To investigate the performance of 27 children with phenylketonuria (PKU) in tests of Executive Functions (EF) and Social Cognition (SC), and their associations with metabolic control inferred by phenylalanine (Phe) levels.
MethodsThe PKU group was dichotomized according to baseline Phe-levels into; "classical PKU"(n = 14), with Phe-levels above 1200 μmol/L (> 20 mg/dL); and "mild PKU" (n = 13) with Phe-between 360 and 1200 μmol/L (6–20 mg/dL). The neuropsychological assessment focused on the EF and SC subtests of the NEPSY-II battery and intellectual performance. Children were compared to age-matched healthy participants.
ResultsParticipants with PKU presented significantly lower Intellectual Quotient (IQ) compared to controls (p = 0.001). Regarding EF analysis adjusted by age and IQ, significant differences between groups were observed only in the executive attention subtests (p = 0.029). The SC set of variables was significantly different between groups (p = 0.003), as in the affective recognition task (p < 0.001). In the PKU group, the relative variation of Phe-achieved 32.1 ± 21.0%. Relative Phe-variation was correlated only with measures of Working Memory (p < 0.001), Verbal Fluency (p = 0.004), Inhibitory Control (p = 0.035) and Theory of Mind (p = 0.003).
ConclusionsPhonological Verbal Fluency, Working Memory, Inhibitory Control, and Theory of Mind were shown to be most vulnerable when there is non-ideal metabolic control. Variations in the level of Phe-may have a selective negative effect on Executive Functions and Social Cognition, but not on intellectual performance.
Phenylketonuria (PKU) is an autosomal recessive disorder caused by phenylalanine hydroxylase (PAH) gene mutations,1 causing partial or total deficiency of the PAH enzyme, which converts phenylalanine (Phe) into tyrosine (Tyr). The accumulation of Phe-results in tissue toxicity, especially in the central nervous system.2 The clinical phenotypes are usually classified as classical PKU, characterized by plasmatic Phe-levels above 1200 μmol/L defined by tandem mass spectrometry (20 mg/dL by fluorometric immunoassay); and mild PKU, when Phe-levels remain between 360 and 1200 μmol/L (6–20 mg/dL).2 According to European guidelines,3 patients with levels above 360 μmol/L (6 mg/dL) should be treated to achieve a therapeutic goal of Phe-between 120 and 360 μmol/L (2–6 mg/dL).1,2 At the time of this study, the treatment for newborns adopted by Brazilian institutions is recommended only for cases with levels above 10 mg/dL or 600 μmol/L.4
To ensure a better prognosis, children with PKU must be submitted to a low-protein diet to control plasmatic Phe-levels as early as possible.2 The time from birth to 12 years of age is a crucial period to maintain stable Phe-levels to preserve neurodevelopment.3 Moreover, diet therapy should be lifelong, since variations in Phe-may result in neurological damage, leading to intellectual deficiency, hyperactivity, and deficits in Executive Functions (EF) and Social Cognition (SC)2,5. EF skills enable individuals to engage in goal-directed activities and SC is important to regulate behavior through understanding other people's intentions or feelings.6 There is evidence that an increase of 100 μmol/L in Phe-level predicts a decrease of 1.3 to 6 points in intelligence quotient (IQ).3,7 Therefore, dietary adherence is important to provide metabolic stability and guarantee cognitive development.2,3
Although nutritional treatment has been shown to improve cognitive and behavioral outcomes, neuropsychological dysfunctions have been reported even in individuals submitted to early intervention. The SC and EF domains seem particularly vulnerable to long-term variations of Phe-levels.2 Inhibitory control8, working memory9, cognitive flexibility10 and verbal fluency11 impairments are usual and prevail in children with delayed diagnosis without intellectual impairment.12 Therefore, the extent of the effects of fluctuations in, and high concentrations of, Phe-on neurodevelopment must be better understood.
This study was designed to analyze the effects of individual variability in Phe-levels on cognitive outcomes in Brazilian PKU children. The authors hypothesized that poor metabolic control, demonstrated by elevated mean Phe-levels in follow-up, would be associated with worse cognitive outcomes. Therefore, the authors aimed to 1) compare intellectual, EF and SC performance of PKU and typically developing children; 2) investigate associations between performance and Phe-fluctuations in the PKU group. Results may provide insights into the PKU neurocognitive phenotype that could be useful for clinical approaches. The interest in FE and CS skills was due to their associations with psychosocial outcomes, such as school performance and peer interaction in the short and mid-terms, and on quality of life in the long term.
MethodsThis cross-sectional exploratory study undertook a quantitative analysis of the neuropsychological performance of children with PKU compared with age-matched healthy controls, approved by the local Ethics Committee (UNIFESP/protocol 2.879.610). All legal guardians provided informed consent and children provided assent for participation.
ParticipantsTwenty-seven children of both sexes diagnosed with Phenylketonuria (PKU group) and 28 children with neurotypical development (control group) aged between 7 and 15 years participated in the study. Children with PKU were recruited from those in clinical follow-up at Instituto Jô Clemente (IJC), a referral center for newborn screening. Control participants were recruited from public and private schools.
The PKU group was dichotomized according to baseline Phe-levels (i.e. before diet therapy) into "classical PKU" (n = 14), with levels above 1200 μmol/L (> 20 mg/dL); and "mild PKU" (n = 13) with levels between 360 and 1200 μmol/L (6–20 mg/dL). Exclusion criteria for the PKU group were lack of regular diet therapy and attendance at follow-up; for controls were reports of learning disabilities, and neurological or psychiatric disorders.
All PKU participants presented Phe-levels above 4 mg/dL until the 15th day of life. They were diagnosed in basic health units or maternity hospitals between the 3rd-5th day of life. Soon after diagnosis, children were referred for clinical follow-up with a multidisciplinary team. After a new assessment of Phe-levels to confirm PKU, nutritional management of Phe-ingestion comprising diet therapy and regular monitoring of levels was started. The frequency of return visits varies according to age: a) weekly for children between 0 and 6 months of age; b) fortnightly for 6–12 months; c) monthly for 1–2 years; d) quarterly for those over 2 years old. Follow-up includes psychological evaluations to monitor neurocognitive development.
Clinical characteristicsClinical (PKU phenotype, Phe-levels), socio-demographic (age, sex, maternal education), and anthropometric (body mass index) variables of PKU participants were collected from medical records. Phe-levels were obtained by fluorometric method following Brazilian guidelines4 and performed by an expert. All met the necessary requirement to minimize the Phe-fluctuation as much as possible in the follow-up sampling: blood collection always at the same time in the morning; a 3-hour fasting period before the measurement of Phe, and the child's last meal before testing consisted of a Phe-source food, with the amount of amino acid prescribed by the dietician. The examination is part of the follow-up routine to monitor Phe-levels.
Four measures of the Phe-levels of each participant were considered. The first was collected after birth and defined the PKU phenotype. The classical PKU phenotype presented mean Phe-levels of 31.2 ± 10.5 mg/dL and the mild PKU had a mean of 15.1 ± 3.81 mg/dL. The other measures comprised the last three blood samples obtained before inclusion in the study. Individual fluctuations in Phe-levels were defined as a percentage variation from the last blood sample, i.e. relative variation (%variation Phe = SD*100 = X/M). The study member that executed the neuropsychological assessment was blinded to the PKU phenotype and to historical Phe-levels.
Neuropsychological investigationThe neuropsychological assessment included intellectual, EF and SC measures. For PKU children it was combined with regular clinical visits and performed individually in appropriate rooms at IJC. Controls were assessed in noise-controlled rooms at their schools, without prejudice to routine activities.
Intellectual performanceThe validated Wechsler Abbreviated Scale of Intelligence (WASI)13 was used to assess intellectual performance. The WASI provides three main index scores: full intelligence quotient (IQ), verbal (VIQ), and performance IQ (PIQ).
Neuropsychological assessmentThe developmental neuropsychological battery NEPSY-II adapted to Portuguese was used to investigate EF and SC.14 The battery covers ages 3–16 years and is widely used in studies with neurodevelopmental disorders. Reports with clinical samples in the manual indicate high-reliability coefficients (rs = 0.85–0.87). Raw scores were used for analyses.
The subtests of the Attention and Executive Functions domain were used: Animal Sorting (AS; assesses cognitive flexibility); Auditory Attention (AA; selective attention); Response Set (RS; auditory attention and vigilance); and Inhibition (IN; inhibitory control and shifting). The protocol also included the digit span and Corsi block-tapping tests of working memory.15 Scores were the extent of digits (maximum 8) or cubes (maximum 7) repeated backward. The number of animals starting with given letters (F,A,S) generated in 60 s was the measures of semantic and verbal fluency, respectively.15
The validated Behavior Rating Inventory of Executive Functions (BRIEF) completed the EF assessment.16 The main caretaker answered 86 questions about behaviors observed at home as "never", "sometimes", and "often". A threshold above 65 indicates a clinical profile.
Social Cognition (SC) was assessed using the subtests of Affect Recognition (AR) and Theory of Mind (ToM). The AR assesses children's ability to identify basic emotions in faces. The ToM assesses abilities to interpret beliefs, intentions, thoughts, and emotions from another person's perspective, among others.
Emotional and behavioral assessmentThe Child Behavior Checklist – CBCL adapted for Brazilian children17 answered by caretakers was used for behavioral assessment. It comprises 113 questions about everyday behavior with responses being "not true", "sometimes/somewhat true" and "very often true", proving quantitative indices related to internalizing (somatic problems, anxiety, depression) and externalizing (rule-breaking behavior, aggressive behavior) profiles. A t-score > 65 indicates a clinically meaningful threshold.
Statistical analysisStatistical analysis was performed using the JAMOVI software (version 2.3.22). Descriptive statistics presented categorical data as absolute and relative frequencies, and numerical data as mean ± standard deviation, median, and confidence intervals when applicable. Generalized Linear Models (GzLM) were used to investigate differences in IQ between groups and identify the impact of variations among Phe-levels on dependent variables. Multivariate analysis of covariance (MANCOVA) was used to identify the effects of groups in a set of dependent variables adjusted by age and IQ. Finally, univariate, and multivariate linear regression analyses were performed to identify the clinical variables with the greatest impact on discrimination between groups. The level of significance adopted was p < 0.05.
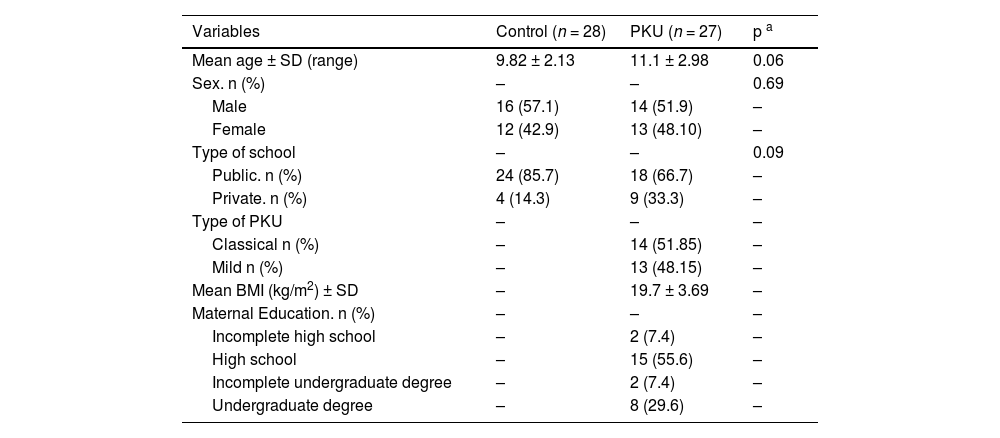
ResultsClinical characteristics were similar in both groups, although the control group had more patients below 10 years of age (Table 1). Therefore, age was added as a covariate to avoid any confounding results. Since no significant difference was observed in IQ between the classical and mild phenotypes (p = 0.415), the PKU patients were not dichotomized into subgroups for the comparative analysis with the control group.
Sociodemographic, anthropometric and clinical characteristics.
| Variables | Control (n = 28) | PKU (n = 27) | p a |
|---|---|---|---|
| Mean age ± SD (range) | 9.82 ± 2.13 | 11.1 ± 2.98 | 0.06 |
| Sex. n (%) | – | – | 0.69 |
| Male | 16 (57.1) | 14 (51.9) | – |
| Female | 12 (42.9) | 13 (48.10) | – |
| Type of school | – | – | 0.09 |
| Public. n (%) | 24 (85.7) | 18 (66.7) | – |
| Private. n (%) | 4 (14.3) | 9 (33.3) | – |
| Type of PKU | – | – | – |
| Classical n (%) | – | 14 (51.85) | – |
| Mild n (%) | – | 13 (48.15) | – |
| Mean BMI (kg/m2) ± SD | – | 19.7 ± 3.69 | – |
| Maternal Education. n (%) | – | – | – |
| Incomplete high school | – | 2 (7.4) | – |
| High school | – | 15 (55.6) | – |
| Incomplete undergraduate degree | – | 2 (7.4) | – |
| Undergraduate degree | – | 8 (29.6) | – |
Participants with PKU presented significantly lower IQ scores compared to controls (89.8 ± 10.8 versus 109.0 ± 9.5, p = 0.001, respectively), with both VIQ (p < 0.001) and PIQ (p < 0.001) different among groups. Thus, IQ was also treated as a covariate in the MANCOVA for EF and SC. The aim was to understand whether the EF and SC performance would be more explained by intelligence level rather than PKU presence.
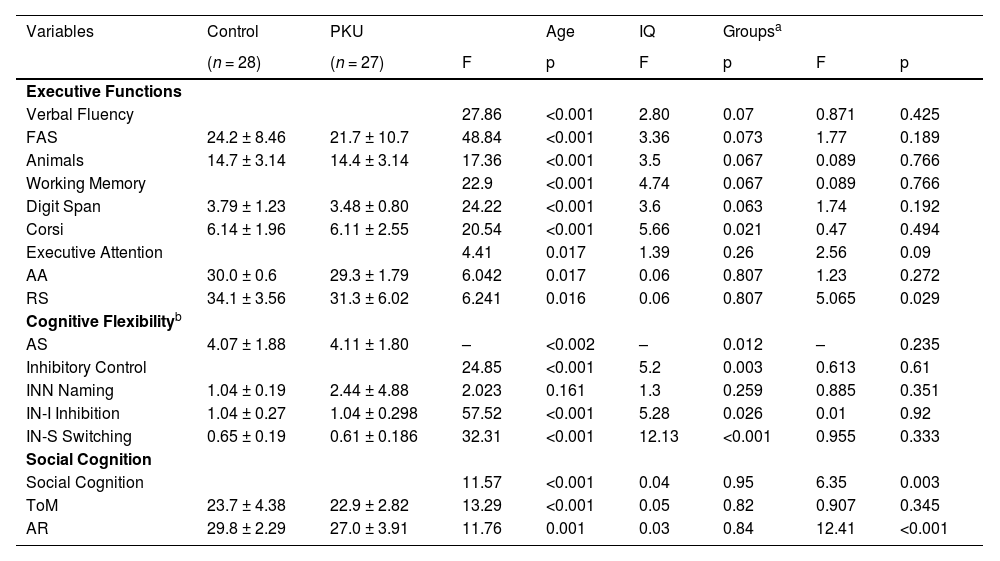
In the EF analysis adjusted by age and IQ, significant differences between the control and PKU groups were observed only in the set of executive attention subtests (RS) of NEPSY-II (p = 0.029). The authors found significant associations of age with the following EF tasks: executive attention (p = 0.017); verbal fluency (p = 0.001), phonemic fluency (p ≤ 0.001), and semantic verbal fluency tasks (p < 0.001); working memory (p = 0.001); cognitive flexibility (p < 0.0021); and inhibitory control (p < 0.001). Yet, IQ was only associated with working memory (p = 0.013), cognitive flexibility (p = 0.012), and inhibitory control (p = 0.003) outcomes.
The SC set of variables (Table 2) was significantly different between groups (p = 0.003), adjusted by age and IQ. Groups differed significantly in the affective recognition (AR) task (p < 0.001), but not in ToM (p = 0.345). Age was associated with the overall SC scores (p = 0.001) and with both AR (p = 0.001) and ToM (p = 0.001) tasks. No significant associations were observed between IQ and the SC set of variables (p = 0.955).
Comparison between groups covariated for age and IQ on EF and CS.
| Variables | Control | PKU | Age | IQ | Groupsa | |||
|---|---|---|---|---|---|---|---|---|
| (n = 28) | (n = 27) | F | p | F | p | F | p | |
| Executive Functions | ||||||||
| Verbal Fluency | 27.86 | <0.001 | 2.80 | 0.07 | 0.871 | 0.425 | ||
| FAS | 24.2 ± 8.46 | 21.7 ± 10.7 | 48.84 | <0.001 | 3.36 | 0.073 | 1.77 | 0.189 |
| Animals | 14.7 ± 3.14 | 14.4 ± 3.14 | 17.36 | <0.001 | 3.5 | 0.067 | 0.089 | 0.766 |
| Working Memory | 22.9 | <0.001 | 4.74 | 0.067 | 0.089 | 0.766 | ||
| Digit Span | 3.79 ± 1.23 | 3.48 ± 0.80 | 24.22 | <0.001 | 3.6 | 0.063 | 1.74 | 0.192 |
| Corsi | 6.14 ± 1.96 | 6.11 ± 2.55 | 20.54 | <0.001 | 5.66 | 0.021 | 0.47 | 0.494 |
| Executive Attention | 4.41 | 0.017 | 1.39 | 0.26 | 2.56 | 0.09 | ||
| AA | 30.0 ± 0.6 | 29.3 ± 1.79 | 6.042 | 0.017 | 0.06 | 0.807 | 1.23 | 0.272 |
| RS | 34.1 ± 3.56 | 31.3 ± 6.02 | 6.241 | 0.016 | 0.06 | 0.807 | 5.065 | 0.029 |
| Cognitive Flexibilityb | ||||||||
| AS | 4.07 ± 1.88 | 4.11 ± 1.80 | – | <0.002 | – | 0.012 | – | 0.235 |
| Inhibitory Control | 24.85 | <0.001 | 5.2 | 0.003 | 0.613 | 0.61 | ||
| INN Naming | 1.04 ± 0.19 | 2.44 ± 4.88 | 2.023 | 0.161 | 1.3 | 0.259 | 0.885 | 0.351 |
| IN-I Inhibition | 1.04 ± 0.27 | 1.04 ± 0.298 | 57.52 | <0.001 | 5.28 | 0.026 | 0.01 | 0.92 |
| IN-S Switching | 0.65 ± 0.19 | 0.61 ± 0.186 | 32.31 | <0.001 | 12.13 | <0.001 | 0.955 | 0.333 |
| Social Cognition | ||||||||
| Social Cognition | 11.57 | <0.001 | 0.04 | 0.95 | 6.35 | 0.003 | ||
| ToM | 23.7 ± 4.38 | 22.9 ± 2.82 | 13.29 | <0.001 | 0.05 | 0.82 | 0.907 | 0.345 |
| AR | 29.8 ± 2.29 | 27.0 ± 3.91 | 11.76 | 0.001 | 0.03 | 0.84 | 12.41 | <0.001 |
Generalized Linear Models (GzLM).
In the PKU group, the mean time variation among the collection of the three blood samples was 4.07 ± 1.67 months, maintaining stable fluctuations (p = 0.225). The Phe-levels were 7.56 ± 3.10 mg/dL at the first collection, 7.49 ± 3.59 mg/dL at the second, and 7.76 ± 3.90 mg/dL at the last. Individual mean variations of Phe-levels were 2.21 ± 1.50 mg/dL, which corresponded to a variation of 32.1 ± 21.0% between collections. To ensure the detection of relative oscillations in Phe-levels in PKU patients, the percentage change between the three time periods was used to define individual variation.
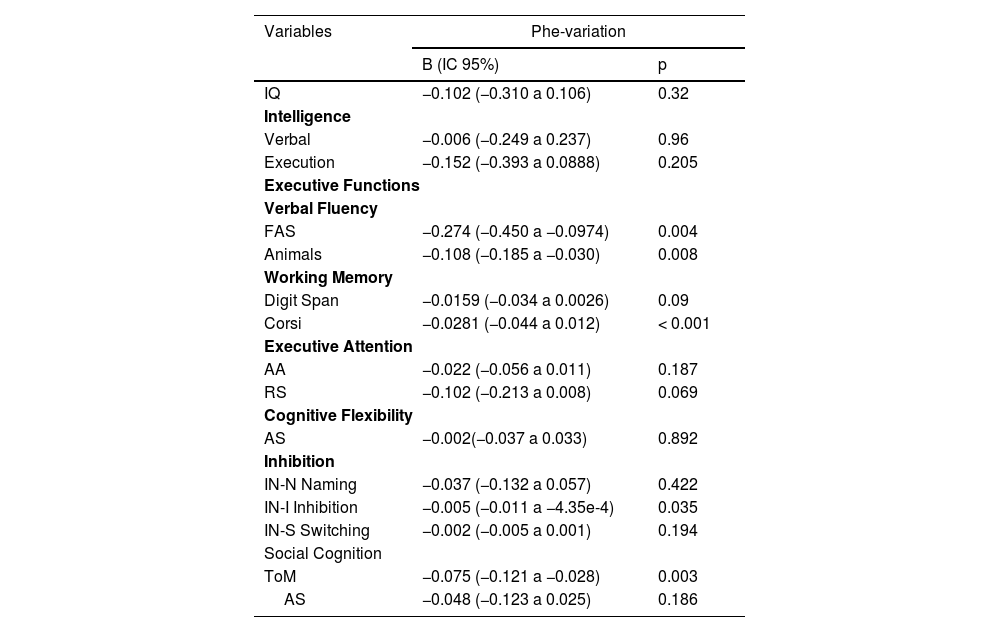
Relative Phe-variation was not correlated with total IQ (p = 0.32, Table 3), or with verbal (p = 0.96) or performance (p = 0.205) quotients. Among EF tests, relative Phe-variation was significantly correlated only with Corsi task scores and working memory index (p = 0.001). Relative Phe-variation was associated with phonemic verbal fluency scores (p = 0.004) and inhibitory control index (p = 0.035). Considering SC variables, it was associated with ToM test scores (p = 0.003).
Influence of Phe-variation on observed outcomes.
Generalized Linear Models (GzLM).
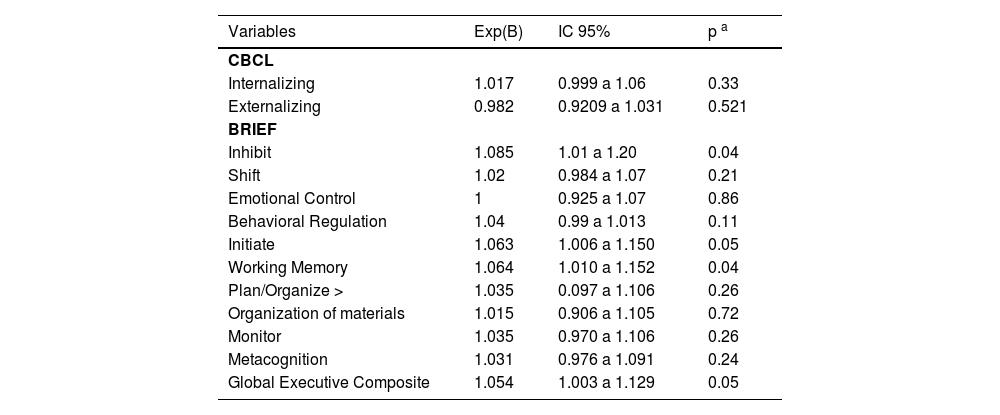
Finally, the authors investigated whether the percentage change in Phe-would be a determinant for behavioral problems and EF, as reported by the main caretakers. Relative Phe-variation was not correlated with CBCL scores related to internalizing profile (p = 0.33) nor to externalizing profile (p = 0.52). In the BRIEF, relative Phe-variation was associated with inhibition (p = 0.04), initiative (p = 0.05) and working memory (p = 0.04) scores, and overall executive functioning index (p = 0.05, Table 4).
Phe-variation as a determinant for CBCL and BRIEF scales.b
| Variables | Exp(B) | IC 95% | p a |
|---|---|---|---|
| CBCL | |||
| Internalizing | 1.017 | 0.999 a 1.06 | 0.33 |
| Externalizing | 0.982 | 0.9209 a 1.031 | 0.521 |
| BRIEF | |||
| Inhibit | 1.085 | 1.01 a 1.20 | 0.04 |
| Shift | 1.02 | 0.984 a 1.07 | 0.21 |
| Emotional Control | 1 | 0.925 a 1.07 | 0.86 |
| Behavioral Regulation | 1.04 | 0.99 a 1.013 | 0.11 |
| Initiate | 1.063 | 1.006 a 1.150 | 0.05 |
| Working Memory | 1.064 | 1.010 a 1.152 | 0.04 |
| Plan/Organize > | 1.035 | 0.097 a 1.106 | 0.26 |
| Organization of materials | 1.015 | 0.906 a 1.105 | 0.72 |
| Monitor | 1.035 | 0.970 a 1.106 | 0.26 |
| Metacognition | 1.031 | 0.976 a 1.091 | 0.24 |
| Global Executive Composite | 1.054 | 1.003 a 1.129 | 0.05 |
The authors also investigated which anthropometric and sociodemographic variables might be predictors of Phe-variation. The authors considered the following variables: age, sex, body mass index (BMI), maternal educational level, type of PKU, and the concentration of blood Phe-present in the first clinical examination.
In the first analysis (Table S1), the authors assessed the association of each variable separately (univariate). The authors considered the variables presenting p < 0.15 to compose the multivariate analysis and determine potential predictors. The authors identified age (p = 0.001) and BMI (p = 0.112) as possible predictors of Phe-variations. However, multivariate analysis showed that only age was an independent predictor for Phe-variations (p = 0.002).
DiscussionThis study investigated EF and SC in a sample of children with classic and mild PKU undergoing diet therapy. Results suggest that poor metabolic control, demonstrated by uncontrolled Phe-levels, has a selective negative effect on cognitive domains, but not on intellectual performance. Phonemic verbal fluency, visuospatial working memory, EF and ToM skills were shown to be the most susceptible to worse metabolic control. There was no evidence of associations with internalizing or externalizing behavioral problems.
An initial topic of discussion concerns the association between metabolic control and intelligence. Children with PKU showed significantly lower IQ scores compared to controls, even in average ranges. These findings are similar to Gassió et al.18 results with a similar number of participants (n = 37) and mean Phe-values (444 ± 145μmol/L; i.e. 7.33 ± 2.39 mg/dL), revealing IQs levels (100 ± 11.0) lower than controls (p = 0.001).
In the present study, Phe-variations did not predict IQ, which differs from previous findings as described by Hood et al.19 in a sample of 47 children followed from birth to 10 years of age, where variations in Phe-levels were negatively correlated with IQs, although IQ was still average. Such results may be explained by the greater variability of Phe-in their sample (202.4 ± 84.4μmol; 3.34 ± 1.39 mg/dL). Conversely, lower IQs have been associated with plasma concentration values obtained at the time of assessment and not with fluctuations in levels.20 Differences in IQ may be less significant if blood Phe-concentrations are maintained between 120 and 360 μmol/L (2–6 mg/dL), in line with therapeutic goals recommended in PKU treatment.2
Regarding EF, the present results showed that greater relative Phe-variation was associated with worse results in verbal fluency, visual-spatial working memory, and inhibitory control. Associations between dietary control (i.e. fluctuations in Phe-levels) and EF were reported in previous studies.21 It is worth mentioning the specific difficulties shown by PKU children in inhibiting motor responses according to task changes. In the BRIEF, caretakers also reported behavioral signs of inhibitory control problems, besides low initiative in social contexts and working memory difficulties. The minor impact of dietary control observed on cognitive flexibility and verbal working memory, as well as discrepancies between verbal and non-verbal working memory, needs to be better understood in future studies.
Regarding SC, the present results suggest that children with PKU also show difficulties in recognizing emotions, but not infer people's mental states (ToM). An unexpected result was that metabolic control turned out to be a predictor only for ToM. A possible explanation is that emotional recognition is associated with the overall neurocognitive phenotypic characteristics of PKU and, therefore, less influenced by variations in Phe-levels. Jahja et al.5 identified broader changes in socio-cognitive functioning which were significantly associated with Phe-variations, especially among children over 12 years old. The authors concluded that Phe-control during childhood and early adolescence is important for better social functioning in adulthood. However, they did not differentiate performance in emotion recognition and ToM.
An important issue to discuss is that age was determinant for the performance of children with PKU in all components of EF and CS, although not in IQ, according to previous evidence.5-11 It can be hypothesized that, due to the later maturation of the prefrontal cortex, self-regulatory executive skills have contributed to better performance among older participants.
The authors found that with each additional year of age, the variation in plasma Phe-concentration levels dropped by four percentage points. Differently from previous knowledge,22 the younger the child, the higher the levels of Phe, suggesting a worse treatment adherence related to age. Due to inherent limitations of the sample size and natural Phe-fluctuations during the day, this data should be interpreted with caution. Future research is necessary to explore this controversy due to particular socio-economical and cultural issues in Latin America compared to European and American reports.23,24
The present study has limitations to be mentioned. Because of its cross-sectional design, the determination of individual Phe-fluctuations based on the relative variation of four measures does not necessarily reflect lifelong Phe-control. Additionally, although neuropsychological assessment was integrated into the patient's follow-up routine, the authors acknowledge that Phe-levels have a natural variation during the day and depend on the time of fasting and type of food ingested. Therefore, conclusions about associations between Phe-fluctuation and neurocognitive development can be limited and demand future studies.
In spite of limitations, the authors believe that the present study innovates by the emphasis done on SC, beyond EF, and associations with metabolic control due to dietary treatment. Furthermore, findings contribute to the literature on PKU, strengthening the important role of dietary control, especially in respect of the development of inhibitory control, visual-spatial working memory, and phonemic verbal fluency. Due to the extended maturation in frontal areas, such functions begin their development in early childhood and are associated with fluid intelligence, which is less influenced by academic learning.
Due to the lack of information on PKU in developing countries, it could be interesting to investigate Brazilian recommended levels of Phe-and practices to compare these data with international recommendations. Greater attention must be paid to the presence of higher Phe-concentrations, as this may be an important early sign of lack of adherence to diet, which may worsen in adolescence.25 Since diet-therapy is the only treatment option that does not seem to be sufficient to preserve neurodevelopment, new therapies have been developed, such as the use of sapropterin dihydrochloride in responsive cases or enzyme replacement therapy with pegvaliase in patients older than 16 years.26 Advantages of these new therapies include the possibility to increase the amount of food containing Phe-or even in some cases responsive to sapropterin dihydrochloride to skip totally the restricted diet, reaching normal protein diet with recommended Phe-levels improving patient nutrition. The disadvantages: this therapy is not universally successful (less than half of patients are responsive) and seems to work more in milder phenotypes; so some patients need to remain with combined treatment of diet and sapropterin, which increases therapeutic cost.27 Instead, the enzyme replacement therapy with pegvaliase corrects the biochemical defect with normalization of Phe-levels under unrestricted diet, but allergic reaction, food neophobia and high cost are the disadvantages.28 There are more options on the horizon coming out such as gene therapy29 and a new oral drug PTC923 with the promise of being even more effective to more patients.30
This study indicated that children with PKU may present selective impairments in executive functioning and social cognition, regardless of age and IQ. The higher the Phe-levels, the more important the executive function impairment seems to be. The association of neuropsychological screening with consultations for metabolic control can contribute to the early identification of cognitive deficits and the design of interventions in clinical settings.
The authors thank the Associação Fundo de Incentivo à Pesquisa (AFIP) and Instituto Jô Clemente