), or ≥ 1.5 with HE; c) no known history of chronic liver disease.
PALF is a rapidly evolving clinical condition, and the standard treatment is liver transplantation (LT).4,5 However, the rarity of PALF and variability in its clinical course complicate the decision process. There are no adequately powered studies to inform diagnostic algorithms, assess markers of disease severity and trajectory, or to guide decisions about LT.6-8 The clinician must construct an individualized diagnostic approach and management strategy. Management requires a multidisciplinary team involving the hepatologist, critical care specialist, and surgeon.4
Early and accurate prognostic assessment of patients with PALF is difficult but critically important for optimum clinical pathways, especially the appropriate utilization of liver transplantation.8 Many different scoring systems have been developed to aid the decision of whether to transplant a patient with PALF or not; however, none of them is accurate enough to predict the outcome.9,10 The etiology of PALF differs worldwide, depending on geographical and socioeconomic characteristics. Although undetermined etiology represents a high percentage in most studies, the viral etiology is predominant in Brazil and Latin America, while acetaminophen-induced PALF and metabolic are more prevalent in high-income countries.11-13
The authors hypothesized that there are specific prognostic factors of Brazilian patients with PALF that could help in the decision-making process. This study aimed to seek risk factors and prognostic models to support the clinical decision to indicate liver transplantation for patients with PALF in a Brazilian LT center.
MethodsThis was a single-center retrospective cohort study of children with PALF admitted to the Liver Transplantation Program of the 'Children's Institute of the University of São Paulo (ICr-USP), Brazil.14 The Liver Transplantation Program of ICr-USP started in 1989. Patients from 2000 until 2019 were included since the electronic health system started at that time and the data was more reliable. The decision to put the patient on the priority list of LT was based on both Kings College and Clichy criteria, following the rule of the Brazilian Ministry of Health.15 When the patient has the criteria for LT, the team waits for a donor while preparing a possible living donor.16 Whilst the patient is awaiting LT, clinical support based on the institutional protocol is given, mainly in the pediatric intensive care unit. Patients may undergo plasmapheresis, continuous renal-replacement therapy, invasive and non-invasive intracranial pressure monitoring. The authors do not have Molecular Adsorbent Recirculating System (MARS).
The inclusion criteria for the study were all pediatric patients of the ICr-USP who met the criteria for PALF, younger than 18 years old. Patients with chronic liver conditions, older than 18 years, and patients with incomplete data in the electronic health record system were excluded.
The sample size was not calculated for this study. Cases were included according to a convenience sample due to the type of exposure and outcome performed. It is an uncommon event in children (PALF), and data were collected retrospectively, including all available cases.
Potential candidate variables for univariate analyses of the association with the outcome of PALF were noted in a spreadsheet for further statistical analysis extracted from the electronic health records of patients. The authors noted demographic data, clinical features, presence of hepatic encephalopathy (HE), etiological diagnosis, and admission laboratory tests, including ammonia, lactate, glucose, total bilirubin (TB), direct bilirubin (DB), aspartate transaminase (AST), alanine transaminase (ALT), INR, albumin, urea, and creatinine.
The main outcome of interest was ""no improvement"" of the patient. The clinical improvement was due to the ""discharge"" and ""no need for transplant"" events. On the other hand, clinical ""not improvement"" was observed in cases with ""need for transplantation"" or ""death"." This has been the primary clinical endpoint defined by the assumption that patients undergoing LT would have otherwise died and are therefore censored from the study when LT is performed. Univariate analyses assessed the association between the selected covariates and survival. The inclusion of multivariable modeling was based on clinical and statistical significance.
All procedures followed the CONSORT guidelines and TRIPOD statement for the transparent reporting of a multivariable prediction model for individual prognosis.17 The study was approved by the Institutional Review Board.
Statistical analysisDescriptive statistics were used to present patient characteristics. Frequencies and percentages were calculated for the qualitative variables. Considering the quantitative variables these were calculated: mean, median, standard deviation, and minimum and maximum values.
Association between qualitative variables and outcome was assessed by Fisher's exact test (when 25% or more of expected values were less than 5) or Pearson's chi-square test. The distribution of quantitative variables was assessed by the Kolmogorov-Smirnov test, and then the Mann-Whitney test was used to compare the groups (improved vs. not improved).
The logistic regression model was used to calculate the regression coefficients and also the odds ratio (OR) values as well as the 95% confidence intervals. The fit of the model was tested by the Hosmer-Lemeshow test under the null hypothesis that the model is good.
The receiver operating characteristic (ROC) curve was used to assess the sensitivity and specificity of the variable associated with the outcome and thus determine the cutoff point. Sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) as well as accuracy were calculated.
The significance level adopted for all tests was 5%. Analysis was performed using the statistical software SPSS v.18 for Windows.
ResultsDemographic characteristics and clinical dataA total of 136 patients with PALF were initially enrolled in the study. However, 12 patients were excluded as they were admitted to the program before 2000, and another 4 patients were excluded due to lack of data, remaining 120 subjects for analysis. Of those, 35 underwent LT and died, 57 underwent LT and survived, 12 died without LT, and 16 survived without LT. Patients were managed according to the institutional clinical protocol that was frequently updated according to medical literature. The demographic and clinical features of patients are described in Table 1.
Demographic and clinical features of patients with PALF.
| Variable | nmissing | Outcome | Total | P value | |
|---|---|---|---|---|---|
| Improved | Not improved | ||||
| n = 16 | n = 104 | n = 120 | |||
| Gender | .053a | ||||
| Female | 12 (75.0) | 51 (49.0) | 63 (52.5) | ||
| Male | 4 (25.0) | 53 (51.0) | 57 (47.5) | ||
| Age in admission (months) | 3 | .745b | |||
| 72.8 (61.8) | 76.3 (57.0) | 75.8 (57.4) | |||
| 63.5 (17.5-121) | 67.0 (19-124) | 67 (18.5-124.4) | |||
| Min-Max | (2-202) | (2-192) | (2-202) | ||
| Origin | 7 | .597c | |||
| São Paulo Estate | 11 (68.8) | 60 (61.9) | 71 (62.8) | ||
| Out of São Paulo Estate | 5 (31.3) | 37 (38.1) | 42 (37.2) | ||
| Jaundice | .035c | ||||
| No | 6 (37.5) | 15 (14.4) | 21 (17.5) | ||
| Yes | 10 (62.5) | 89 (85.6) | 99 (82.5) | ||
| PICU | .005c | ||||
| No | 9 (56.3) | 22 (21.2) | 31 (25.8) | ||
| Yes | 7 (43.8) | 82 (78.8) | 89 (74.2) | ||
| ICP monitoring | .021c | ||||
| No | 16 (100) | 77 (74.0) | 93 (77.5) | ||
| Yes | 0 | 27 (26.0) | 27 (22.5) | ||
| MV | <.001c | ||||
| No | 11 (68.8) | 13 (12.5) | 24 (20.0) | ||
| Yes | 5 (31.3) | 91 (87.5) | 96 (80.0) | ||
| Dialysis | .212c | ||||
| No | 16 (100) | 91 (87.5) | 107 (89.2) | ||
| Yes | 0 | 13 (12.5) | 13 (10.8) | ||
PICU, pediatric intensive care unit; ICP, intracranial pressure; MV, mechanical ventilation.
Note: Values presented as n (%), median (range) or mean ± SD.
Overall, 52.7% of patients were female, the median age at diagnosis was 5.6 (IQR, 1.5-10.4), and 62.8% were from São Paulo State; 99% of patients had jaundice, 10.8% had dialysis, 22.5% had intracranial pressure monitoring, 80% were intubated and put in mechanical ventilation, and 89% were admitted to the pediatric intensive care unit (PICU). Most patients had an indeterminate cause of PALF, and the second cause was viral hepatitis, mainly caused by the hepatitis A virus (Supplemental Digital Content 1).
The factors associated with clinical improvement were assessed by binary logistic regression where the event of interest was "no improvement" in the clinical condition. Initially, all independent variables were evaluated with the outcome of the clinical condition, and the odds ratios (OR) were calculated with their respective 95% confidence intervals. Afterward, multiple regression models were constructed and presented by the values of the correlation coefficients to calculate the probability of "no clinical improvement". It was found that 13.3% (16/120) of the children showed clinical improvement.
Table 2 shows the univariate analysis of variables associated with survival in patients with PALF. The odds ratio (OR) and confidence interval (CI) showed that the presence of jaundice 3.56 (1.13-11.25), PICU admission 4.79 (1.61-14.31), mechanical ventilation 15.40 (4.61-51.45), presence of encephalopathy 8.53 (2.37-30,74), INR 2.109 (1.343-3.310), ALT 0.9996 (0.999-1.00), and TB (1.084 (1.019-1.154) were independently associated with the clinical condition.
Univariate analysis of variables associated with survival in patients with PALF.
OR, Odds ratio; CI95%, confidence interval of 95%; SD, Standard Deviation; NE, not evaluable.
LV, liver transplantation; PICU, pediatric intensive care unit; ICP, intracranial pressure; MV, mechanical ventilation; HAV, hepatitis A virus; INR, international normalized ratio; AST, aspartate transaminase; ALT, alanine transaminase.
To better understand the variables associated with death or liver transplantation in the present study's cohort, the authors conducted a multiple logistic regression analysis that could explain the condition of the clinical worsening of children with PALF. Two prognostic models were tested based on the results of the univariate analysis, which resulted in 2 prognostic models to be tested: model 1 (INR and ALT) and model 2 (INR and TB). The regression coefficients for each possible model were calculated, as well as measures of accuracy. Table 3 shows the description of variables and models constructed for children admitted with PALF. Both models were subjected to the Hosmer-Lemeshow test that assesses the fit of the model under the null hypothesis that ""the model is good"." It appears that for model 1, the null hypothesis is not rejected (p = 0.607). Alternatively, model 2 rejected the null hypothesis that the model is good (p = 0.025). Both prognostic models 1 and 2 showed high sensitivity (97.9%/96.9%) and accuracy (88.4%/88.5%), respectively. Another key value for prognostic models is the predictive value. Model 1 has a positive predictive value (PPV) of 89.52%, while model 2 has a PPV of 90.38%.
Variables and models of prediction for PALF.
CI95%, confidence interval of 95% for beta (β); ALT, alanine transaminase; TB, total bilirubin.
Prognostic model 1 (INR/ALT) has the statistical features of a good prognostic model. The equation to determine the probability of not improving the clinical condition of children with PALF according to prognostic model 1 is the following.
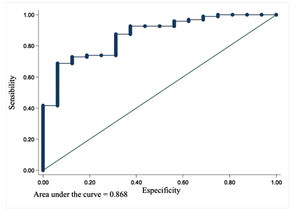
The ROC for the prognostic, model 1 was analyzed, and the area under the curve (AUC) was 0.868 (Fig. 1).
DiscussionTo our knowledge, this is the first prognostic model for PALF built in Latin America. The analysis of patients’ recorded data allowed us to determine risk factors associated with the outcome and build a prognostic model for patients with PALF. Univariate analyses assessed the association between the selected covariates and survival without LT. The authors tested all independent variables and only found statistical significance for the presence of encephalopathy (p = 0.001), INR (p = 0.001), ALT (p = 0.029), and TB (p = 0.011). Multivariate logistic regression was employed to build two prognostic models: model 1 (INR/ALT) and model 2 (INR/TB), based on the variables previously studied by the univariate analysis. The receiver operating characteristic (ROC) was calculated for both models, and the area under the curve (AUC) was 0.87 for model 1 and 0.88 for model 2, which is good considering that 1.0 is ideal and > 0.8 is considered acceptable.9 The AUC or ""c-statistic"" summarizes how good the model is at discriminating between outcomes. It allows us to create an ROC curve and a complete sensitivity/specificity report. The ROC curve is a fundamental tool for diagnostic test evaluation.18 The prognostic model 1 (INR/ALT) has an AUC = 0. The Hosmer-Lemeshow test is a ""goodness of fit"" test for logistic regression, especially for risk prediction models. A goodness of fit test tells you how well your data fit the model.19 Model 2 (INR/TB) failed to pass the Hosmer-Lemeshow test, rejecting the null hypothesis (the model is good) (p = 0.025). However, model 1 (INR/ALT) did not reject the null hypothesis (p = 0.607) and was considered a good prognostic model. To summarize, in terms of accuracy, both models 1 and 2 have good discrimination (c-statistic), but only model 1 has good calibration (goodness-of-fit) and was elected as the prognostic model that the authors were looking for.
In PALF, an appropriate balance between sensitivity and specificity is essential, as reduced sensitivity (low positive predictive value) could lead to the failure to list a patient for LT who would have subsequently died, but reduced specificity (low negative predictive value) carries a risk of unnecessary LT in a patient who was likely to recover spontaneously.9,18,19
Multiple models to identify early prognostic indicators and estimate prognosis for liver failure have been proposed and widely used as a resource; however, their accuracy and efficacy have been extensively debated.7,10,20 Multivariable prognostic models, including the King's College Hospital criteria, the Pediatric End-Stage Liver Disease (PELD)/Model for End-Stage Liver Disease (MELD) score, and Liver Injury Units (LIU), have been widely employed to determine the prognosis of PALF, but the results are far from satisfactory.7 Moreover, some prognostic models, such as the KCH criteria, have proven to have a low sensitivity to determine the outcome, although their specificity is acceptable.21,22 Dynamic prognostic models try to predict the outcome with multiple measures since PALF is a dynamic syndrome, although it lacks a wider validation.23 Determining the likelihood of either spontaneous native liver recovery or death in patients with PALF is the most challenging assessment in this scenario.9 Besides that, the results are contradictory depending on the region of the world the prognostic model is employed.24,25
The authors did not compare the just build model to other established ones for many reasons. First, as it was a retrospective study, the authors did not have all data required to conduct that comparison. Second, as there is no gold-standard model to compare with, the authors would be comparing it to other imperfect ones. The authors also have to point out that the authors’ model provides a percentage of the chance of non-recovery for PALF patients, which there is no specific threshold.
Several prognostic scores have been developed to aid decision-making for liver transplantation; however, none of them were based in a developing country population.
A prognostic model in medicine is designed to produce indices to enable the estimation of the risk of future events in individual patients/groups and to risk stratify these patients.9,19,20 The ideal model-derivation population should be large, representative of the diseased cohort, and entail a reasonable proportion of the outcome measures.3,23 In this study, the authors had a fair number of subjects (n = 120), considering the rarity of the disease; and the sample is representative of the Brazilian population since it is the most important center of LT in Brazil, which is a reference for PALF, admitting almost all cases in Brazil. The authors built a prognostic model based on variables of the studied population, and we think it is one of a strength of this study.
A prognostic model that fits all cases seems unfeasible. There are even specific prognostic models for certain etiologies of PALF, such as for hepatitis A-induced PALF and Wilson disease.26,27 Maybe different prognostic variables or PALF scoring systems could be adapted according to regional variables significantly associated with worse outcomes in different areas. The prognostic model that resulted from this study should be validated in other centers in Brazil and Latin America.
Another issue is that the etiology of PALF seems to determine the clinical course and progression of the disease as well as the need for specific therapy.1 The present study's data showed that indeterminate cause is the major etiology accounting for roughly 60% of cases, which is high compared to reported studies in other countries such as Italy (47%),28 Spain (36.7%),13 and Canada and the USA (30.8%).29 A recently published review found that the main causes of PALF in Latin America and the Caribbean are viral hepatitis and poisonings, and 38.4% of subjects had undetermined causes.30 The mortality rate varies among different centers.6,12,13,31,32 The mortality rate is in line with the PALFSG databank, which reported a mortality rate of 11% among 769 patients.32
This study has several limitations. Firstly, this study was a retrospective study, and some data were unavailable. Secondly, this was a single-center study, and the results cannot be generalized to other centers or other parts of the world. Also, the authors were unable to determine the cause of PALF in roughly half of the patients. Another issue is that most patients were referred to the present study's center, and these patients could be in worse clinical condition since only 13% had a spontaneous recovery, which could be a selection bias. The authors also consider a limitation the fact that this is not a dynamic model. Lastly, most prognostic models are derived and validated retrospectively; hence, missing information and different time points for data collection (admission versus study enrollment data; early versus late transfer to tertiary center) can be confounders.
The construction of this prognostic model is considered the first step of a program that aims to validate it internally and externally, both in Brazil and in Latin America. It would be a challenge to also validate it in high-income countries, which have a different populations, phenotypes, and prevalence of PALF.
ConclusionUsing clinical data derived from a Brazilian single-center registry database, the authors build a multivariable logistic regression prognostic model using INR and ALT. Validation studies are required.
FundingThe authors received no grants for this study.