To assess the prevalence of chronic neutropenia (CN) and the clinical profile of patients with CN aged up to 18 years, followed in the pediatric hematology, rheumatology, or immunology outpatient clinic of a tertiary medical center from May 1, 2018, to 30 April 2019.
MethodsRetrospective observational study carried out by collecting data from the patient's medical charts. CN was defined as absolute neutrophil count (ANC) below 1.5 × 109/L lasting over three months. Autoimmune neutropenia (AIN) was defined by clinical criteria and an over twofold increase in ANC after glucocorticoid stimulation. AIN was considered secondary when associated with autoimmune or immunoregulatory disorders. Wilcoxon and Fisher's exact tests were used to compare variables; the significance level was 5 %.
ResultsA total of 1,039 patients were evaluated; 217 (20 %) presented CN. Twenty-one (2 %) had AIN, classified as primary in 57 % of the cases. The average age at the onset of symptoms was 38.6 months. During follow-up, patients had 4.2 infections on average; frequency was higher among patients with secondary AIN (p = 003). Isolated neutropenia occurred in 43 % of the patients with AIN. Neutropenia resolved in eight (38 %) of the 21 patients with AIN within 19.6 months on average. Eight patients with secondary AIN met the criteria for Inborn Errors of Immunity.
ConclusionAIN prevalence was 2 %. Most cases were first evaluated by a pediatric immunologist or rheumatologist rather than a pediatric hematologist. This study highlights the need for a multidisciplinary approach involving a pediatric immunologist, rheumatologist, and hematologist.
Neutropenia is defined as absolute neutrophil count (ANC) below normal levels for age.1 The prevalence of childhood neutropenia varies considerably with geoethnicity and is usually benign. Denic et al. analyzed ANC in 26,542 children aged from one day to six years and found that neutropenia frequency ranged from 15.4 % in North African Arabs to less than 1 % in Peninsular-Arabs.2
The lower limit of ANC is 6 × 109/L at birth, 1 × 109/L in children aged between two weeks and one year, and 1.5 × 109/L in children older than one year. The accepted lower limit of ANC in African-American children is 1.2 × 109/L. Moderate and severe neutropenia is considered when ANC lies between 0.5 and 1.0 × 109/L and below 0.5 × 109/L, respectively.3,4 Neutropenia lasting longer than three months is considered chronic neutropenia; shorter-duration neutropenia is generally associated with acute and convalescent phases of viral infections. Increased risk of mortality from severe or opportunistic infections occurs only in 12–20 % of children and is associated with ANC lower than 0.2 × 109/L and persistent neutropenia.5
The main causes of chronic neutropenia are reduced neutrophil production or inability to transfer neutrophils from the bone marrow to peripheral blood, increased peripheral neutrophil sequestration (pseudoneutropenia), and neutrophil destruction. Chronic neutropenia can be classified into congenital neutropenia and acquired neutropenia. Congenital neutropenia includes cyclic neutropenia, myelokathexis, Shwachman-Diamond-Oski syndrome, Kostmann syndrome, Chédiak-Higashi syndrome, and reticular dysgenesis. In turn, the major causes of acquired neutropenia are infections, hypersplenism, treatment with drugs (in neonate consider maternal treatment), nutritional deficiency (vitamin B12 or folate), maternal hypertension (neonate), autoimmune neutropenia/chronic benign neutropenia (neonatal isoimmune, alloimmune, and primary and secondary to systemic autoimmune diseases), diseases replacing the bone marrow (neoplasms, leukemia, and myelodysplastic syndromes), bone marrow failures (aplastic anemia, Fanconi anemia, and congenital dyskeratosis), pure white cell aplasia (Blackfan-Diamond), and immunodeficiency disorders.4,6
The terms autoimmune neutropenia (AIN) and idiopathic neutropenia (IN) are confused in the literature and have similar clinical profiles and outcomes. The gold standard to confirm AIN is the presence of serum antineutrophil antibodies directed against neutrophil cell surface membrane antigens (HNA-1a, HNA-1b, HNA-1c, HNA-2a, HNA-3a, HNA-4a, and HNA-5a).3 However, it is technically difficult to detect specific autoantibodies in clinical practice, so AIN and IN are considered as possibly the exact etiology and follow-up is similar in both cases. Antineutrophil detection methods are poorly sensitive and specific, so two tests must be combined to confirm a diagnosis of AIN, for example, the semiquantified granulocyte indirect immunofluorescence test (GIIFT) and the agglutination test.7,8
AIN is classified as secondary when it is associated with systemic autoimmune diseases, like systemic lupus erythematosus. Secondary AIN has a good prognosis, and neutropenia usually resolves spontaneously.9
In clinical practice, the differential diagnosis remains a challenge, especially when chronic neutropenia appears in isolation or as the first manifestation of a systemic disease.
Few studies have evaluated chronic neutropenia prevalence and clinical course in specific groups and conducted long-term follow-up. This study aims to evaluate the prevalence of chronic neutropenia and the clinical profile of patients with a diagnosis of this condition, aged up to 18 years and followed in the pediatric hematology, rheumatology, or immunology outpatient clinic of a tertiary medical center.
Materials and methodsPopulationThis retrospective observational study was conducted between May 2018 and April 2019 at the Clinics Hospital of the Ribeirão Preto Medical School, University of São Paulo, Brazil. Patients aged up to 18 years with chronic neutropenia were eligible for the study. Patients were selected among those who attended the pediatric rheumatology, immunology, or hematology outpatient clinic.
The authors evaluated all blood counts performed by patients followed at these outpatient clinics during the study period. The records of patients with neutrophil counts below 1.5 × 109/L were selected for chart review, classified as chronic neutropenia (CN), and then classified as AIN according to the criteria described below.
Data collectionDemographic data, clinical features, laboratory findings, and therapeutic data were retrieved from the patient's medical charts.
CN was defined as ANC below 1.5 × 109/L, lasting over three months, and confirmed in three blood counts collected at different times4. Severe neutropenia was defined as ANC below 0.5 × 109/L.3,4
AIN was defined according to the criteria suggested by Newburger et al.5 In patients with confirmed chronic neutropenia, patient and familiar clinical history, leukocyte morphology, quantitative IgG, IgA, and IgM, and antinuclear antibodies were evaluated. If the diagnosis remained uncertain, AIN was considered if ANC increased over twofold after the glucocorticoid stimulation test.5 The bone marrow was examined following the hematologist's recommendation.
AIN was considered secondary when it was associated with autoimmune or immunoregulatory diseases. In turn, AIN was considered primary when no other genetic, infectious, inflammatory, or malignant causes were identified.10
Neutropenia was considered resolved when ANC in peripheral blood remained above 1500/mm3 in three samples after over three months.11
Ethical considerationsThe study was approved by the Clinics Hospital of the Ribeirão Preto Medical School Research Ethics Committee (process number 2.689.688)
Statistical analysisThe characteristics of the study sample were summarized by using descriptive statistics. Ordinal data are presented as percentages, and continuous data are presented as means, standard deviations [SD] or medians, and interquartile ranges, as appropriate.
The Wilcoxon test was used to compare the means of continuous variables between the groups. Fisher's exact test was employed to compare categorical variables between the groups. A significance level of 5 % was adopted in all the analyses. Data were analyzed by using the SAS 9.4 software (SAS Institute Inc., SAS/STAT® User's Guide, Version 9.4, Cary, NC: SAS Institute Inc., 2013).
ResultsOf the 1039 patients followed up at the outpatient clinics during the study period, 217 (20 %) had chronic neutropenia. Among the patients with CN, 196 (90.3 %) had hematological causes that justified the condition, including leukemia, lymphoma, myelodysplastic syndrome, bone marrow aplasia, neutropenia induced by chemotherapy drugs, immunosuppressant, and congenital neutropenia. Three cases of congenital and severe neutropenia were genetically confirmed: two cases presenting a germline pathogenic mutation in ELANE and one case with a pathogenic mutation in the GFI1gene.
Two percent of the 1039 patients selected for this study met the classification criteria established for AIN. When the authors considered only the patients with chronic neutropenia, 9.6 % had AIN, and 57.2 % of them were classified as primary AIN.
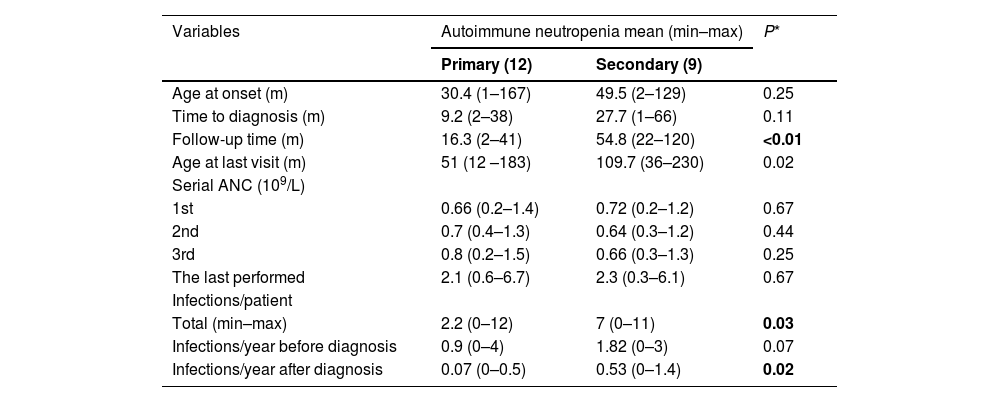
Table 1 describes the clinical profile of the 21 patients with AIN. Almost 50 % of these patients had isolated neutropenia in the blood count. They were followed up at the pediatric hematology, immunology, or rheumatology outpatient clinic—10 (47.6 %), 9 (42.9 %), and 2 (9.5 %) patients, respectively. Bone marrow aspiration, performed in 12/21 (57 %) patients, did not show significant changes. During follow-up, the authors found an average of 4.2 infections per patient; the most frequent infections were sinusitis and pneumonia, which led to nine hospitalizations. Ten patients had severe infections; six of these patients required admission to the intensive care unit, and four had febrile neutropenia.
Demographic, clinical, and follow-up data of 21 patients aged under 18 years with autoimmune neutropenia.
N, number of cases; min, minimum; max, maximum.
When the authors compared the clinical data of the patients with primary and secondary AIN, the follow-up period (p < 0.01) and the number of infections (p = 0.03) were higher in the latter group. In eight of the 21 patients with AIN, neutropenia resolved at 11.5 (3–20) and 27.7 (7–60) months in patients with primary (four patients) and secondary (four patients) AIN, respectively. Three patients with primary AIN had a spontaneous resolution, and one patient with primary AIN required corticosteroids. All the patients with secondary AIN required specific treatments (Table 2).
Comparison between primary and secondary autoimmune neutropenia in 21 patients aged under 18 years.
ANC, absolute neutrophil count. *Wilcoxon test; m (months).
The patients with and without resolved neutropenia, as well as the patients with criteria for severe and non-severe neutropenia, did not differ significantly in terms of the studied variables (age of onset, time to diagnosis, follow-up time, and number of infections).
Eight patients with secondary AIN met the criteria for Inborn Errors of Immunity, namely autoimmune lymphoproliferative syndrome (ALPS), adenosine deaminase 2 deficiency (DADA2), common variable immunodeficiency (CVID), DiGeorge Syndrome, severe combined immunodeficiency (SCID), and Wiskott-Aldrich Syndrome (WAS), and one patient had juvenile dermatomyositis and developed non-medication-related neutropenia. In patients with ALPS, DADA2, and WAS, hematological alterations were the first manifestation of the disease. Table 3 describes the clinical profile of these patients.
Demographic, clinical, and follow-up data of nine patients aged under 18 years with secondary autoimmune neutropenia.
M: masculine; F: feminine; Y: years; ANC: absolute neutrophil count; RI: recurrent infections; LPS: lymphoproliferative symptoms; NR not done; PTH: parathyroid hormone; TB: tuberculosis; CMV: cytomegalovirus; EBV: Epstein-Barr Virus; DNT: double negative T cells (CD3+TCR alpha/beta+CD4-CD8-); EMG: electromyography; p: percentile G-CSF: Granulocyte colony-stimulating factor; IV: intravenous; SC: subcutaneous, IG: human immunoglobulin; MMF: mycophenolate mofetil; MTX: methotrexate; DADA2: deficiency adenosine deaminase 2; JDM: juvenile dermatomyositis; ALPS: Autoimmune Lymphoproliferative Syndrome; CVID: Common variable immunodeficiency; SCID: severe combined immunodeficiency; MLPA: multiplex ligation-dependent probe amplification method; WASp: Wiskott-Aldrich syndrome protein ⁎⁎ compound heterozygous mutation – ADA2 gene (NM_0001282225) – exon 3 c.369_397delp. (His 133LeufsTer44) and exon 4 c563T>C p. (Leu188Pro).
The prevalence of patients with chronic neutropenia followed at the rheumatology, hematology, or immunology outpatient clinic was 20 %. Two percent of the patients with chronic neutropenia had chronic idiopathic neutropenia, which the authors classified as AIN according to clinical criteria.5 Karapinar et al. found a frequency of 0.9 % of chronic idiopathic neutropenia among patients of a hematology outpatient clinic.12 Here, to evaluate neutropenia, the authors deliberately chose patients of the pediatric hematology, rheumatology, or immunology outpatient clinic because the incidence of neutropenia among them was likely higher. If the authors had not considered only the outpatient clinics related to oncological and autoimmune diseases, the incidence of neutropenia would probably have been lower. In a population database, Anderson et al. retrospectively evaluated routine complete blood cell count in individuals aged from 1 to 90 years, to find a 0.9 % prevalence of acute neutropenia and a 0.06 % prevalence of chronic neutropenia.13
According to the Italian Neutropenia Registry, a positive GIIFT indicated AIN, whereas four negative GIIFTs without associated underlying diseases indicated “idiopathic neutropenia”; however, the two groups were clinically very similar. Kobayashi described similar results when evaluating children with chronic neutropenia by quantitative analysis of antineutrophil antibodies by GIIFT and the micro-leukocyte agglutination test (MLAT) — age at diagnosis, neutropenia severity, and frequency of infection among the patients with negative, weakly positive, and strongly positive antibody titers did not differ significantly.8 Therefore, IN and AIN in childhood have similar clinical characteristics, as suggested by several authors. Most of these cases probably have autoimmune origin even though antineutrophil antibodies may not always be detected.14 Thus, in the present real-life study, the authors considered IN/AIN in the same group of patients for analysis.
Although antineutrophil antibodies were not available, all the patients fulfilled the autoimmune neutropenia classification criteria suggested by Newburger et al.5
Among the patients with AIN, 62 % were male. Studies have reported different prevalence of AIN in terms of gender: in adults, the condition usually predominates in female patients, at an 8:1 ratio.15 In children, the female-to-male ratio is lower16. In the Italian Neutropenia Registry, 56.8 % of the patients with AIN and 50.6 % of the patients with IN were male.8 The different methodologies adopted in the studies make comparison difficult.
In the present study, the mean onset of AIN was 38.6 months, and the mean time to diagnosis was 17 months; in some cases, specific diagnosis was delayed up to five years. In the Italian Neutropenia Registry, the mean age at diagnosis was lower: 9.6 and 14.4 months for AIN and IN, respectively.8 Concerning severe neutropenia, the mean age of the patients with IN was 15 months (spanning from 3 months to 17 years) in a single center in Pittsburgh within a four-year period.17 The results of this study differed from the literature results possibly because the authors conducted the study at a tertiary hospital and, when the patient was referred to this hospital, they already had some symptoms and featured more prolonged evolution, not to mention that many patients had not been subjected to any previous routine exams to confirm the onset of neutropenia.
Most patients with a diagnosis of AIN only had hematological alterations in their initial clinical profile, and 43 % had isolated neutropenia. Nevertheless, 52 % of the cases were first evaluated by a pediatric immunologist or rheumatologist and not by a pediatric hematologist, showing that it is important to recognize and update how these professionals manage these diseases.
The main symptoms associated with hematological changes were hepatomegaly (19 %) and splenomegaly (14.2 %). Other symptoms, such as adenomegaly, fever of unknown origin, and cutaneous livedo reticularis are generally associated with the causes of secondary AIN. This clinical profile places secondary AIN in the group of differential diagnoses of other possible diseases that may present with neutropenia, including neoplasms and infections.18
The authors classified 57 % of the AIN cases as primary. Farrugia et al. showed a prevalence of 91 % primary AIN and 9 % secondary AIN in patients with AIN.19 The high frequency of secondary AIN in the present study could be justified by the profile of the outpatient clinics where the patients were followed. Many cases of primary, mild, and transient neutropenia are probably followed in less complex hospitals.
Inborn Errors of Immunity (IEI) were the main final diagnoses related to secondary neutropenias in this study. Three diagnoses were confirmed by the molecular test that detected pathogenic variants in genes ADA220 and JAK3 and a 22q11.2 deletion; the test used multiplex ligation-dependent probe amplification on DNA (MLPA). By the increase ANC over twofold after the glucocorticoid treatment, the authors considered the AIN diagnosis in this case.
The South African Primary Immunodeficiency Registry showed that 62 % of patients with IEI had cytopenia at presentation, with 19 % having neutropenia.21 Several mechanisms can contribute to non-immune cytopenia in IEI, including cytoskeletal dysfunction with megakaryocyte dysfunction responding by thrombocytopenia and defective myeloid cell differentiation or release of granulocytes from the bone marrow, enhanced apoptosis, and increased destruction of peripheral-blood granulocytes as the leading causes of neutropenia.22 Furthermore, a strong relationship between autoimmune cytopenias and IEI has been reported.23 When the very early onset of neutropenia is present, this group of diseases must be considered in addition to other associated suggestive signs, such as severe infections by atypical agents, vasculitis, livedo reticularis, other autoimmune symptoms, and family history.23,24 These patients should be screened for humoral and cellular immune response, and they generally need immunomodulatory and immunosuppressant drugs to control cytopenias, another reason why early diagnosis is paramount.
Unfortunately, the authors did not identify signs or symptoms that would allow primary and secondary AIN to be differentiated early or help in diagnosis or prognosis. Only the number of infections was significantly higher in the patients with secondary AIN and could serve as an alert for diagnosing primary immunodeficiencies. Farruggia et al. compared cases of primary and secondary AIN and identified that monocytosis was more associated with primary AIN, and that female gender and the occurrence of severe infections were more associated with secondary AIN.19 Fioredda et al. showed that neutropenia either appeared in patients aged over five years or lasted longer than three years, and the condition had a far inferior resolution rate and increased autoimmunity frequency over the course of the disease.11
Infections are among the greatest concerns in patients with neutropenia. In this study, there were an average of 4.2 infections per patient; one of the patients had 12 infections during the evaluation period. After diagnosis, the average number of infections dropped from 1.2 per year to 0.2 per year, reinforcing that the causes of neutropenia must be identified early and that specific treatment must be introduced as soon as possible. Kirk et al. identified that infections occurred in 77 % of the patients with AIN, and that the most frequent infections were acute otitis media, skin infections, and pneumonia.25 Farruggia et al. found a higher frequency of infections in patients with secondary AIN (40 %) when compared to patients with primary AIN (11.8 %).19 The authors also found a higher frequency of infection in the patients with secondary AIN.
Regarding patient evolution, neutropenia resolved in 38 %. The median time to recovery of neutrophil levels was 19.6 months (from 3 to 60 months). Follow-up was longer in the patients with secondary AIN compared to the patients with primary AIN, reflecting the shorter time needed for primary autoimmune neutropenia to resolve. In the literature, similar times to neutropenia resolution have been reported. Kirk et al. observed a mean time to neutropenia resolution of 18 months (from 2 to 85 months).25 Dale and Bolyard observed neutropenia remission mainly in children aged between three and five years; the mean duration was 17 months.16
The present study has some limitations: it is retrospective and based on patients’ medical charts, and a limited number of patients were evaluated. Another challenge was to perform clinical characterization of the patients with autoimmune neutropenia without analyzing antineutrophil antibodies. Given that this is a common difficulty in the literature, the authors performed the diagnosis based on clinical criteria, exclusion of other causes, and response to treatment in a real-life study.5 Despite these limitations, this study has addressed relevant points: it has associated neutropenia frequency with immunodeficiency and autoimmune disorders, confirmed good prognosis in most cases, and verified the need for an integrated follow-up with a pediatric hematologist, immunologist, and rheumatologist when confirmation and management are difficult.
In the present study, the authors found that the AIN prevalence was 2 %. Most cases were first assessed by a pediatric immunologist or rheumatologist and not by a pediatric hematologist, so it is necessary to recognize and update how these diseases are managed by these professionals. In conclusion, this study highlights the need for a multidisciplinary approach that involves a pediatric immunologist, rheumatologist, and hematologist.