To meta-analyze health-related quality of life in pediatric patients with chronic kidney disease in comparison to healthy patients according to the dimensions of the PedsQL instrument.
Sources of dataA systematic review was performed with meta-analysis for the mean difference in each of the health-related quality of life dimensions. The authors searched for ten scientific databases including PubMed, Scopus, SciELO, Science Direct, ProQuest, Google Scholar. Reproducibility by the Kappa index was evaluated, and Dersimonian and Laird's tests, RI coefficient, Begg statistic, Forest Plot, and sensitivity analysis were carried out.
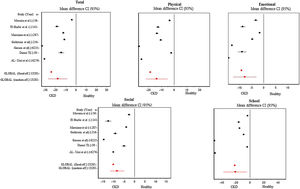
Summary of the findings17 investigations were included in the qualitative synthesis and 7 in the quantitative synthesis with a population of 1214 of both healthy and sick pediatric patients with 3−5 chronic kidney disease stages. The health-related quality of life in pediatric chronic kidney disease patients presented lower scores in all the evaluated dimensions: in the physical dimension the difference is of 13.6 points, in the emotional dimension 7.8, in the social dimension 8.2, in the school dimension 20.8, with the total difference being 17.7 points.
ConclusionThe findings of this study show that pediatric patients have lower health-related quality of life in all the evaluated dimensions, with the “school” dimension being the most affected and the “emotional” one the least. In this sense, we suggest monitoring the health-related quality of life of pediatric patients with chronic kidney disease so that interventions can be oriented to strengthen the affected dimensions, including adjustments to daily life and prevention of complications related to the disease.
Chronic kidney disease (CKD) has steadily increased its frequency and prevalence.1,2 Globally, the epidemiological panorama shows that CKD is considered the sixth fastest-growing cause of death, affecting around 10% of the population in industrialized countries.3,4 It is estimated that around 850 million people – between 6% and 12% of the world population5,6 - suffer from chronic kidney disease, of which at least 2.4 million die every year.4 The global affliction for chronic renal failure (CRF) has increased from 10 to15% each year. More than one million patients approximately are on peritoneal dialysis treatment and close to 1.6 million people are on hemodialysis therapy,7 a rate that is most likely to double in the next 10 years, thus, increasing healthcare costs to more than 1.1 trillion USD.8,9
In concern to the pediatric population worldwide, incidences of ESRD has been stable over the past 30 years.10,11 In 2008, the incidence of children and adolescents with ESRD was around 9per million persons regarding the age-related population (pmarp), with the United States at a higher median of 15.5 pmarp.12 In France, the most recent incidence was 8.7 pmarp in 2015. The average European pediatric ESRD incidence was 5.5 cases pmarp in children aged 0–14 years and 8.3 pmarp in children aged 0–19 years.13
The presence of CKD during childhood and adolescence is rare, but when present it displays significant effects in life development Since it involves other social life dynamics (playing, studying, sexual identity), it generates medical dependency and an uncertainty in receiving a transplant, within their active and working life, as well as with the future itself.14–16 Each of the stated changes has an impact on the quality of life of pediatric patients.
The World Health Organization (WHO) defines Quality of life as the perception that an individual has of his or her place in existence, in the context of culture, and the value system in which he or she lives, as well as on their expectations, norms, and concerns.17,18 In general, Quality of life (QL) is a multidimensional concept based on the subjective perception of the patient,19 in which "non-clinical" factors, such as family, friends, religious beliefs, work, income, and other life circumstances are also involved.20 However, the interest in evaluating the results of the interventions, and in knowing the influence of the health conditions to healthcare services, as well as in recognizing important activities that affect the individual’s well-being, led to this exploration of Health-related quality of life (HRQL). The HRQL focuses on life aspects influenced specifically by personal health and health activities,21 Bulpit22 defines it as the subjective degree of well-being attributed to or associated with the lack of illness or its symptoms, the psychological condition and the activities that a person wants to do.
The quality of life-related to health is considered important in the clinical setting because it allows doctors to predict mortality, therapeutic adherence, hospitalizations, and reflects the preferences of patients through their physical and psychological health, social relations and the environment.23,24 Likewise, it has become a common measure for health professionals during therapeutic monitoring and personal care customization.
In congruence with the abovementioned and given the importance that the study of HRQL has gained along with the impact that chronic kidney disease generates in pediatric patients, European countries as well as North and South American ones have conducted studies with results that differ in the scope of the most affected HRQL. A study carried out in Brazil25 shows that the most affected measurement is the emotional dimension, while in Bulgaria26 a study carried out with 55 pediatricians showed that the social dimension is affected as much as school activities, and its physical dimension was as reported by Marciano et al.27
Moreover, it is important to note that the evaluation of HRQL in pediatric patients has become relevant in recent times because it begins to be studied as a holistic, multidimensional and comprehensive concept, where the children's perception of their well-being is taken into account.28,29 Currently, there are generic and specific instruments to measure the HRQL indicator, the generic being designed to measure this both in the healthy population and patients with different pathologies, considering the common interests of all these groups. Its importance comes from the capacity it holds to compare healthy and sick persons, as well as groups with different pathologies; while the second seeks to capture particular items, such as specific symptoms of a disease or adverse effects of the treatments, they cannot be used outside the context for which they were designed.30
The instruments for measuring HRQL has been built from multiple aspects of the patient's life and situations, which are grouped around various dimensions: physical functioning, psychological well-being, emotional state, pain, social functioning, the general perception of health, and other factors that include sexual functioning, the degree of satisfaction with life, the impact on work productivity and everyday activities. The scores or summary scores range between 0–100 points and they are used for the analysis and extraction of conclusions concerning the variables of the HRQL indicator.31 This indicator is valued globally or multi-dimensionally, indicating that the higher the score is, the higher the quality of life related to the patient’s health.
Based on this background, this study was designed with the aim of meta-analyzing health-related quality of life in pediatric CKD patients. The study consisted of the comparison with healthy patients according to the dimensions of the Pediatric Quality of Life (QL) Inventory 4.0 instrument, as well as with systematically reviewing the demographic and clinical characteristics of pediatric patients with chronic kidney disease and their health-related quality of life profile.
MethodsType of studyA systematic review with meta-analysis. The studied population consisted of pediatric patients with chronic kidney disease in different stages (1−5) undergoing hemodialysis, peritoneal dialysis and kidney transplantation, compared with healthy pediatric patients.
Instrument for measuring health-related quality of lifeTo develop the meta-analysis, we used the instrument “Pediatric Quality of Life (QL) Inventory 4.0”32 which measures the health-related quality of life (HRQL) in healthy children and adolescents, as well as in those with acute and chronic health conditions. It is a generic instrument made up of 23 items with a Likert-type response scale (0 = never and 4 = almost always) and four dimensions: physical functioning (8 items), emotional functioning (5 items), social functioning (5 items) and functioning at school (5 items). The scale has four versions according to the age of the child: 2−4 years (preschool), 5–7 years (young children), 8–12 years (children) and 13–18 years (adolescents).32
Good psychometric properties with internal consistency were shown in previous studies33,34 for the full scale (alpha=0.88 children). Similarly, in Colombia, Vélez et al.35 reported a Cronbach's alpha of 0.96.
Research protocolAccording to the criteria of the PRISMA guide (Preferred reporting items for systematic reviews and meta-analyses),36 a systematic review was carried out with meta-analysis following the order of the stages of identification, screening, eligibility, and inclusion.
Search and identification of studiesIn order to search for the information, the keywords adolescent, children, Quality of life, kidney disease, renal failure, dialysis were selected, in combination with the Boolean connector AND. With the selected keywords, different search algorithms were built in six databases: PubMed, Scopus, Scielo, Science Direct, ProQuest, Google Scholar. Some of the algorithms used in PubMed were [((QUALITY OF LIFE [Title / Abstract]) AND ADOLESCENT [Title / Abstract]) AND KIDNEY DISEASE [Title / Abstract]; in Scopus [TITLE-ABS-KEY ("quality of life" AND adolescent AND dialysis); in Scielo [(ab: (QUALITY OF LIFE)) AND (ab: (ADOLESCENT)) AND (ab: (RENAL FAILURE))]; for Science Direct [Title, abstract, keywords: "quality of life" AND children AND "kidney disease"]; in ProQuest [ab (quality of life) AND ab (young) AND ab (dialysis); and finally, in Google Scholar [all in title: Young quality of life kidney disease].
To guarantee the sensitivity and exhaustiveness of the search, no time limits were included in the publications.
Inclusion or screening criteriaThe identified studies were screened by reading the abstracts and the inclusion criteria considered were: 1) articles written in English, Spanish or Portuguese, 2) articles from original studies that have made observations or interventions directly to patients, 3) quality of life in patients with chronic kidney disease or on hemodialysis as the main subject of the study, 4) studies other than case reports.
Exclusion criteriaOnce the screening stage was completed, the exclusion criteria were applied by reading the articles chosen in the previous stage, excluding 1) articles that did not include the population of interest (children and/or adolescents), 2) articles that do not specify the methodology for evaluating the quality of life, 3) the evaluation of Quality of Life was studied from the caregiver's perspective, and 4) articles that were not found in the full text version despite searching for it on multiple sources. Finally, to carry out the meta-analysis, articles that 1) used scales other than PedsQoL were excluded, 2) did not report the score on a scale from 0 to 100, and 3) did not include the mean, standard deviation or standard error in each dimension.
Collection of informationThe articles identified in the databases were exported to the Zotero program version 4.0.28.7 for the storage of references and the elimination of duplicates.
Subsequently, a matrix was prepared in Excel 2016, which contained the variables such as year of publication, authors, country, number of patients included, follow-up time, age (mean, SD), stage of kidney disease, type of treatment (dialysis, hemodialysis), disease progression time, comorbidities, the name of the scale used to assess the HRQL, the domains and the score in each domain (mean, SD). Likewise, we separated the scores reported in each dimension of the scale for patients with CKD and those who are healthy.
Reproducibility and methodological quality assessmentThe identification and selection of the articles were carried out by two researchers independently; the inconsistencies were resolved through a third person as the reviewer. The reproducibility in the information extraction was evaluated with the Kappa index of 1.0 for the qualitative variables and an intra-class correlation coefficient of 1.0 for the quantitative variables. The evaluation of the methodological quality of the studies included in the meta-analysis was made with the STROBE guide (Strengthening the Reporting of Observational Studies in Epidemiology).37
Analysis of the informationThe descriptions of the studies were done with frequency measurements. A comparison between the HRQL dimensions of pediatric CKD patients and healthy individuals was done with a standardized mean difference meta-analysis. Heterogeneity was assessed with the Galbraith graph, the Dersimonian and Laird's test (Q statistic), and the RI coefficient (proportion of total variance due to the variance between studies). Publication bias was assessed with the Funnel Plot and the Begg statistic. The global results are shown with the Forest Plot for each dimension of the HRQL. Finally, a sensitivity analysis was done to assess the effect of each study on the overall result. The analyses were done in Epidat version 3.1.
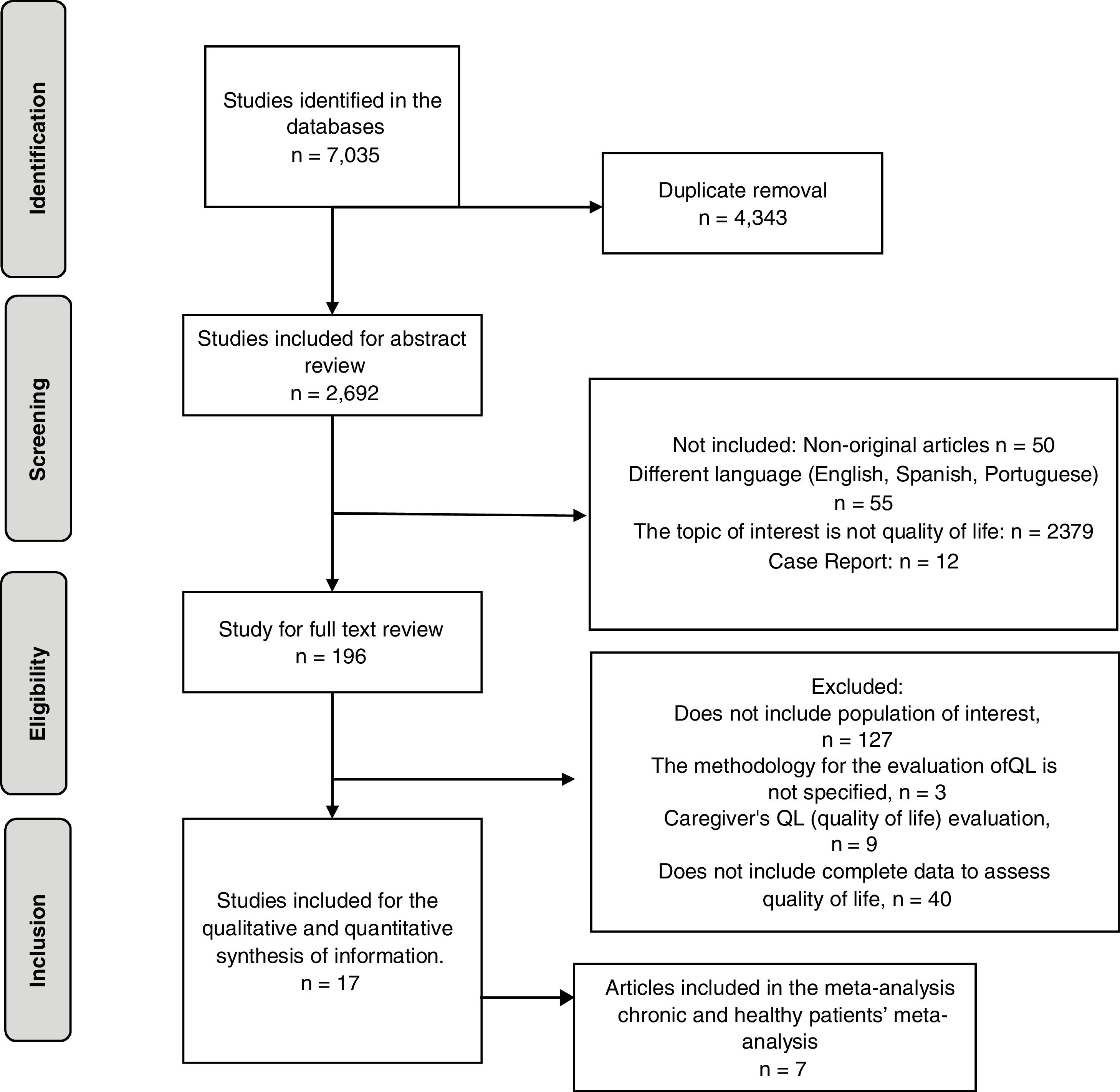
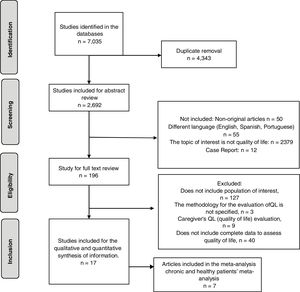
Results7035 articles were identified, of which 4343 were eliminated for being duplicates and 2675 for not meeting the inclusion and exclusion criteria, leaving 17 articles for the systematic review and 7 for the meta-analysis (Fig. 1).
The evaluation of HRQL in pediatric patients with CKD has been a topic of interest in different countries of the world, highlighting publications from Brazil,25,27 Bulgaria,26 Argentina,38 Egypt,39 Singapore,40 United States,41–44 Thailand,45 England,46 France,47 Stockholm,48 Greece,49 Australia,50 and Holland.51
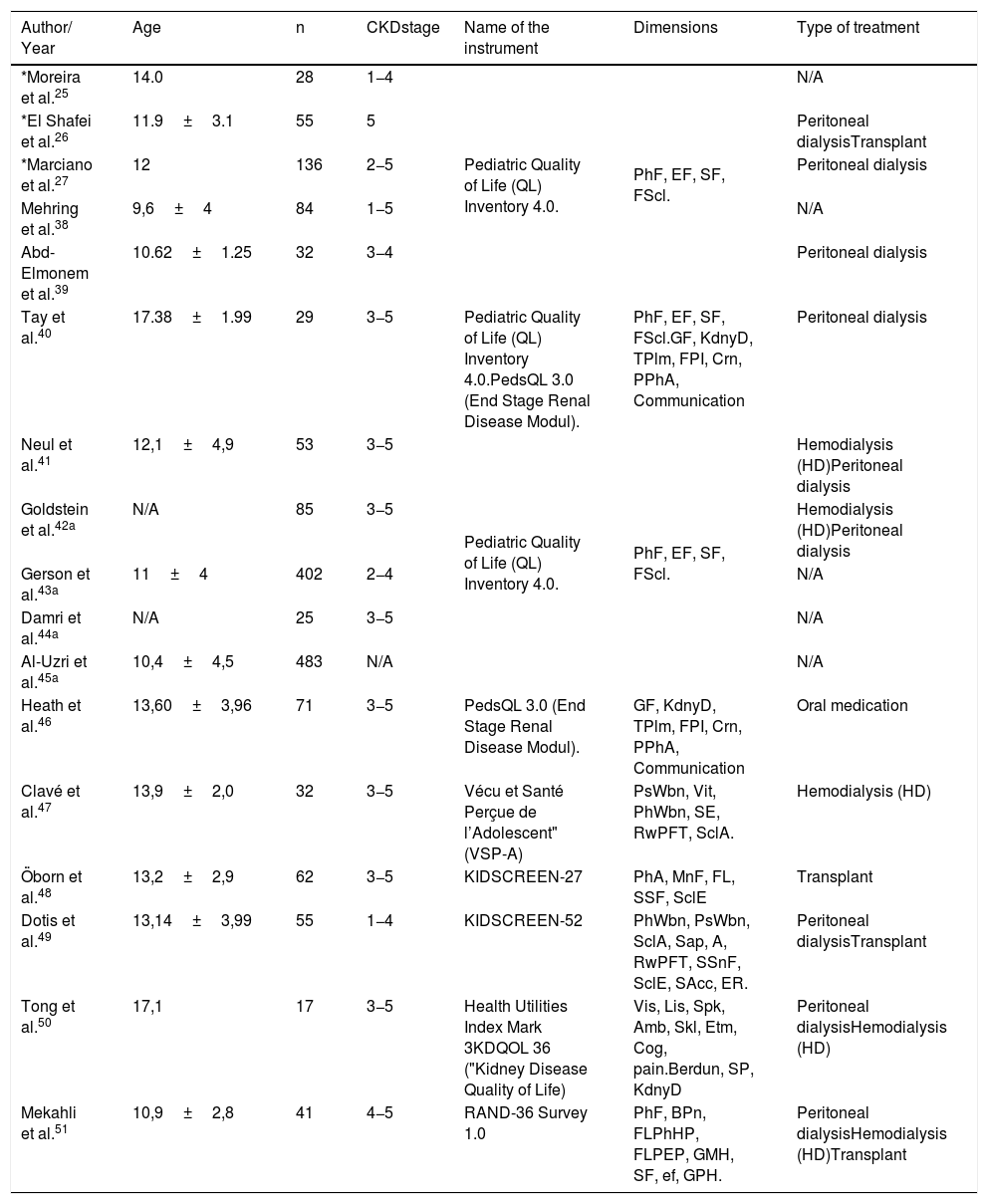
In the same sense, the HRQL assessment was carried out with a wide variety of instruments, generic and specific, and with dimensions that account for the impact of CKD on social relationships, physical health, emotional health, family interaction, interaction with peers, autonomy, and the school environment. Instruments used include PedsQoL 3.0–4.0, Kidscreen, VSP-A, Health Utilities Index, KDQOL, and RAND-36; the most used was PedsQoL 4.0. Specifically, the studies that applied the PedsQoL25–27,42–45 are included in this meta-analysis reported data within the dimensions of the instrument in the healthy population and in individuals with CKD, also including patients between 14 years25 and 10.4±4.5 years,45 59% were men, with chronic kidney disease from stage I to V, and with several patients included that ranged from 28 in the study by Moreira et al.25 to 483 in the research by Al-Uzri et al.45 We highlight that the study carried out by Goldstein et al.,42 does not report in its data differentiated information regarding the age (mean) and gender of the participants; Damri et al.,44 for age and Al-Uzri et al.45 for the stage of disease (Table 1).
Characteristics of the studies included in the Qualitative Synthesis and the Meta-analysis.
| Author/ Year | Age | n | CKDstage | Name of the instrument | Dimensions | Type of treatment |
|---|---|---|---|---|---|---|
| *Moreira et al.25 | 14.0 | 28 | 1−4 | Pediatric Quality of Life (QL) Inventory 4.0. | PhF, EF, SF, FScl. | N/A |
| *El Shafei et al.26 | 11.9±3.1 | 55 | 5 | Peritoneal dialysisTransplant | ||
| *Marciano et al.27 | 12 | 136 | 2−5 | Peritoneal dialysis | ||
| Mehring et al.38 | 9,6±4 | 84 | 1−5 | N/A | ||
| Abd-Elmonem et al.39 | 10.62±1.25 | 32 | 3−4 | Peritoneal dialysis | ||
| Tay et al.40 | 17.38±1.99 | 29 | 3−5 | Pediatric Quality of Life (QL) Inventory 4.0.PedsQL 3.0 (End Stage Renal Disease Modul). | PhF, EF, SF, FScl.GF, KdnyD, TPlm, FPI, Crn, PPhA, Communication | Peritoneal dialysis |
| Neul et al.41 | 12,1±4,9 | 53 | 3−5 | Pediatric Quality of Life (QL) Inventory 4.0. | PhF, EF, SF, FScl. | Hemodialysis (HD)Peritoneal dialysis |
| Goldstein et al.42a | N/A | 85 | 3−5 | Hemodialysis (HD)Peritoneal dialysis | ||
| Gerson et al.43a | 11±4 | 402 | 2−4 | N/A | ||
| Damri et al.44a | N/A | 25 | 3−5 | N/A | ||
| Al-Uzri et al.45a | 10,4±4,5 | 483 | N/A | N/A | ||
| Heath et al.46 | 13,60±3,96 | 71 | 3−5 | PedsQL 3.0 (End Stage Renal Disease Modul). | GF, KdnyD, TPlm, FPI, Crn, PPhA, Communication | Oral medication |
| Clavé et al.47 | 13,9±2,0 | 32 | 3−5 | Vécu et Santé Perçue de l’Adolescent" (VSP-A) | PsWbn, Vit, PhWbn, SE, RwPFT, SclA. | Hemodialysis (HD) |
| Öborn et al.48 | 13,2±2,9 | 62 | 3−5 | KIDSCREEN-27 | PhA, MnF, FL, SSF, SclE | Transplant |
| Dotis et al.49 | 13,14±3,99 | 55 | 1−4 | KIDSCREEN-52 | PhWbn, PsWbn, SclA, Sap, A, RwPFT, SSnF, SclE, SAcc, ER. | Peritoneal dialysisTransplant |
| Tong et al.50 | 17,1 | 17 | 3−5 | Health Utilities Index Mark 3KDQOL 36 ("Kidney Disease Quality of Life) | Vis, Lis, Spk, Amb, Skl, Etm, Cog, pain.Berdun, SP, KdnyD | Peritoneal dialysisHemodialysis (HD) |
| Mekahli et al.51 | 10,9±2,8 | 41 | 4−5 | RAND-36 Survey 1.0 | PhF, BPn, FLPhHP, FLPEP, GMH, SF, ef, GPH. | Peritoneal dialysisHemodialysis (HD)Transplant |
A, Autonomy; Amb, ambulation; BPn, body pain; Cog, cognition; Crn, concern; EF, Emotional functioning; ef, energy / fatigue; ER, Economic resources; Etm, emotion; FL, family life and free time; FLPEP, functional limitations due to personal or emotional problems; FLPhHP, functional limitations due to physical health problems; FPI, Family and peer interaction; FScl, Functioning at School; GF, General fatigue; GMH, general mental health; GPH, general perceptions of health; KdnyD, About my kidney disease; Lis, listen; M, Mood; MnF, mood and feelings; PhA, physical activity and health; PhF, Physical functioning; PhWbn, physical well-being; PPhA, Perceived physical appearance; PsWbn, psychological well-being; RwPFT, relationship with parents, friends and teachers; SAcc, Social acceptance; Sap, Self-appreciation; SclA, school activities and leisure; SclE, school environment; SE, self-esteem; SF, Social functioning; Skl, skill; SP, symptoms / problems; Spk, speak; SSnF, social support and friends; TPblm, treatment problems; Vis, Vision; Vit, vitality; N/A, data not reported in the articles.
Studies26,27,39–42,46–51 indicate the treatment modality of the pediatric population with CKD, including peritoneal dialysis, hemodialysis and kidney transplantation, only the study by Heath et al.46 included a population that received oral medication.
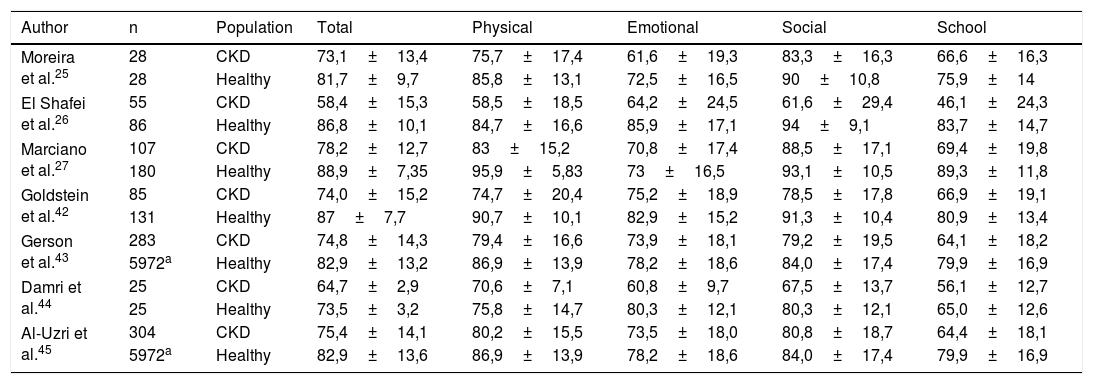
The investigations reported populations with healthy individuals and a population with chronic kidney disease (CKD). The physical dimension of HRQL in patients with CKD showed values between 56.2±4.6 and 80.2±15.5, for the emotional dimension 60.8±9.7 and 75.2±18.9, for the social 56.2±4.6 and 88.5±17.1, with the school dimension being the one that reported lower values 44.4±3.1 and 66.9±19.1.
For the healthy population, the physical dimension revealed data 75.8±14.7 and 95.9±5.83, emotional 72.5±16.5 and 85.9±17.1, social 80.3±12.1 and 94±9.1, and school 65.0±12.6 and 89.3±11.8 (Table 2).
Health-related quality of life profile according to PedsQL 4.0 dimensions in the pediatric population with chronic kidney disease and healthy controls.
| Author | n | Population | Total | Physical | Emotional | Social | School |
|---|---|---|---|---|---|---|---|
| Moreira et al.25 | 28 | CKD | 73,1±13,4 | 75,7±17,4 | 61,6±19,3 | 83,3±16,3 | 66,6±16,3 |
| 28 | Healthy | 81,7±9,7 | 85,8±13,1 | 72,5±16,5 | 90±10,8 | 75,9±14 | |
| El Shafei et al.26 | 55 | CKD | 58,4±15,3 | 58,5±18,5 | 64,2±24,5 | 61,6±29,4 | 46,1±24,3 |
| 86 | Healthy | 86,8±10,1 | 84,7±16,6 | 85,9±17,1 | 94±9,1 | 83,7±14,7 | |
| Marciano et al.27 | 107 | CKD | 78,2±12,7 | 83±15,2 | 70,8±17,4 | 88,5±17,1 | 69,4±19,8 |
| 180 | Healthy | 88,9±7,35 | 95,9±5,83 | 73±16,5 | 93,1±10,5 | 89,3±11,8 | |
| Goldstein et al.42 | 85 | CKD | 74,0±15,2 | 74,7±20,4 | 75,2±18,9 | 78,5±17,8 | 66,9±19,1 |
| 131 | Healthy | 87±7,7 | 90,7±10,1 | 82,9±15,2 | 91,3±10,4 | 80,9±13,4 | |
| Gerson et al.43 | 283 | CKD | 74,8±14,3 | 79,4±16,6 | 73,9±18,1 | 79,2±19,5 | 64,1±18,2 |
| 5972a | Healthy | 82,9±13,2 | 86,9±13,9 | 78,2±18,6 | 84,0±17,4 | 79,9±16,9 | |
| Damri et al.44 | 25 | CKD | 64,7±2,9 | 70,6±7,1 | 60,8±9,7 | 67,5±13,7 | 56,1±12,7 |
| 25 | Healthy | 73,5±3,2 | 75,8±14,7 | 80,3±12,1 | 80,3±12,1 | 65,0±12,6 | |
| Al-Uzri et al.45 | 304 | CKD | 75,4±14,1 | 80,2±15,5 | 73,5±18,0 | 80,8±18,7 | 64,4±18,1 |
| 5972a | Healthy | 82,9±13,6 | 86,9±13,9 | 78,2±18,6 | 84,0±17,4 | 79,9±16,9 |
Note: Studies 25, 26, 27, 42 and 44) report primary source data for healthy populations.
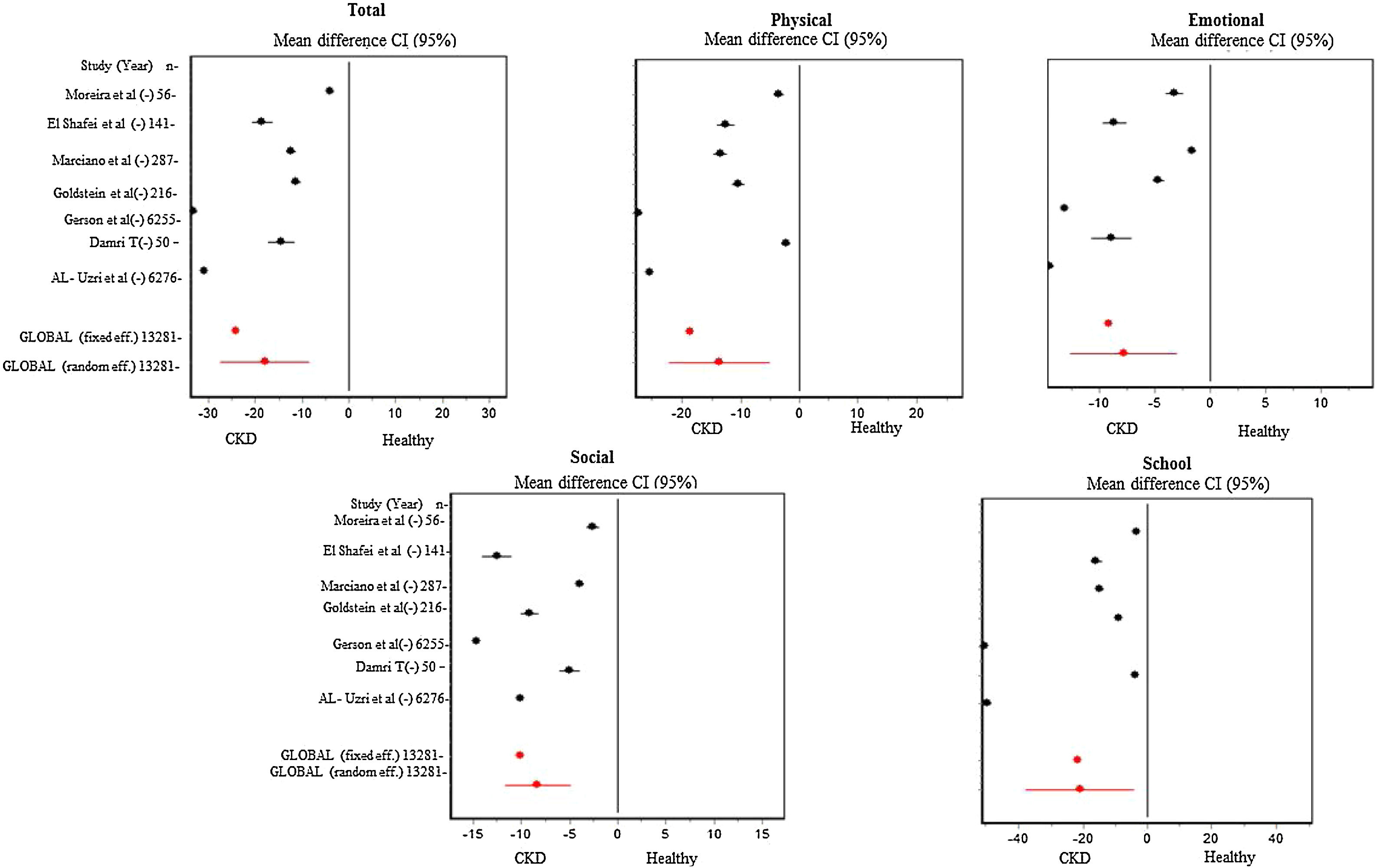
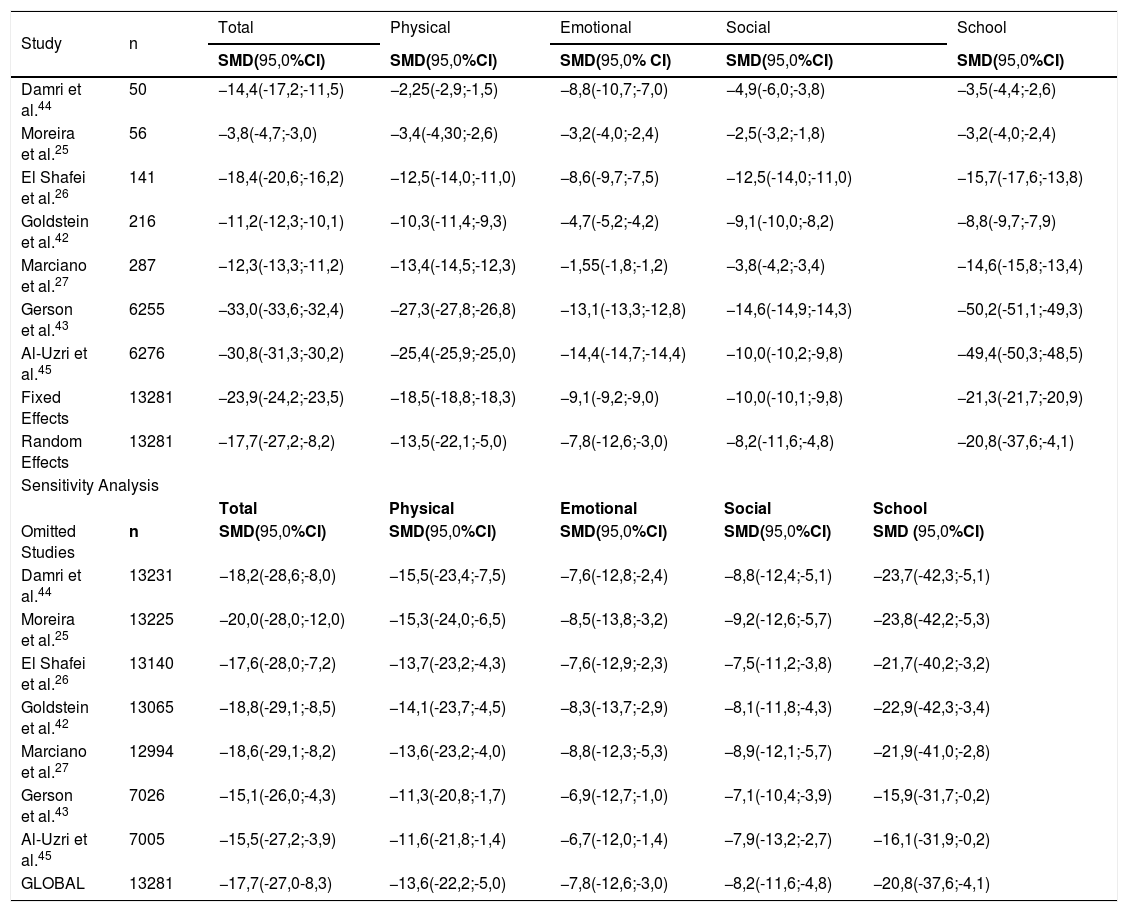
In the meta-analysis for each of the HRQL dimensions, we found heterogeneity with p values of 0.001 in Dersimonian Laird’s statistic, and RI coefficient of 0.9988. In addition to this, the absence of publication bias was evident with p values of 0.7639 in the Begg statistic. Taking into account the data abovementioned, we carried out a meta-analysis of standardized differences of means (SMD) under the random-effects model, which showed that pediatric patients with CKD have lower HRQL values than healthy patients in all dimensions evaluated; the values are as follows: in the physical dimension, the difference is SMD:13.6 (95%CI: 22.2; 5.0) points, in the emotional dimension SMD:7.8 (95% CI: 12.6; 3.0) points, in the social dimension SMD: 8.2 (95% CI:11.6; 4.8) points, in the school dimension SMD 20.8 (95% CI:37.6; 4.1) points and in the total SMD:17.7 (95%CI:27.0; 8.3) points (Fig. 2). The sensitivity analysis reflected that the overall difference in dimensions is not significantly influenced by any of the individual studies (Table 3).
Meta-analysis of mean difference and sensitivity analysis of the HRQL of patients with CKD and healthy ones according to the dimensions of the PedsQL.
| Study | n | Total | Physical | Emotional | Social | School | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| SMD(95,0%CI) | SMD(95,0%CI) | SMD(95,0% CI) | SMD(95,0%CI) | SMD(95,0%CI) | ||||||
| Damri et al.44 | 50 | −14,4(-17,2;-11,5) | −2,25(-2,9;-1,5) | −8,8(-10,7;-7,0) | −4,9(-6,0;-3,8) | −3,5(-4,4;-2,6) | ||||
| Moreira et al.25 | 56 | −3,8(-4,7;-3,0) | −3,4(-4,30;-2,6) | −3,2(-4,0;-2,4) | −2,5(-3,2;-1,8) | −3,2(-4,0;-2,4) | ||||
| El Shafei et al.26 | 141 | −18,4(-20,6;-16,2) | −12,5(-14,0;-11,0) | −8,6(-9,7;-7,5) | −12,5(-14,0;-11,0) | −15,7(-17,6;-13,8) | ||||
| Goldstein et al.42 | 216 | −11,2(-12,3;-10,1) | −10,3(-11,4;-9,3) | −4,7(-5,2;-4,2) | −9,1(-10,0;-8,2) | −8,8(-9,7;-7,9) | ||||
| Marciano et al.27 | 287 | −12,3(-13,3;-11,2) | −13,4(-14,5;-12,3) | −1,55(-1,8;-1,2) | −3,8(-4,2;-3,4) | −14,6(-15,8;-13,4) | ||||
| Gerson et al.43 | 6255 | −33,0(-33,6;-32,4) | −27,3(-27,8;-26,8) | −13,1(-13,3;-12,8) | −14,6(-14,9;-14,3) | −50,2(-51,1;-49,3) | ||||
| Al-Uzri et al.45 | 6276 | −30,8(-31,3;-30,2) | −25,4(-25,9;-25,0) | −14,4(-14,7;-14,4) | −10,0(-10,2;-9,8) | −49,4(-50,3;-48,5) | ||||
| Fixed Effects | 13281 | −23,9(-24,2;-23,5) | −18,5(-18,8;-18,3) | −9,1(-9,2;-9,0) | −10,0(-10,1;-9,8) | −21,3(-21,7;-20,9) | ||||
| Random Effects | 13281 | −17,7(-27,2;-8,2) | −13,5(-22,1;-5,0) | −7,8(-12,6;-3,0) | −8,2(-11,6;-4,8) | −20,8(-37,6;-4,1) | ||||
| Sensitivity Analysis | ||||||||||
| Total | Physical | Emotional | Social | School | ||||||
| Omitted Studies | n | SMD(95,0%CI) | SMD(95,0%CI) | SMD(95,0%CI) | SMD(95,0%CI) | SMD (95,0%CI) | ||||
| Damri et al.44 | 13231 | −18,2(-28,6;-8,0) | −15,5(-23,4;-7,5) | −7,6(-12,8;-2,4) | −8,8(-12,4;-5,1) | −23,7(-42,3;-5,1) | ||||
| Moreira et al.25 | 13225 | −20,0(-28,0;-12,0) | −15,3(-24,0;-6,5) | −8,5(-13,8;-3,2) | −9,2(-12,6;-5,7) | −23,8(-42,2;-5,3) | ||||
| El Shafei et al.26 | 13140 | −17,6(-28,0;-7,2) | −13,7(-23,2;-4,3) | −7,6(-12,9;-2,3) | −7,5(-11,2;-3,8) | −21,7(-40,2;-3,2) | ||||
| Goldstein et al.42 | 13065 | −18,8(-29,1;-8,5) | −14,1(-23,7;-4,5) | −8,3(-13,7;-2,9) | −8,1(-11,8;-4,3) | −22,9(-42,3;-3,4) | ||||
| Marciano et al.27 | 12994 | −18,6(-29,1;-8,2) | −13,6(-23,2;-4,0) | −8,8(-12,3;-5,3) | −8,9(-12,1;-5,7) | −21,9(-41,0;-2,8) | ||||
| Gerson et al.43 | 7026 | −15,1(-26,0;-4,3) | −11,3(-20,8;-1,7) | −6,9(-12,7;-1,0) | −7,1(-10,4;-3,9) | −15,9(-31,7;-0,2) | ||||
| Al-Uzri et al.45 | 7005 | −15,5(-27,2;-3,9) | −11,6(-21,8;-1,4) | −6,7(-12,0;-1,4) | −7,9(-13,2;-2,7) | −16,1(-31,9;-0,2) | ||||
| GLOBAL | 13281 | −17,7(-27,0-8,3) | −13,6(-22,2;-5,0) | −7,8(-12,6;-3,0) | −8,2(-11,6;-4,8) | −20,8(-37,6;-4,1) | ||||
SMD, standard mean difference.
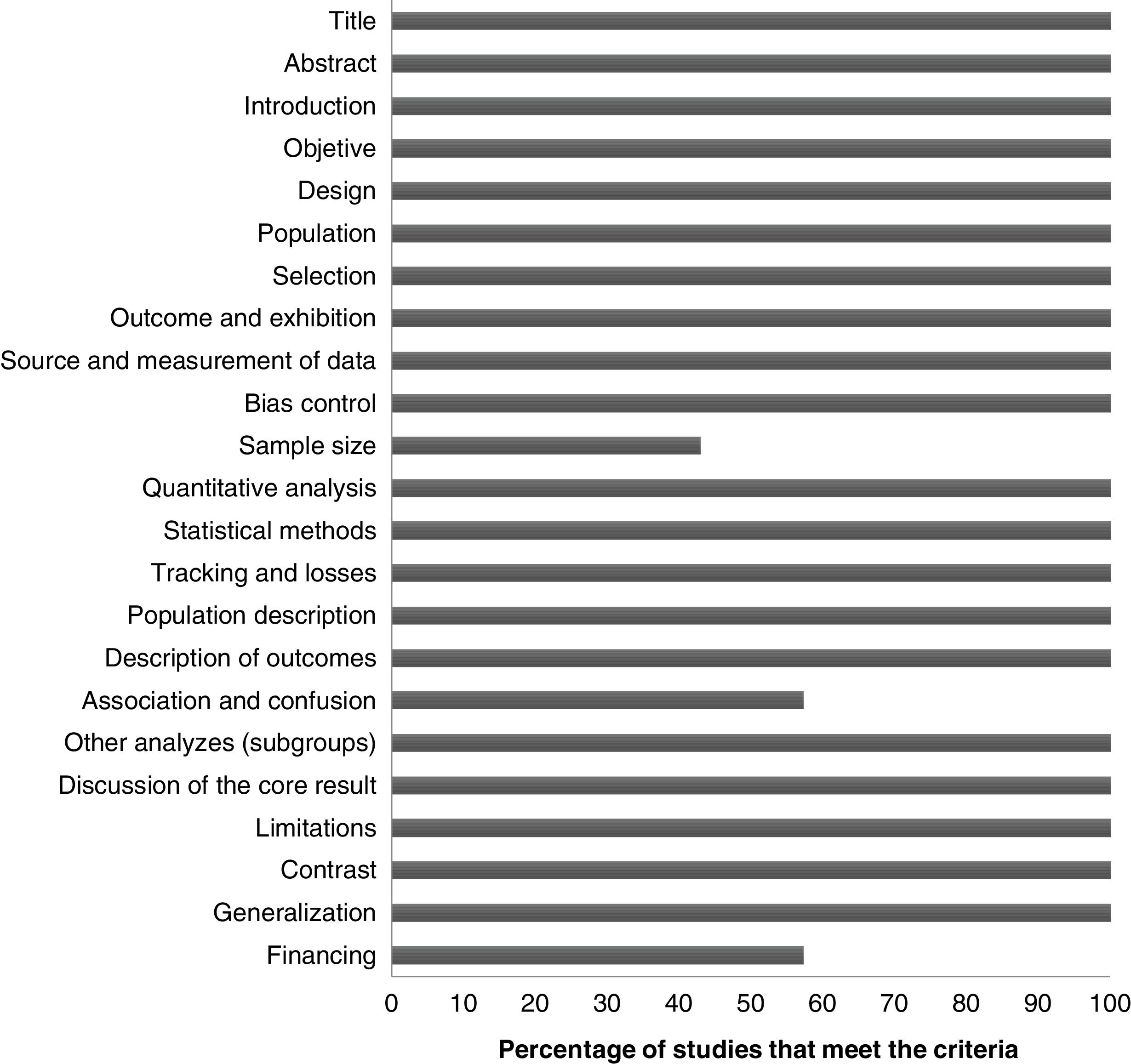
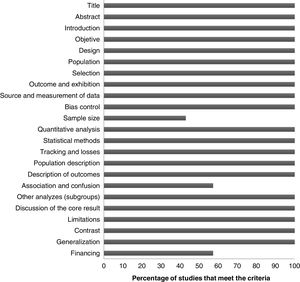
Finally, when evaluating the methodological quality of the investigations, we found that 20 of the 23 items evaluated were met in all publications. The items with the most shortcomings were the calculation of the sample size and the procedure for identifying confounding factors (Fig. 3).
DiscussionThe global findings of this study reveal that pediatric patients with chronic kidney disease have lower HRQL than healthy patients, specifically in the school, physical, emotional and social dimensions. These data are related to the research carried out by Aparicio et al.52 where patients show their perception of a poor quality of life related to their health, with significant differences in the domains of physical function, physical role, and general health status awareness. Likewise, a multi-center study carried out in the USA53 concluded that sick patients present greate in their physical activity as well as with their emotional and social health.
Regarding the physical dimension, we found that pediatric patients with CKD have lower scores than healthy individuals in 13.6 (22.2; 5.0) points. This is consistent with the results found by Goldstein et al.42 and McKenna et al.,54 who reported that the physical dimension in a pediatric patient presents lower rates compared to the healthy control individual. This difference can be attributed to the renal treatment modality (IQR 0.39–0.91, p<0.001) for dialysis patients (IQR 0.59−0.97, p=0.4), those with kidney transplants,55 or the presence of associated co-morbidities to the disease, such as short stature and below average weight (p=0.001) for stages 5 of the disease,56 metabolic alterations with blood hemoglobin levels less than 11.0g/dl in children aged 0.5–5 years and under 12.0g/dl in children aged 12–15 years,57 likewise, protein-calorie malnutrition, increased protein catabolism, and osteo-dystrophy kidney changes.58 In addition, frequent hospitalizations, and the pain that CKD patients feel from undergoing medical procedures (42%)55 cause restriction and alterations to their activities on top of the physical dimension deterioration.
In this sense, it is suggested that care programs for CKD pediatric patients involve different pharmacological, educational and alternative strategies that help them reduce signs and symptoms of their disease and should thus lead to physical activities tailored to their needs and their health/disease condition.59
On the other hand, the emotional dimension was the one that reflected the least differences between pediatric patients with CKD and healthy individuals with 7.8 (12.6; 3.0) points, a similar finding reported by Morales et al.60 where the emotional dimension in patients with peritoneal dialysis was 14.4 and in hemodialysis 12.3. Moreover, Tjaden et al.61 reported the experiences of 143 children undergoing therapy, where the emotional dimension is lower in the groups that present CKD and undergo dialysis treatment versus healthy controls. These data are closely related to the domains identified by Tjaden et al.61 as loss of control, high dependence on caregivers, relentless dependence on a machine, impaired bodily integrity, restricted lifestyle, limited opportunities for social interaction, academic struggle, positive determination, and self-awareness. In addition, children face limitations in developing similar activities as their healthy peers, such as having fun, playing, feeling good about themselves, especially their body image as they grow up.58
Accordingly, the interdisciplinary health team that is made up of play therapists, social workers, psychologists, and family psychotherapists together with doctors and nurses need to constantly address the real potential problems and emotional distress suffered by children and young people, where together with a coordinated plan of care that includes self-care strategies, psychosocial programs, and planned sessions for self-management, information, and contact with other patients can improve their emotional condition.62–64
Going forward to the social dimension, this study showed that there is a difference of 8.2 (11.6; 4.8) points between pediatric patients with CKD and healthy individuals, contrary data to those reported by Alvarado et al.,64 who evidenced individual weight differences below 30 points. In general terms, the dimension obtained a score of 19. This difference could be due to the frequency and duration of medical care and treatment modality, as well as the impossibility of carrying out social activities with friends because of their disease complications. Although receiving treatment may interrupt relationships with peers, it could strengthen family relationships, as reported by Morales et al.,60 where the family aspect obtained weight of 79 points, on the contrary to what was referred by Tjaden et al.61 in their qualitative review of the literature where the children expressed greater overprotection of their parents and presented limitations in relationships with friends. A hypothesis that emerges from this finding is that the presence of chronic kidney disease at an early age strengthens the family bond since the social support that arises within the family allows the child to adapt to the changes produced by the disease, improving his or her self-esteem, confidence, and self-confidence.
In these patients, the use of social networks, according to the globalized world, allows for the receiving of formal and informal support that can increase adherence to treatment, as well as in obtaining a greater sense of self-management, having access to information and in sharing experiences with peers, which entails understanding the disease and its stages in addition to taking on new self-care strategies.65
We should note that the school dimension was the most affected in pediatric CKD patients with a difference compared to healthy individuals of 20.8 (37.6; 4.1) points. Similar results are presented in investigations41,64,66 where the school environment reaches measures close to 78 points. At the time when the disease is diagnosed, the school grade in which the patient is in, and his/her age show significant correlation (p=0.03), the treatment modality also often decreases school participation. Buyan et al.67 in a cohort of 211 children with CKD reported that 50% of dialysis-treated patients and 37.9% of transplant patients had dropped out of school, indicating that the complications of the disease and the patient's hemodynamic imbalance may prevent them from continuing with their academic activities, moreover, being subjected to periodical school interruptions, delayed school work submission impacts negatively the learning process as stated by Powe et al.66 Similarly, Tjaden et al.61 says that prolonged absence from school for treatment-related problems can lead to a detrimental effect on maintaining peer relationships, self-esteem, and academic performance, negatively influencing the patient’s quality of life. However, care efforts have focused and expanded on gradually addressing the individual needs of the patient and their cultural characteristics, hence pro-schooling academic institutions have provided virtual learning platforms and support services for parents/caregivers so that children can continue with their academic activities.68 We should also point out that Renal units and academic institutions are entities that strengthen study opportunities and offer complimentary teaching strategies to avoid absenteeism.61
Finally, regarding the total of the HRQL scale, we found a difference of 17.7 (27.0; 8.3) points between pediatric patients with CKD and healthy individuals. This is consistent with that expressed by Goldstein et al.42 who reported that the quality of life is lower in patients with CKD than in healthy controls. Another study conducted in Texas69 confirmed similar findings, where the quality of life measurement in children is associated with certain treatments that affect their lives. As a result, a study evaluated the HRQL in relation to the treatment and found that transplant patients reported significantly better scores than dialysis patients.41 Similar data was reported in a study carried out in Peru,60 where the quality of life in children with a transplant was of 66.97 while in dialysis patients it was 29.79. This research found a total quality of life of 35.22 points, influenced by the school dimension (7.37), family relations (1265) and friends (23), likewise, recurrent hospitalizations, lack of information about the disease, bad or low self-perception, separation from friends and family, and interrupting school activities influenced low HRQL. We estimate that when children reach their youth, they have more stable levels of health-related quality of life and are comparable with the healthy population, supported by the perspective of education, treatment adherence, and future achievements.70
This meta-analysis on the impact of the quality of life in the pediatric population with chronic kidney disease is paramount because it allows for the achievement of a greater degree in generalizing the results in different contexts. In this sense, it was necessary to evaluate and investigate those dimensions affected by chronic kidney disease. We raised a hypothesis that forms the basis for investigating social and cultural factors that generate differences in each of the dimensions of quality of life related to health, as well as focused analytical studies on the family, self-perception, and changes in the disease and the environment.
During this study, we found heterogeneity in the articles included for analysis, which may be influenced by the characteristics of the population, the stage of chronic kidney disease, the country of origin, the characteristics of the environment and the individual perception of quality of life. Despite the above, Tufanaru et al.71 suggest that the decision to combine statistically heterogeneous studies should not be based solely on statistical heterogeneity. Studies that are conceptually similar with respect to participants, interventions, comparators, design, and risk of bias can be combined in a meta-analysis.71 Along the same lines, Jones et al.72 argues that it is common to find some degree of heterogeneity in meta-analysis, in which case, as was done in this manuscript, the random effects model should be performed.
Regarding the limitations of the study, first, it was not possible to perform a meta-regression between HRQoL and the important aspects of chronic kidney disease such as in creatinine levels, glomerular filtration rate, time of diagnosis, access, adherence treatment and social support, given that most studies did not report these variables. Second, most of the included studies have small sample sizes, and the sampling was not random, implying that external validity should be assessed with caution. Third, the studies included in this meta-analysis were descriptive, which is why their objective was not to test or falsify hypotheses but rather to describe HRQoL in healthy CKD patients. In this sense, it is important to specify that although the sum of the descriptive studies does not produce an analytical one, combining the results of the aforementioned investigations allowed the consolidation of the hypothesis on the great impact of CKD in pediatric patients by increasing the extrapolation framework of the conclusions, reducing the probability of error ßeta and improving the precision of the results, thus serving as basis for the design of subsequent analytical investigations that can explain the differential profile of HRQL. Additionally, the inclusion of descriptive studies was justified because prospective or analytical researches on this topic are limited.
Finally, manuscripts published in a language other than English, Spanish or Portuguese were not included, which is why further relevant research could have been omitted, as well as access to full texts on the web due to costs or invalidity in the downloading process.
ConclusionThis meta-analysis revealed the impact of chronic kidney disease on health-related quality of life in pediatric populations. The most affected dimension was regarding the school due to frequent visits to the doctor and the symptoms of the disease; the physical dimension showed low scores in pediatric patients, which is attributed to the modality of treatments associated with the disease that decreased their energy levels, making it hard to pursue normal daily activities; the social dimension showed that the complications of the disease reduce opportunities for social interaction with peers and to carry on recreational activities, and finally the emotional dimension was the one with the fewest weight differences.
FundingThis article is based on a doctoral thesis project “Significance of Quality of Life for adolescents with chronic kidney disease”. This work was supported by agreement of the Sinú University Elías Bechara Zainúm, Cartagena, No 2020-05.
Conflicts of interestThe authors declare no conflicts of interest.