To describe the clinical, laboratory, and radiological characteristics, as well as the outcomes of children with MIS-C.
MethodMulticenter, prospective cohort study, conducted in 17 pediatric intensive care units in five states in Brazil, from March to July 2020. Patients from 1 month to 19 years who met the MIS-C diagnostic criteria were included consecutively.
ResultsFifty-six patients were included, with the following conditions: Kawasaki-like disease (n = 26), incomplete Kawasaki disease (n = 16), acute cardiac dysfunction (n = 10), toxic shock syndrome (n = 3), and macrophage activation syndrome (n = 1). Median age was 6.2 years (IQR 2.4−10.3), 70% were boys, 59% were non-whites, 20% had comorbidities, 48% reported a contact with COVID-19 cases, and 55% had a recent SARS-CoV-2 infection confirmed by RT-PCR and/or serology. Gastrointestinal symptoms were present in 71%, shock symptoms in 59%, and severe respiratory symptoms in less than 20%. d-Dimer was increased in 80% and cardiac dysfunction markers in more than 75%. Treatment included immunoglobulin (89%); corticosteroids, antibiotics, and enoxaparin in about 50%; and oseltamivir and antifungal therapy in less than 10%. Only 11% needed invasive mechanical ventilation, with a median duration of five days (IQR 5–6.5). The median length of PICU stay was six days (IQR 5–11), and one death occurred (1.8%).
ConclusionsMost characteristics of the present MIS-C patients were similar to that of other cohorts. The present results may contribute to a broader understanding of SARS-CoV-2 infection in children and its short-term consequences. Long-term multidisciplinary follow-up is needed, since it is not known whether these patients will have chronic cardiac impairment or other sequelae.
In April 2020, a few months after the beginning of the SARS-CoV-2 virus pandemic, some reports called attention to the increase in hospitalizations of children and adolescents with clinical features similar to toxic shock syndrome or Kawasaki disease (KD).1,2 This condition, initially called pediatric multisystem inflammatory syndrome temporally associated with COVID-19, was later named multisystemic inflammatory syndrome in children (MIS-C). Several studies have described this new syndrome,3–7 and both the Centers for Disease Control and Prevention (CDC) and the World Health Organization (WHO) have released a case definition.8,9 Currently, it is already known that MIS-C and KD are different clinical entities,10,11 and that MIS-C may present with different phenotypes.11,12
To date, few studies have been published on COVID-19 or MIS-C in Latin America. A multicenter study from Chile, Colombia, and other countries reported 17 children with COVID-19, but no case of MIS-C13; another multicenter study in Brazil reported 79 confirmed cases of SARS-CoV-2 infection, of which ten were classified as MIS-C14; and the other two were case series from southeastern Brazil (66 cases, six of MIS-C),15 and from northern Brazil (11 cases of MIS-C).16
The present study aimed to describe the epidemiological, clinical, laboratory, and radiological characteristics, as well as outcomes of a broader cohort of children with MIS-C in Brazil, the epicenter of the COVID-19 epidemic in South America.
Materials and methodsStudy design, patient selection, and settingThis was an observational, multicenter study, partly retrospective, partly prospective, conducted in 17 PICUs from five states in Brazil: 11 in the Southeast, five in the Northeast, and one in the North; 12 private, four public, and one mixed; all associated with the Brazilian Research Network in Pediatric Intensive Care (BRnet-PIC). From March 25 to August 23, 2020, pediatric patients (age range: 1 month–19 years) were consecutively included if they met the CDC case definition8 for MIS-C: 1) fever >38.0 °C for ≥24 h (objective or subjective); 2) laboratory evidence of inflammation, including, but not limited to, one or more of the following: high values of C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), fibrinogen, procalcitonin, d-dimer, ferritin, lactic acid dehydrogenase (LDH), or interleukin 6 (IL-6); elevated neutrophils, reduced lymphocytes, and low albumin; 3) no alternative plausible diagnosis; 4) current or recent SARS-CoV-2 infection diagnosed by a positive reverse transcription polymerase chain reaction (RT-PCR) or positive serological tests (IgM, IgG or IgA), or exposure to a suspected or confirmed COVID-19 case within the four weeks prior to the onset of symptoms.
The study was approved by the Research Ethics Committees of all institutions. The families agreed to participate by signing an informed consent form.
Data collectionDemographic, clinical, epidemiological, radiological, laboratory, and outcome data were prospectively collected, using standardized case report forms (REDCap; Vanderbilt University – TN, United States). Laboratory tests were ordered at the discretion of the health team in each pediatric intensive care unit (PICU) and according to local availability. Tests to detect SARS-CoV-2 infection were performed by RT-PCR using oro/nasopharyngeal swabs or tracheal aspirates and/or serology. The serological tests to detect the presence of specific COVID-19 antibodies in the blood/serum of the patients were of two types: 1) rapid immunodiagnostic tests (commercially available immunochromatographic lateral flow immunoassays) for detection of IgM and/or IgG; 2) enzyme-linked immunosorbent assays (ELISA) for detection of IgM, IgG, and IgA, according to the local availability of these diagnostic tests.
Data processing and statistical analysisContinuous variables were described as medians and interquartile ranges (IQRs) and categorical variables as frequencies and percentages. Demographic, clinical, laboratory, and outcome data were categorized in the following groups: Kawasaki-like disease, incomplete KD, acute cardiac dysfunction, toxic shock syndrome, and macrophage activation syndrome. The analyses were performed using the software R (v. 3.6.1, R Foundation – Vienna, Austria).
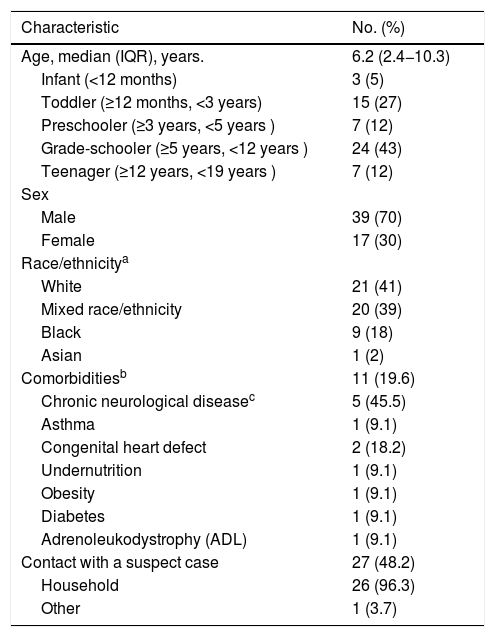
ResultsFifty-six patients were included. The median age was 6.2 years; 70% were boys; 57% were of mixed-race or Black; about 20% had comorbidities, of which almost half were chronic neurological diseases; and of the patients who reported contact with a suspected case (approximately half), almost 100% were household contacts. SARS-CoV-2 infection could be confirmed in 55% of patients, since RT-PCR and serology immunoassays were not available in all PICU: 14 out of 30 tested positive by RT-PCR, of whom five also presented serology (two positive); 19 out of 22 tested positive by serology, of whom 18 also underwent RT-PCR (16 negative; Table 1). All cases described as confirmed SARS-CoV-2 infection in this study had either a positive RT-PCR or a positive serology result.
Demographic, clinical history and diagnostic data of the MIS-C cohort (n = 56).
| Characteristic | No. (%) |
|---|---|
| Age, median (IQR), years. | 6.2 (2.4−10.3) |
| Infant (<12 months) | 3 (5) |
| Toddler (≥12 months, <3 years) | 15 (27) |
| Preschooler (≥3 years, <5 years ) | 7 (12) |
| Grade-schooler (≥5 years, <12 years ) | 24 (43) |
| Teenager (≥12 years, <19 years ) | 7 (12) |
| Sex | |
| Male | 39 (70) |
| Female | 17 (30) |
| Race/ethnicitya | |
| White | 21 (41) |
| Mixed race/ethnicity | 20 (39) |
| Black | 9 (18) |
| Asian | 1 (2) |
| Comorbiditiesb | 11 (19.6) |
| Chronic neurological diseasec | 5 (45.5) |
| Asthma | 1 (9.1) |
| Congenital heart defect | 2 (18.2) |
| Undernutrition | 1 (9.1) |
| Obesity | 1 (9.1) |
| Diabetes | 1 (9.1) |
| Adrenoleukodystrophy (ADL) | 1 (9.1) |
| Contact with a suspect case | 27 (48.2) |
| Household | 26 (96.3) |
| Other | 1 (3.7) |
| SARS-CoV-2 tests | Confirmed COVID-19 N (%) | Non-confirmed COVID-19 (N%) |
|---|---|---|
| Total | 31 (55.4) | 25 (44.6) |
| RT-PCR | ||
| Negative | 16 (51.6) | 11 (44) |
| Positived | 14 (45.2) | 0 (0) |
| Not tested | 1 (3.2) | 14 (56) |
| Serology | ||
| Negative | 3 (9.7) | 7 (28) |
| Positived | 19 (61.3) | 0 (0) |
| Not tested | 9 (29) | 18 (72) |
MIS-C, multisystem inflammatory syndrome in children; IQR, interquartile range; RT-PCR, reverse transcription polymerase chain reaction.
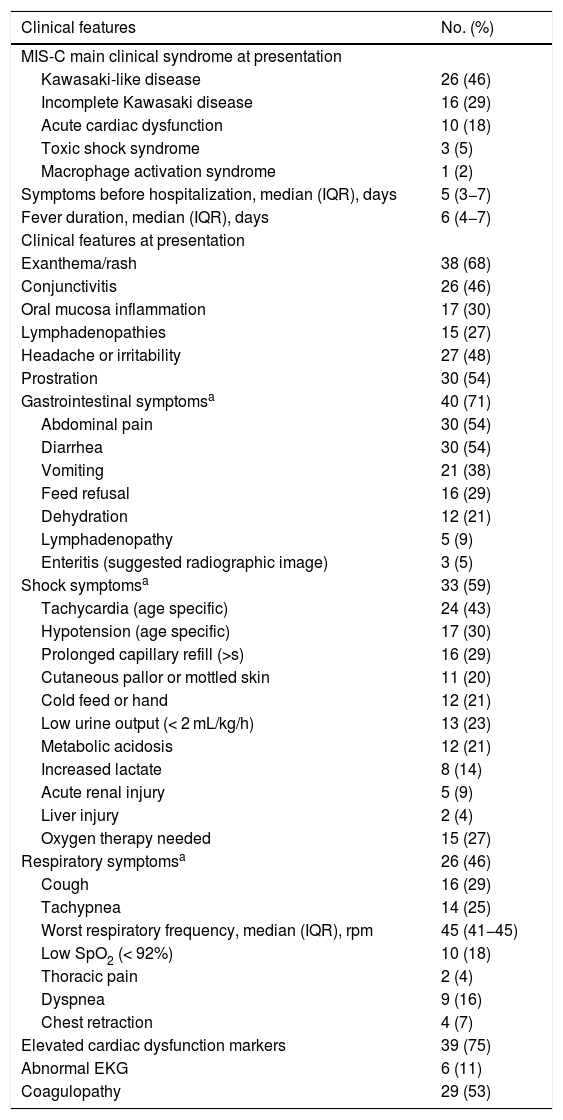
The main multisystem inflammatory phenotype at presentation was Kawasaki-like disease (46%) and incomplete KD (29%), followed by acute cardiac dysfunction (18%), toxic shock syndrome (5%), and macrophage activation syndrome (2%). The median duration of symptoms before hospitalization was five days, and the median duration of fever was six days. Gastrointestinal symptoms were present in 71% of patients, mainly abdominal pain (54%), diarrhea (54%), and vomiting (38%). Skin rash (68%), prostration (54%), and headache or irritability (48%) were also very common. Shock symptoms were present in 59% of patients, mainly tachycardia (43%), hypotension (30%), and prolonged capillary refill (29%). Respiratory symptoms occurred in 46% of patients, but severe symptoms, such as low SpO2 (18%) and dyspnea (16%) were not very frequent. All patients had elevated inflammatory markers, and over 75% had elevated cardiac dysfunction markers. More than half (53%) also had laboratory signs of coagulopathy (Table 2).
Clinical features and radiologic findings of the MIS-C cohort (n = 56).
| Clinical features | No. (%) |
|---|---|
| MIS-C main clinical syndrome at presentation | |
| Kawasaki-like disease | 26 (46) |
| Incomplete Kawasaki disease | 16 (29) |
| Acute cardiac dysfunction | 10 (18) |
| Toxic shock syndrome | 3 (5) |
| Macrophage activation syndrome | 1 (2) |
| Symptoms before hospitalization, median (IQR), days | 5 (3−7) |
| Fever duration, median (IQR), days | 6 (4−7) |
| Clinical features at presentation | |
| Exanthema/rash | 38 (68) |
| Conjunctivitis | 26 (46) |
| Oral mucosa inflammation | 17 (30) |
| Lymphadenopathies | 15 (27) |
| Headache or irritability | 27 (48) |
| Prostration | 30 (54) |
| Gastrointestinal symptomsa | 40 (71) |
| Abdominal pain | 30 (54) |
| Diarrhea | 30 (54) |
| Vomiting | 21 (38) |
| Feed refusal | 16 (29) |
| Dehydration | 12 (21) |
| Lymphadenopathy | 5 (9) |
| Enteritis (suggested radiographic image) | 3 (5) |
| Shock symptomsa | 33 (59) |
| Tachycardia (age specific) | 24 (43) |
| Hypotension (age specific) | 17 (30) |
| Prolonged capillary refill (>s) | 16 (29) |
| Cutaneous pallor or mottled skin | 11 (20) |
| Cold feed or hand | 12 (21) |
| Low urine output (< 2 mL/kg/h) | 13 (23) |
| Metabolic acidosis | 12 (21) |
| Increased lactate | 8 (14) |
| Acute renal injury | 5 (9) |
| Liver injury | 2 (4) |
| Oxygen therapy needed | 15 (27) |
| Respiratory symptomsa | 26 (46) |
| Cough | 16 (29) |
| Tachypnea | 14 (25) |
| Worst respiratory frequency, median (IQR), rpm | 45 (41−45) |
| Low SpO2 (< 92%) | 10 (18) |
| Thoracic pain | 2 (4) |
| Dyspnea | 9 (16) |
| Chest retraction | 4 (7) |
| Elevated cardiac dysfunction markers | 39 (75) |
| Abnormal EKG | 6 (11) |
| Coagulopathy | 29 (53) |
| Image findings | No. (%) |
|---|---|
| Chest radiographies | 48 (86) |
| Altered | 25/48 (52.1) |
| Diffuse interstitial infiltrate, bilateral | 11/48 (22.9) |
| Pleural effusion | 7/48 (14.6) |
| Consolidation | 3/48 (6.3) |
| Interstitial infiltrate, localized | 3/48 (6.3) |
| Atelectasis | 1/48 (2.1) |
| Chest computed tomography | 35 (63) |
| Chest computed tomography with ground-glass opacities | 11 (31) |
| Echocardiogram altered, No. | 34 |
| Mild pericardial effusion/pericarditis | 17/34 (50) |
| Abnormal left ventricular function | 9/34 (26.5) |
| Coronary artery dilatation/ectasy | 9/34 (26.5) |
IQR, interquartile range; MIS-C, multisystem inflammatory syndrome in children; SpO2, pulse oximeter oxygen saturation; EKG, electrocardiogram.
Altered chest radiographs were found in 25 of 48 patients (52%), mainly bilateral diffuse interstitial infiltrate (23%), while ground glass opacities were observed in 11 of 35 patients (31%) who underwent chest computerized tomography. The echocardiogram was altered in 34 patients (61%), mainly showing mild pericardial effusion (50%), although 27% of the patients had left ventricular dysfunction and also 27% had signs of coronary dilatation (Table 2).
Laboratory findingsOf the 56 patients included, 18 had C-reactive protein (CRP) below 10.0 mg/dL, two patients had borderline results between 1.0 and 3.0 mg/dL and only three had CRP below 1.0 mg/dL. From these five patients with CRP below 3.0 mg/dL, three of them had altered ESR (>20 mm) and the two remaining had altered fibrinogen (>300 mg/dL). Thus, all patients had altered inflammatory markers, in accordance with the CDC criteria.
Cardiac dysfunction markers were increased in more than 75% of the patients tested. Troponin I was higher than 0.1 ng/mL in 53% of patients; Pro-B type natriuretic peptide (proBNP) was elevated in more than 75% of patients, and above 1000 pg/mL in 42% of those; and 38% had creatinine kinase myocardial band (CK-MB) values higher than 25 U/L. d-Dimer was high (>500 ng/mL) in 80% of 41 patients tested; fibrinogen and activated prothrombin time (aPTT) were also elevated in more than half of the patients. Blood count was performed in all patients: anemia, leukocytosis, lymphopenia, and thrombocytopenia were observed in more than 30% of the patients, but thrombocytosis in only 14%. Lactate dehydrogenase (LDH) was high (>295 U/L) in 78% of 50 patients; the results of liver function tests were slightly abnormal in some patients, but hypoalbuminemia (<3 g/dL) was present in 62%. Renal function was normal in all patients (Table 3).
Laboratory findings of pediatric patients with MIS-C.
| Laboratory test, number tested/total (No.) | Worst values |
|---|---|
| Median (IRQ) or No. (%) | |
| Inflammatory markers | |
| C-reactive protein (mg/dL), No. 55/56 | 15.0 (9.1−36.6) |
| Erythrocyte sedimentation rate, No. 42/56 | 92.5 (49.3−120.0) |
| Procalcitonin (ng/mL), No. 14/56 | 1.0 (0.4−2.5) |
| Ferritin (ng/mL), No. 46/56 | 464.5 (187.0−852.7) |
| Interleukin-6 (pg/mL), No. 3/56 | 194.3 (101.4−452.7) |
| Cardiac disfunction markers | |
| Troponin (ng/mL), No. 45/56 | 0.2 (0.1−8.7) |
| ProBNP (pg/mL), No. 36/56 | 5,818.0 (603.8−12,748.0) |
| Creatinine kinase, total (U/L), No. 38/56 | 76.4 (43.5−136.3) |
| Creatinine kinase, myocardial band (U/L), No. 26/56 | 14.2 (1.5−28.8) |
| Coagulation markers | |
| Activated prothrombin time (PT), No.18/56 | 17.2 (15.9−19.0) |
| Activated Partial Thromboplastin Time (aPTT), No. 16/56 | 31.0 (26.8−35.6) |
| d-Dimer (ng/mL), No. 46/56 | 3,270.5 (1213.8−5175.0) |
| Fibrinogen (mg/dL), No. 10/56 | 506.0 (387.3−665.0) |
| Biochemical tests | |
| Lactate (mg/dL), No. 8/56 | 2.3 (1.8−2.7) |
| Bicarbonate (mEq/L), No. 12/56 | 15.5 (13.0−17.1) |
| Alanine aminotransferase (U/L), No. 54/56 | 51.4 (32.0−82.1) |
| Aspartate aminotransferase (U/L), No. 54/56 | 51.1 (29.5−73.4) |
| Lactate dehydrogenase (U/L), No. 49/56 | 506.0 (318.0−671.0) |
| Gamma-glutamyltransferase (U/L), No. 2/56 | 192.0 (182.0−202.0) |
| Albumin (g/dL), No. 38/56 | 2.7 (2.2−3.0) |
| Urea (mg/dL), No. 55/56 | 30.0 (22.4−44.7) |
| Creatinine (mg/dL), No. 55/56 | 0.5 (0.3−0.7) |
| Altered hematological tests | |
| Anemia | 24 (43%) |
| Hemoglobin (g/dL), No. 24 | 8.8 (7.8−10.0) |
| Hematocrit (%), No. 24 | 25.9 (23.3−30.1) |
| Leukocytosis (>15,000/µL) | 25 (44.6%) |
| Total leukocyte count (×1000/µL), No.25 | 23,900.0 (18,350.0−26,000.0) |
| Lymphopenia | 26 (46.4%) |
| Lymphocyte count (cells/mm3) No. 26 | 796 (479−1048) |
| Thrombocytosis (>450,000/µL), | 10 (17.9%) |
| Platelet count (×1000/µL), No. 10 | 668,000 (535,500−845,250) |
| Thrombocytopenia (<150,000/µL), | 23 (41.1%) |
| Platelet count (×1000/µL), No. 23 | 94,000 (65,750−102,500) |
MIS-C, multisystem inflammatory syndrome in children; IQR, interquartile range; proBNP, Pro-B type natriuretic peptide, NA, non-available.
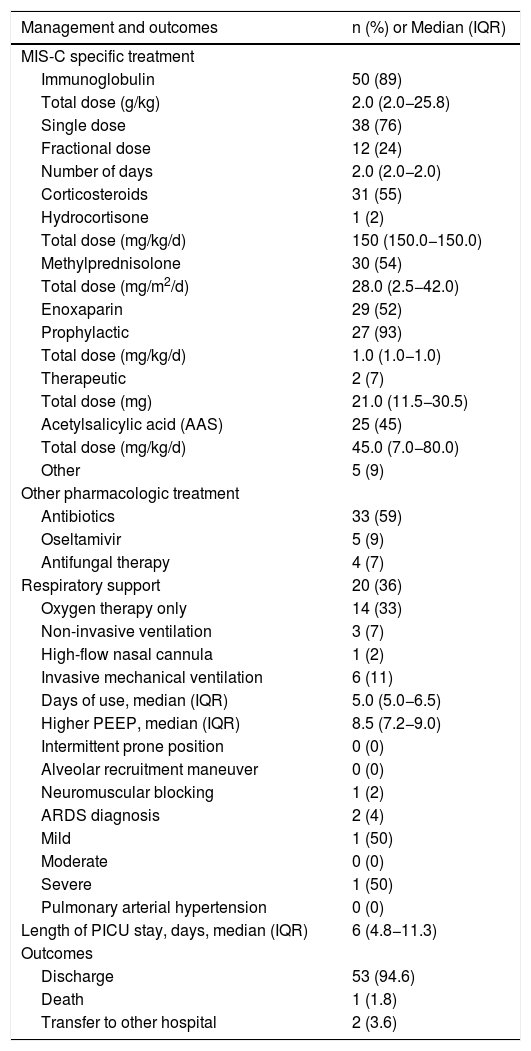
A total of 89% of patients received intravenous immunoglobulin (IVIG), and more than 50% received corticosteroids and enoxaparin (prophylactic or therapeutic). Acetylsalicylic acid (AAS) was administered in 45%, antibiotics in 59%, but oseltamivir and antifungal therapy were used in less than 10% of patients. Of the 20 patients (36%) who needed respiratory support, only 11% required invasive mechanical ventilation (IMV), of whom only two patients had a more severe course, with acute respiratory distress syndrome (ARDS), but no with pulmonary arterial hypertension. The median duration of IMV was five days and the highest median positive end expiratory pressure (PEEP) was 8.5. No special ventilatory strategies, such as intermittent prone position or alveolar recruitment maneuver, were necessary. There was only one death in this cohort (1.8%; Table 4).
Management and clinical outcomes of pediatric patients with MIS-C (n = 56).
| Management and outcomes | n (%) or Median (IQR) |
|---|---|
| MIS-C specific treatment | |
| Immunoglobulin | 50 (89) |
| Total dose (g/kg) | 2.0 (2.0−25.8) |
| Single dose | 38 (76) |
| Fractional dose | 12 (24) |
| Number of days | 2.0 (2.0−2.0) |
| Corticosteroids | 31 (55) |
| Hydrocortisone | 1 (2) |
| Total dose (mg/kg/d) | 150 (150.0−150.0) |
| Methylprednisolone | 30 (54) |
| Total dose (mg/m2/d) | 28.0 (2.5−42.0) |
| Enoxaparin | 29 (52) |
| Prophylactic | 27 (93) |
| Total dose (mg/kg/d) | 1.0 (1.0−1.0) |
| Therapeutic | 2 (7) |
| Total dose (mg) | 21.0 (11.5−30.5) |
| Acetylsalicylic acid (AAS) | 25 (45) |
| Total dose (mg/kg/d) | 45.0 (7.0−80.0) |
| Other | 5 (9) |
| Other pharmacologic treatment | |
| Antibiotics | 33 (59) |
| Oseltamivir | 5 (9) |
| Antifungal therapy | 4 (7) |
| Respiratory support | 20 (36) |
| Oxygen therapy only | 14 (33) |
| Non-invasive ventilation | 3 (7) |
| High-flow nasal cannula | 1 (2) |
| Invasive mechanical ventilation | 6 (11) |
| Days of use, median (IQR) | 5.0 (5.0−6.5) |
| Higher PEEP, median (IQR) | 8.5 (7.2−9.0) |
| Intermittent prone position | 0 (0) |
| Alveolar recruitment maneuver | 0 (0) |
| Neuromuscular blocking | 1 (2) |
| ARDS diagnosis | 2 (4) |
| Mild | 1 (50) |
| Moderate | 0 (0) |
| Severe | 1 (50) |
| Pulmonary arterial hypertension | 0 (0) |
| Length of PICU stay, days, median (IQR) | 6 (4.8−11.3) |
| Outcomes | |
| Discharge | 53 (94.6) |
| Death | 1 (1.8) |
| Transfer to other hospital | 2 (3.6) |
ARDS, acute respiratory distress syndrome; IQR, interquartile range; PEEP, positive end expiratory pressure; PICU, pediatric intensive care unit; NA, non-available.
Most patients (75%) had the complete KD or incomplete Kawasaki phenotype, of which boys (76%) were most frequent. In the group with acute cardiac dysfunction, the median age was higher than in the other groups and non-whites represented 88% of patients. Comorbidities were not frequent in any phenotype. More than 70% of patients of all groups were treated with IGIV. The median PICU length of stay was six days, except for the only macrophagic activation syndrome patient (22 days; Table S1 – Supplementary files).
DiscussionThis is the first Brazilian multicenter study that described a cohort of patients with MIS-C only. The clinical features of the present patients are very similar to those previously reported in literature: most patients did not meet the complete criteria for KD; the median age was higher than that described for patients with COVID-19 only, but slightly lower than that described for MIS-C,3–7,10 and greater than that observed in classic KD.17
In this study, males were much more frequent than females (2:1), and this predominance was even higher in the Kawasaki-like phenotype (3:1). This high proportion of males was also observed in other multicenter studies of MIS-C in the United Kingdom and the United States,5,6 and in critically ill children with COVID-19 alone.14 Biological differences (genetic and epigenetic) between males and females may affect the immune response to SARS-CoV-2 infection, as has already been described in KD.18,19 Most of the present patients were non-white, similar to what has been described in Europe and America, although this may reflect the general ethnical distribution of Brazil.5,6,11 As to the frequency of comorbidities, the present results were also similar to other studies of MIS-C,5,6 but the predominance of chronic neurological diseases has not been previously reported. Almost half of the present patients had a history of a contact with someone with COVID-19 and 55% had SARS-CoV-2 infection either detected by RT-PCR, serological test or both, corroborating a causal association with the new coronavirus. Fourteen patients tested positive for SARS-CoV-2 by RT-PCR, which generally reflects an acute phase of the infection, although the virus or its fragments may be detected for longer periods in some patients and could be responsible for these results, outside the classical period of positivity of the acute phase of COVID-19. However, it is still unclear how long after the acute phase of SARS-CoV-2 infection it takes for the first signs and symptoms of MIS-C to appear, or even whether this condition may still occur during the acute phase of COVID-19. The high frequency of gastrointestinal symptoms, the low prevalence of severe respiratory failure, and the lower degree of mucosal involvement have already been described, which characterizes MIS-C as a distinct entity, unrelated to classic KD.5,6
As expected in proinflammatory states and as part of the criteria for defining MIS-C, all children in the present cohort had increased inflammatory markers, such as CRP, ESR, and ferritin, which is also reported in other MIS-C cohorts in England, United States, France, Switzerland, and Italy.3–5,7,10 The proinflammatory effect of SARS-CoV-2 infection has been reported in adults with severe COVID-19, with whom MIS-C shares some characteristics, such as dysregulated innate immune response and cytokine storm.6,10,20–22 In this study, IL-6 was measured in only three patients, due to its low availability and high cost. IL-6 is a pro-inflammatory cytokine that has been studied for many years as a sepsis biomarker, and its concentrations appear to correlate positively with sepsis severity.23 IL-6 appears to play an important role in severe adult patients with COVID-19, in whom the compassionate use of tocilizumab for pharmacologic inhibition of IL-6 has been described.24 The levels of d-dimer, fibrinogen, and aPTT were also elevated in most patients, reflecting a state of coagulopathy associated with hyperinflammation, which has been described in severe COVID-19 and MIS-C.10,20,24,25
Lymphopenia and thrombocytopenia — which have been described as distinct hematological features of MIS-C and are not present in classic KD — was observed in 30%–45% of patients in the present cohort. Anemia and hypoalbuminemia were also common in our patients, but these findings are characteristics of KD.2 Although MIS-C is already recognized as a distinct clinical entity, the overlap of many features of other multisystem inflammatory syndrome phenotypes is frequently reported. A striking difference between MIS-C and severe COVID-19 in adults is the absence of renal impairment,10 this is described in other cohorts of MIS-C and we also found normal renal function in our patients. Cardiac dysfunction markers were altered in most of our patients. Normal troponin levels of <0.1 ng/mL have been described in healthy children under 1 year of age.26 Pro-BNP cut-off points of 502 ng/L, 456 ng/L, 445 ng/L, and.355 ng/L have been suggested for detecting cardiac failure in children aged 1–3 years, 4−7 years, and 8–14 years, respectively.27 In the present cohort, troponin levels were highly increased in at least half of the patients who were tested, while pro-BNP was highly increased in more than three quarters of the tested patients. Echocardiographic findings similar to those of KD were also common in this cohort. These findings are compatible with myocardial dysfunction and inflammation consistently described in MIS-C reports.3–6 Its mechanism is not fully understood, but it may be related to microvascular damage, stress cardiomyopathy (Takotsubo syndrome) and systemic inflammatory response syndrome.28
Although there is no current evidence for the best management of MIS-C, guidelines from different organizations recommend treatment based on the clinical phenotype.6,29 In the present cohort, the clinical syndromes at admission were mostly complete or incomplete KD, followed by acute cardiac dysfunction, and in a lesser extent toxic shock syndrome and macrophage activation syndrome. Accordingly, IGIV was used in the vast majority of patients, and corticosteroids and AAS in approximately half. The role of AAS in the treatment of KD is well established and has been used in all patients with a phenotype similar to complete KD. Although there is no evidence of the benefit of corticosteroid for pediatric patients with severe COVID-19 and/or MIS-C, the use of corticosteroid in MIS-C patients has been described in many studies in an attempt to reduce the hyperinflammatory response.5,6,10 In addition, the recent multicenter CoDEX trial showed that, in severe adult patients with COVID-19, the use of dexamethasone increased the ventilator-free-days in the first 28 days by two-thirds.30 Enoxaparin was also used in more than half of the present patients, since d-dimer was highly elevated in most of them. Actually, coagulopathy and thrombosis are important features in severe COVID-19 in adults.28 Children with MIS-C are at risk for thrombotic complications of multiple causes, including hypercoagulable state, possible endothelial injury, immobilization stasis, ventricular dysfunction, and coronary artery aneurysm. For these reasons, antiplatelet and/or anticoagulation treatment is recommended, based on coagulation tests and clinical presentation.28
Although no information on associated bacterial or fungal infection and/or co-detection of other viruses is available, the described use of antibiotics, antifungal therapy and oseltamivir in this study can be justified by these possibilities. Empirical antibiotic therapy in hospitalized patients with MIS-C is recommended, as symptoms overlap with severe bacterial sepsis.28 The protocol of the Brazilian Ministry of Heath for severe respiratory acute syndrome recommends the use of oseltamivir until an influenza infection can be excluded.
Although children diagnosed with MIS-C often require intensive care treatment, studies have shown good outcomes and a low mortality rate.5,10 This study found similar outcomes, with only one death, but with a longer length of PICU stay than that reported in other cohorts.3,7,10,11 The comparison of demographic and clinical features among the different phenotypes showed that they were relatively equivalent.
The present study has some limitations. Brazil is a country with great racial miscegenation, which may limit the generalization of the present results, especially considering that individual genetic variations has been reported to affect the severity and phenotypes of SARS-CoV-2 infection. In addition, although this is a multicenter study, the number of patients is small, making comparison among phenotypes difficult. However, the present number is similar to other cohorts of MIS-C patients previously described.3–7,10 Additionally, only 55% of the patients had confirmed laboratorial SARS-CoV-2 infection and not all patients had inflammatory and cardiac dysfunction markers checked. Also, information about the history of previous symptoms that suggest another previous viral disease or when it has occurred was not available.
Nevertheless, this article provides relevant information on the clinical features and outcomes of the novel described MIS-C in hospitalized children and adolescents in Brazil, which the authors believe to be an important contribution to the understanding of SARS-CoV-2 infection in children and its short-term consequences. It is important to reinforce the need for long-term multidisciplinary follow-up, since it is still not known whether these patients will have chronic cardiac impairment or other sequelae.
FundingThis study was supported by grants from the following Brazilian research promotion agencies: Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq (National Council for Scientific and Technological Development – CNPq), Process 401597/2020-2; and Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro FAPERJ (Carlos Chagas Filho Foundation for Research Support of the State of Rio de Janeiro – FAPERJ), Process E-26/010.000160/2020, grant 2020/0996.
Conflict of interestThe authors declare no conflicts of interest.