This study aimed to evaluate the diagnostic utility, disease activity, and phenotypic association of serum anti-Saccharomyces cerevisiae antibody (ASCA), perinuclear anti-neutrophil cytoplasmic antibody (pANCA), PR3-ANCA, and MPO-ANCA in pediatric patients with inflammatory bowel disease (IBD).
MethodsPediatric patients diagnosed with IBD were recruited and classified as Crohn's disease (CD), ulcerative colitis (UC), and IBD-unclassified (IBD-U) through full investigation. The Paris classification was used to evaluate disease phenotypes of pediatric CD and UC.
ResultsIn all, 229 pediatric patients with IBD (CD 147, UC 53, IBD-U 29) were included. The ASCA IgG seropositivity significantly differed among the three groups (CD 75.4%, UC 17.5%, and IBD-U 60.0%; p < 0.001). PR3-ANCA positive rates were the highest in UC (24.0%), followed by IBD-U (17.6%), and none in CD (p = 0.002); pANCA-positive rates were higher in IBD-U (33.6%), followed by UC (28.0%) than in CD (1.4%) (p < 0.001). Regarding disease phenotype, perianal disease revealed higher serum ASCA IgG titers (median 36.7 U/mL in P1 vs. 25.2 U/mL in P0, p = 0.019). Serum ASCA IgG and IgA cutoff values to distinguish CD were 32.7 (U/mL) and 11.9 (U/mL), respectively, with a specificity of 80.0%.
ConclusionSerological biomarkers of ASCA IgG and IgA were effective for differentiating CD in pediatric IBD patients, and serum pANCA and PR3-ANCA, but not MPO-ANCA, were effective in distinguishing UC and IBD-U. Furthermore, measuring serological titers of ASCA IgG and IgA may help differentiate CD and evaluate the disease activity and phenotype of pediatric IBD in practice.
Inflammatory bowel disease (IBD) is a chronic relapsing inflammatory intestinal disease classified as Crohn's disease (CD), ulcerative colitis (UC), and IBD type unclassified (IBD-U). Subtypes of IBD are diagnosed according to the revised Porto criteria, and IBD-U is referred to when the diagnostic criteria for CD or UC are not met.1,2 In the screening and diagnostic evaluation of IBD in practice, serological markers such as the anti-Saccharomyces cerevisiae antibody (ASCA) and anti-neutrophil cytoplasmic antibody (ANCA) are shown to be practical non-invasive tests when it is challenging to diagnose CD or UC because of many indistinct clinical features of pediatric IBD.3–6
Of serological markers of IBD, ASCA IgG, and IgA are directed against the mannan cell wall of Saccharomyces cerevisiae, which shares homology with intestinal bacteria, and serum ASCA noted in CD patients indicates the loss of tolerance against commensal antigens.7 ANCAs are autoantibodies directed against various neutrophil cytoplasmic granules.8 There are two testing methods for detecting ANCA. One is the standard indirect immunofluorescence method (IIF), representing perinuclear ANCA (pANCA) or cytoplasmic ANCA (cANCA). The other method is enzyme-linked immunosorbent assay (ELISA), chemiluminescence immunoassay, which can be a quantitative analysis of antibodies against specific antigens representing anti-proteinase-3 with cytoplasmic ANCA (PR3-ANCA) or anti-myeloperoxidase with perinuclear ANCA (MPO-ANCA).8 PR3-ANCA was usually associated with c-ANCA as a distinct marker for microscopic polyangiitis in adults, and MPO-ANCA was associated with pANCA in IBD, Wegener granulomatosis, etc.8
To date, the diagnostic accuracies of ASCA and pANCA as serological markers of IBD have been evaluated in many previous studies; however, only a few studies on PR3-ANCA and MPO-ANCA in IBD, especially in children with UC.5,9 Moreover, no studies have examined the association between serological titers of these markers and the clinical phenotype and disease activity of pediatric IBD.
Therefore, this study aimed to evaluate the diagnostic utility of serum ASCA IgG, ASCA IgA, pANCA, PR3-ANCA, and MPO-ANCA in pediatric IBD and to determine the association between these serological biomarkers and their titers and disease activity, inflammatory markers, and disease phenotype of each pediatric IBD subtype.
MethodsStudy subjectsPediatric IBD patients diagnosed through full investigation between March 2004 and July 2021 at a single tertiary institution were recruited. All participant data were obtained retrospectively from their medical records, and all clinical data were collected at the time of diagnosis of IBD. The data of clinical and demographic characteristics (age, sex, disease activity, disease phenotype, serum and fecal inflammatory markers, and serological biomarkers of IBD such as ASCA IgG, ASCA IgA, pANCA, PR3-ANCA, and MPO-ANCA) were collected in all study subjects.
The following exclusion criteria were applied to the retrospective pediatric IBD patients: 1) infectious colitis, 2) diagnosis of primary immune deficiency, or 3) infantile IBD (diagnosed in children before 2 years of age).
Diagnosis of pediatric IBDThe diagnosis of IBD was confirmed based on clinical, endoscopic, radiologic, and pathological evaluations, and the subjects were divided into three subgroups representing CD, UC, and IBD-U, based on the revised Porto criteria.1 Small bowel lesions were examined by magnetic resonance enterography (MRE), capsule endoscopy, or if MRE failed due to claustrophobia, small bowel series. Patients in whom the symptomatic distinctions between UC and CD could not be identified were considered IBD-U cases; all the characteristics were consistent with UC but were rectal sparing, duodenal, or esophageal ulcers and not attributable to other causes (non-steroidal anti-inflammatory drugs, Helicobacter pylori, celiac disease, etc.) are examples of IBD-U.1
Disease phenotype and disease activity of pediatric IBDThe Paris classification was used to categorize pediatric patients with IBD based on their disease phenotypes.10 CD patients with perianal disease were defined as 'P1′, and those without were defined as 'P0′. To assess the disease activity of IBD in children, the pediatric Crohn's disease activity index (PCDAI) was used for CD, and the Pediatric ulcerative colitis activity index (PUCAI) was used for UC.11,12 In pediatric patients with IBD-U, both PCDAI and PUCAI criteria were applied for each disease phenotype.
Laboratory markers of inflammationAll laboratory values were obtained at diagnosis and the following markers were measured; C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), total white blood cell count (WBC), absolute neutrophil count (ANC), hemoglobin (Hb), hematocrit (Hct), platelet count, and serum levels of total protein and albumin. Fecal calprotectin was measured using a fluorescence enzyme immunoassay (Calprotectin, Phadia AB, Sweden).
Serological markers of IBDSerological biomarkers were collected at the time of diagnosis, and the serum ASCA IgG and IgA levels were analyzed by ELISA (INOVA Diagnostics Inc., San Diego, USA) following the manufacturer's protocol. The manufacturer's serum ASCA IgG and IgA cutoff values were set at ≥ 20 U/mL.
The entire ANCA (pANCA, PR3-ANCA, and MPO-ANCA) was tested simultaneously as the diagnosis, regardless of whether the pANCA was positive. Serum pANCA levels were analyzed using an indirect IIF antibody test (Axioskop, Jena, Germany) with a cut-off value of≥ 1:40 IU/mL. Serum PR3‐ANCA and MPO‐ANCA levels were analyzed using ELISA (Immunocap 250 kit, Tokyo, Japan) with cutoff values set to ≥ 2.0 IU/mL for PR3‐ANCA and ≥ 3.5 IU/mL for MPO-ANCA. All ANCA panels were analyzed according to the manufacturer's instructions, and the Institute for Reference Materials and Measurements recently certified reference materials for PR3-ANCA and MPO-ANCA.13 PR3 is an antigen recognized by c-ANCA, and c-ANCA testing could not be performed alone in this hospital. Other serological techniques could not be performed owing to hospital constraints. Serum ANCA positive rate means that any of the pANCA, PR3-ANCA, and MPO-ANCA is positive.
Statistical analysisStatistical analyses were performed in a non-parametric manner. For continuous variables, the Kruskal-Wallis test was used for three-group analyses. For statistically significant variables, post-hoc Bonferroni analysis was performed. All values are expressed as median and interquartile range (IQR). The χ2 test or Fisher's exact test was used for numeric variables. Bivariate correlation analyses were performed using Pearson correlation. Receiver operating characteristic (ROC) curve analysis was performed to assess the diagnostic accuracy of serum ASCA IgG and IgA as serological markers for CD. Statistical analyses were performed using SPSS version 25.0 (IBM Corp. Armonk, NY, USA) A p values below 0.05 were considered statistically significant.
Ethical considerationsThis study was approved by the Institutional Review Board of the Seoul National University Hospital Institutional Review Board (IRB approval number: B-2111-720-101). The requirement for informed consent from enrolled patients and parents was waived by the institutional review board.
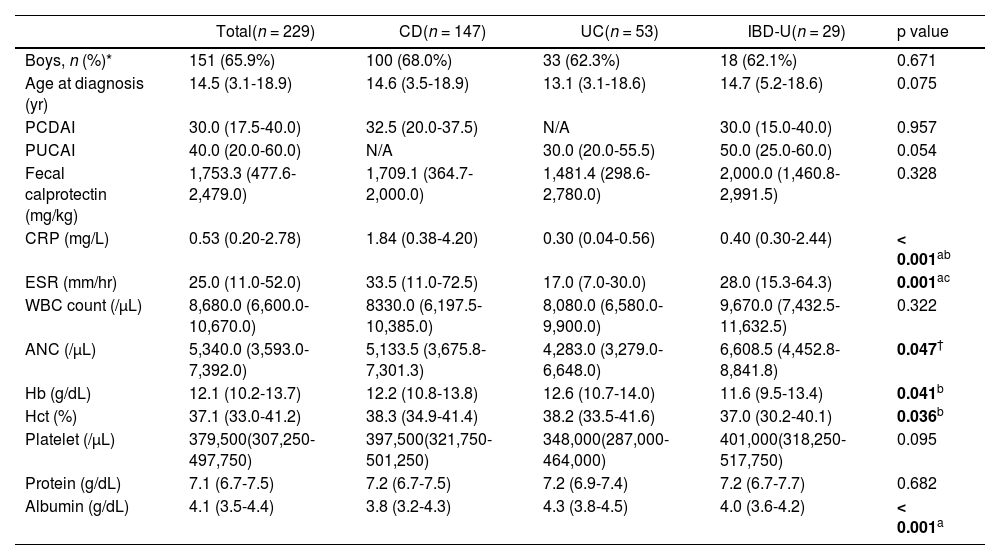
ResultsPatient characteristicsA total of 229 participants (151 boys, 78 girls; mean age: 14.0 ± 3.3 years) were included (CD: 147, UC: 53, and IBD-U: 29). The mean duration from initial symptom onset to diagnosis was 153.2 ± 208.1 days, with a significant difference among the three IBD subgroups (p = 0.032). Post-hoc analysis showed a difference between CD and UC (median 96.0 vs. 48.0 days, p = 0.026), but not IBD-U. The demographics, clinical features, and laboratory findings of the study participants are shown in Table 1.
Demographic parameters, disease activity of pediatric inflammatory bowel disease, and inflammatory parameters of the study subjects at diagnosis.
| Total(n = 229) | CD(n = 147) | UC(n = 53) | IBD-U(n = 29) | p value | |
|---|---|---|---|---|---|
| Boys, n (%)* | 151 (65.9%) | 100 (68.0%) | 33 (62.3%) | 18 (62.1%) | 0.671 |
| Age at diagnosis (yr) | 14.5 (3.1-18.9) | 14.6 (3.5-18.9) | 13.1 (3.1-18.6) | 14.7 (5.2-18.6) | 0.075 |
| PCDAI | 30.0 (17.5-40.0) | 32.5 (20.0-37.5) | N/A | 30.0 (15.0-40.0) | 0.957 |
| PUCAI | 40.0 (20.0-60.0) | N/A | 30.0 (20.0-55.5) | 50.0 (25.0-60.0) | 0.054 |
| Fecal calprotectin (mg/kg) | 1,753.3 (477.6-2,479.0) | 1,709.1 (364.7-2,000.0) | 1,481.4 (298.6-2,780.0) | 2,000.0 (1,460.8-2,991.5) | 0.328 |
| CRP (mg/L) | 0.53 (0.20-2.78) | 1.84 (0.38-4.20) | 0.30 (0.04-0.56) | 0.40 (0.30-2.44) | < 0.001ab |
| ESR (mm/hr) | 25.0 (11.0-52.0) | 33.5 (11.0-72.5) | 17.0 (7.0-30.0) | 28.0 (15.3-64.3) | 0.001ac |
| WBC count (/μL) | 8,680.0 (6,600.0-10,670.0) | 8330.0 (6,197.5-10,385.0) | 8,080.0 (6,580.0-9,900.0) | 9,670.0 (7,432.5-11,632.5) | 0.322 |
| ANC (/μL) | 5,340.0 (3,593.0-7,392.0) | 5,133.5 (3,675.8-7,301.3) | 4,283.0 (3,279.0-6,648.0) | 6,608.5 (4,452.8-8,841.8) | 0.047† |
| Hb (g/dL) | 12.1 (10.2-13.7) | 12.2 (10.8-13.8) | 12.6 (10.7-14.0) | 11.6 (9.5-13.4) | 0.041b |
| Hct (%) | 37.1 (33.0-41.2) | 38.3 (34.9-41.4) | 38.2 (33.5-41.6) | 37.0 (30.2-40.1) | 0.036b |
| Platelet (/μL) | 379,500(307,250-497,750) | 397,500(321,750-501,250) | 348,000(287,000-464,000) | 401,000(318,250-517,750) | 0.095 |
| Protein (g/dL) | 7.1 (6.7-7.5) | 7.2 (6.7-7.5) | 7.2 (6.9-7.4) | 7.2 (6.7-7.7) | 0.682 |
| Albumin (g/dL) | 4.1 (3.5-4.4) | 3.8 (3.2-4.3) | 4.3 (3.8-4.5) | 4.0 (3.6-4.2) | < 0.001a |
1Values are presented as number of patients (%) *or median (IQR).
2A p value of less than 0.05 was considered statistically significant.
3For numeric variables, the Chi-square test was used, and for continuous variables, the Kruskal-Wallis test was used. Significance values have been adjusted by the Bonferroni correction. P value < 0.05 for comparison of CD vs. UC, CD vs. IBD-U and UC vs. IBD-U;
4CD, Crohn's disease; UC, ulcerative colitis; IBD-U, inflammatory bowel disease unclassified; PCDAI, Pediatric Crohn's disease activity index; PUCAI, Pediatric ulcerative colitis activity index; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; WBC, white blood cell; ANC, absolute neutrophil count; Hb, hemoglobin; Hct, hematocrit
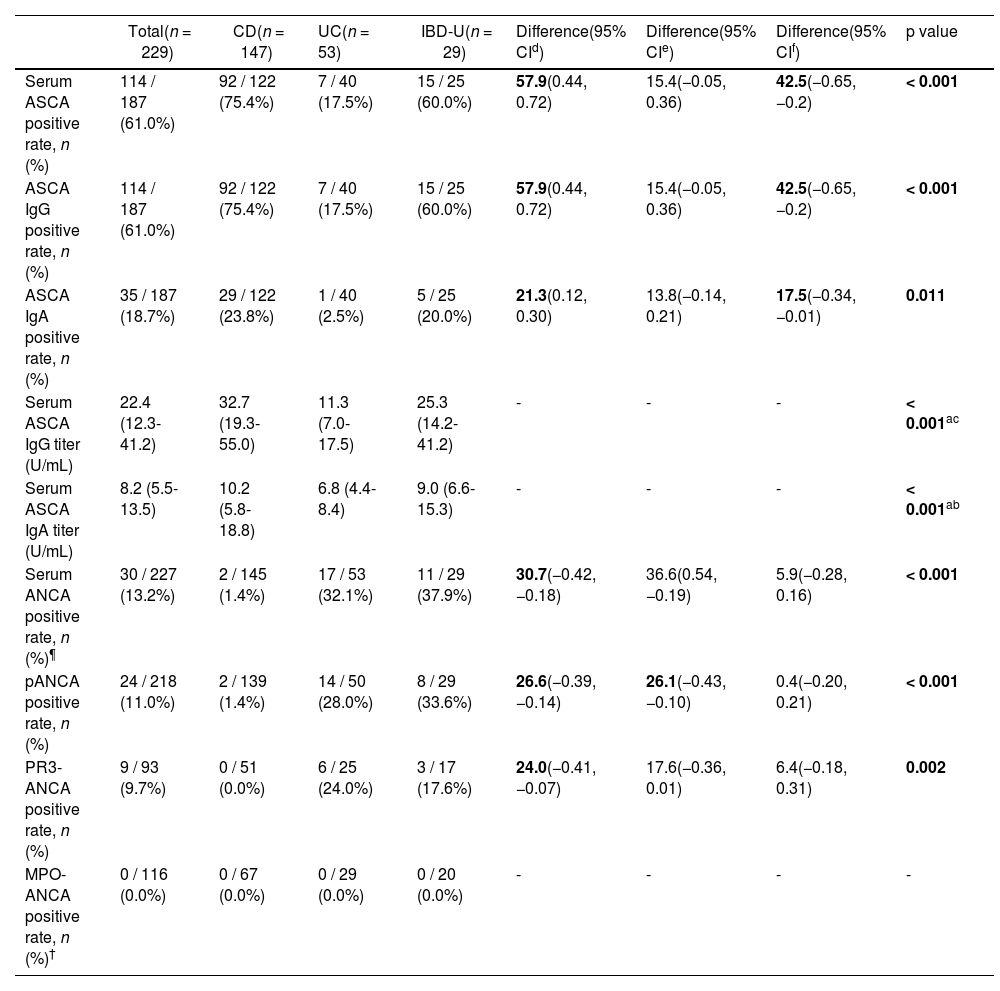
The serological results of ASCA IgG and IgA are reported in Table 2. Serum ASCA IgG and IgA titers were significantly higher in CD (median 32.7 U/mL and 10.2 U/mL, respectively), followed by IBD-U (median 25.3 U/mL and 9.0 U/mL, respectively) than in UC (median 11.3 U/mL and 6.8 U/mL, respectively) (p < 0.001 for both ASCA IgG and IgA).
Comparison of seropositivity and serum titers of anti-Saccharomyces cerevisiae antibodies and anti-neutrophil cytoplasmic antibodies among disease subtypes of pediatric inflammatory bowel disease.
| Total(n = 229) | CD(n = 147) | UC(n = 53) | IBD-U(n = 29) | Difference(95% CId) | Difference(95% CIe) | Difference(95% CIf) | p value | |
|---|---|---|---|---|---|---|---|---|
| Serum ASCA positive rate, n (%) | 114 / 187 (61.0%) | 92 / 122 (75.4%) | 7 / 40 (17.5%) | 15 / 25 (60.0%) | 57.9(0.44, 0.72) | 15.4(−0.05, 0.36) | 42.5(−0.65, −0.2) | < 0.001 |
| ASCA IgG positive rate, n (%) | 114 / 187 (61.0%) | 92 / 122 (75.4%) | 7 / 40 (17.5%) | 15 / 25 (60.0%) | 57.9(0.44, 0.72) | 15.4(−0.05, 0.36) | 42.5(−0.65, −0.2) | < 0.001 |
| ASCA IgA positive rate, n (%) | 35 / 187 (18.7%) | 29 / 122 (23.8%) | 1 / 40 (2.5%) | 5 / 25 (20.0%) | 21.3(0.12, 0.30) | 13.8(−0.14, 0.21) | 17.5(−0.34, −0.01) | 0.011 |
| Serum ASCA IgG titer (U/mL) | 22.4 (12.3-41.2) | 32.7 (19.3-55.0) | 11.3 (7.0-17.5) | 25.3 (14.2-41.2) | - | - | - | < 0.001ac |
| Serum ASCA IgA titer (U/mL) | 8.2 (5.5-13.5) | 10.2 (5.8-18.8) | 6.8 (4.4-8.4) | 9.0 (6.6-15.3) | - | - | - | < 0.001ab |
| Serum ANCA positive rate, n (%)¶ | 30 / 227 (13.2%) | 2 / 145 (1.4%) | 17 / 53 (32.1%) | 11 / 29 (37.9%) | 30.7(−0.42, −0.18) | 36.6(0.54, −0.19) | 5.9(−0.28, 0.16) | < 0.001 |
| pANCA positive rate, n (%) | 24 / 218 (11.0%) | 2 / 139 (1.4%) | 14 / 50 (28.0%) | 8 / 29 (33.6%) | 26.6(−0.39, −0.14) | 26.1(−0.43, −0.10) | 0.4(−0.20, 0.21) | < 0.001 |
| PR3-ANCA positive rate, n (%) | 9 / 93 (9.7%) | 0 / 51 (0.0%) | 6 / 25 (24.0%) | 3 / 17 (17.6%) | 24.0(−0.41, −0.07) | 17.6(−0.36, 0.01) | 6.4(−0.18, 0.31) | 0.002 |
| MPO-ANCA positive rate, n (%)† | 0 / 116 (0.0%) | 0 / 67 (0.0%) | 0 / 29 (0.0%) | 0 / 20 (0.0%) | - | - | - | - |
1The Chi-square test was used for numeric variables, and the values are presented as number of patients (%) or median (IQR).
2The Kruskal-Wallis test was used for continuous variables, and the Bonferroni correction adjusted the significance values.
3The difference was expressed with a 95% CI by comparing the size of the positivity rate between the two groups. 95% CI.d,CD vs. UC; 95% CI; e CD vs. IBD-U; 95% CI f UC vs. IBD-U
4A p value of less than 0.05 was considered statistically significant.
5CI, Confidence interval; ASCA, anti-Saccharomyces cerevisiae antibody; ANCA, anti-neutrophil cytoplasmic antibody; CD, Crohn's disease; UC, ulcerative colitis; IBD-U, inflammatory bowel disease unclassified
The serological results of pANCA, PR3-ANCA, and MPO-ANCA are presented in Table 2. pANCA-positive rates were higher in IBD-U (33.6%) and UC (28.0%) than in CD (1.4%), with a significant difference among the 3 subgroups (p < 0.001). In Table 2, all serological markers showed statistically significant p values, and the difference was expressed with a 95% confidence interval (CI) by comparing the size of the positivity rate between the two groups.
Inflammatory markers compared across disease phenotypes of pediatric IBDRegarding inflammatory markers based on disease phenotypes in CD, age subgroups showed statistically significant differences in Hct levels (p = 0.018), with Hb in A1a significantly lower than in A2 in post-hoc analysis (36.1% in A1a vs. 40.4% in A2, p = 0.016). As for the disease location of CD, the L1, L2, L3, and L4 subgroups revealed significant differences in fecal calprotectin (p < 0.001), ESR (p < 0.001), WBC (p = 0.025), ANC (p = 0.015), Hb (p = 0.008), platelet count (p < 0.001), and albumin levels (p = 0.010). The results of the post-hoc analysis are in Supplemental Table 1. Regarding the existence of perianal disease (P1) in CD patients, there were significant differences in Hb (12.8 g/dL in P0 vs. 12.1 g/dL in P1, p = 0.015) and Hct (39.0% in P0 vs. 37.0% in P1, p = 0.038).
In a subgroup analysis of inflammatory markers based on disease extent [E1∼E4] in UC, there were significant differences in fecal calprotectin (p = 0.049), CRP (p < 0.001), ESR (p < 0.001), Hb (p < 0.001), Hct (p < 0.001), platelet count (p = 0.003), protein (p = 0.003), and albumin (p < 0.001). The results of the post-hoc analysis are in Supplemental Table 2.
As for the severity of UC [S0∼S1], fecal calprotectin (p = 0.006), CRP (p = 0.015), ESR (p < 0.001), WBC (p < 0.001), ANC (p = 0.002), Hb (p < 0.001), Hct (p = 0.001), platelet count (p < 0.001), and albumin (p = 0.007) were significantly different. Results of post-hoc analysis are described in Supplementary Table 3. In subgroup analysis, IBD-U showed an overall similar trend to CD and UC.
Serological markers of ASCA IgG and IgA compared according to disease phenotypes of pediatric IBDWith regards to titer analysis of serological markers according to disease phenotypes in CD and IBD-U, only perianal disease was associated with higher serum ASCA IgG titers [median 36.7 (21.4-60.0) U/mL in P1 vs. 25.2 (16.6-52.0) U/mL in P0, p = 0.019], but not ASCA IgA titers. ASCA IgA titers were significantly higher shown in the L3 subgroup than in L2 (p = 0.025).
One patient with UC and primary sclerosing cholangitis was positive for ASCA IgG with a titer of 41.8 U/mL, while all other markers were negative.
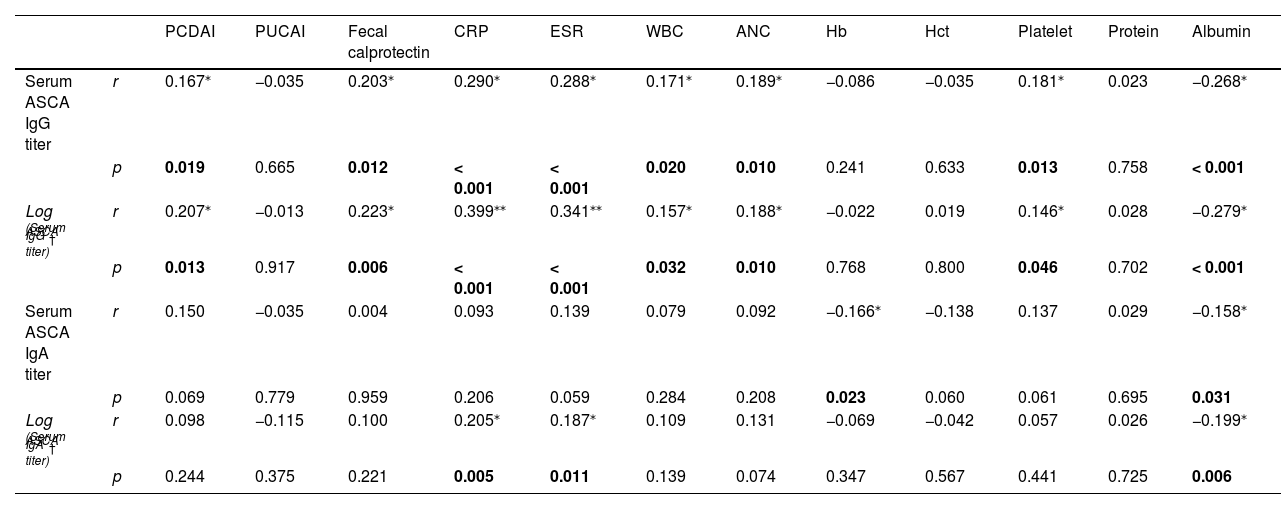
Correlation of serum ASCA titers with disease activity and inflammatory markers of pediatric IBDThe results of correlation analyses of serum ASCA IgG and IgA titers with disease activity and other inflammatory markers in pediatric patients with IBD are described in Table 3.
Correlation analyses of serum anti-Saccharomyces cerevisiae antibody titers with disease activity and inflammatory parameters of pediatric inflammatory bowel disease.
| PCDAI | PUCAI | Fecal calprotectin | CRP | ESR | WBC | ANC | Hb | Hct | Platelet | Protein | Albumin | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Serum ASCA IgG titer | r | 0.167⁎ | −0.035 | 0.203⁎ | 0.290⁎ | 0.288⁎ | 0.171⁎ | 0.189⁎ | −0.086 | −0.035 | 0.181⁎ | 0.023 | −0.268⁎ |
| p | 0.019 | 0.665 | 0.012 | < 0.001 | < 0.001 | 0.020 | 0.010 | 0.241 | 0.633 | 0.013 | 0.758 | < 0.001 | |
| Log (Serum ASCA IgG titer)† | r | 0.207⁎ | −0.013 | 0.223⁎ | 0.399⁎⁎ | 0.341⁎⁎ | 0.157⁎ | 0.188⁎ | −0.022 | 0.019 | 0.146⁎ | 0.028 | −0.279⁎ |
| p | 0.013 | 0.917 | 0.006 | < 0.001 | < 0.001 | 0.032 | 0.010 | 0.768 | 0.800 | 0.046 | 0.702 | < 0.001 | |
| Serum ASCA IgA titer | r | 0.150 | −0.035 | 0.004 | 0.093 | 0.139 | 0.079 | 0.092 | −0.166⁎ | −0.138 | 0.137 | 0.029 | −0.158⁎ |
| p | 0.069 | 0.779 | 0.959 | 0.206 | 0.059 | 0.284 | 0.208 | 0.023 | 0.060 | 0.061 | 0.695 | 0.031 | |
| Log (Serum ASCA IgA titer)† | r | 0.098 | −0.115 | 0.100 | 0.205⁎ | 0.187⁎ | 0.109 | 0.131 | −0.069 | −0.042 | 0.057 | 0.026 | −0.199⁎ |
| p | 0.244 | 0.375 | 0.221 | 0.005 | 0.011 | 0.139 | 0.074 | 0.347 | 0.567 | 0.441 | 0.725 | 0.006 |
1The above data were analyzed by all three IBD groups (CD, UC, and IBD-U), and bivariate correlation analysis was performed on the variables using the Pearson correlation.
2Based on the absolute value of r, it is indicated as follows.
3A p value of less than 0.05 was considered statistically significant.
4ASCA, anti-Saccharomyces cerevisiae antibody; PCDAI, pediatric Crohn's disease activity index; PUCAI, pediatric ulcerative colitis activity index; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; WBC, white blood cell; ANC, absolute neutrophil count; Hb, hemoglobin; Hct, hematocrit
The authors also performed correlation analyses using logarithmic values for all variables to identify clinically significant differences. In subgroup analysis, logarithmic values of serum ASCA IgG titers in pediatric CD patients were positively correlated with PCDAI (r = 0.187, p = 0.042), fecal calprotectin (r = 0.360, p < 0.001), CRP (r = 0.297, p < 0.001), ESR (r = 0.263, p = 0.004), ANC (r = 0.195, p = 0.031), and platelet counts (r = 0.212, p = 0.019) and negatively correlated with Hb (r = -0.192, p = 0.034). Serum ASCA IgA titers were negatively correlated only with Hb (r = -0.191, p = 0.035); however, log values of serum ASCA IgA titers showed no correlation.
In the UC and IBD-U subgroup correlation analysis, there were no values showing a significant correlation.
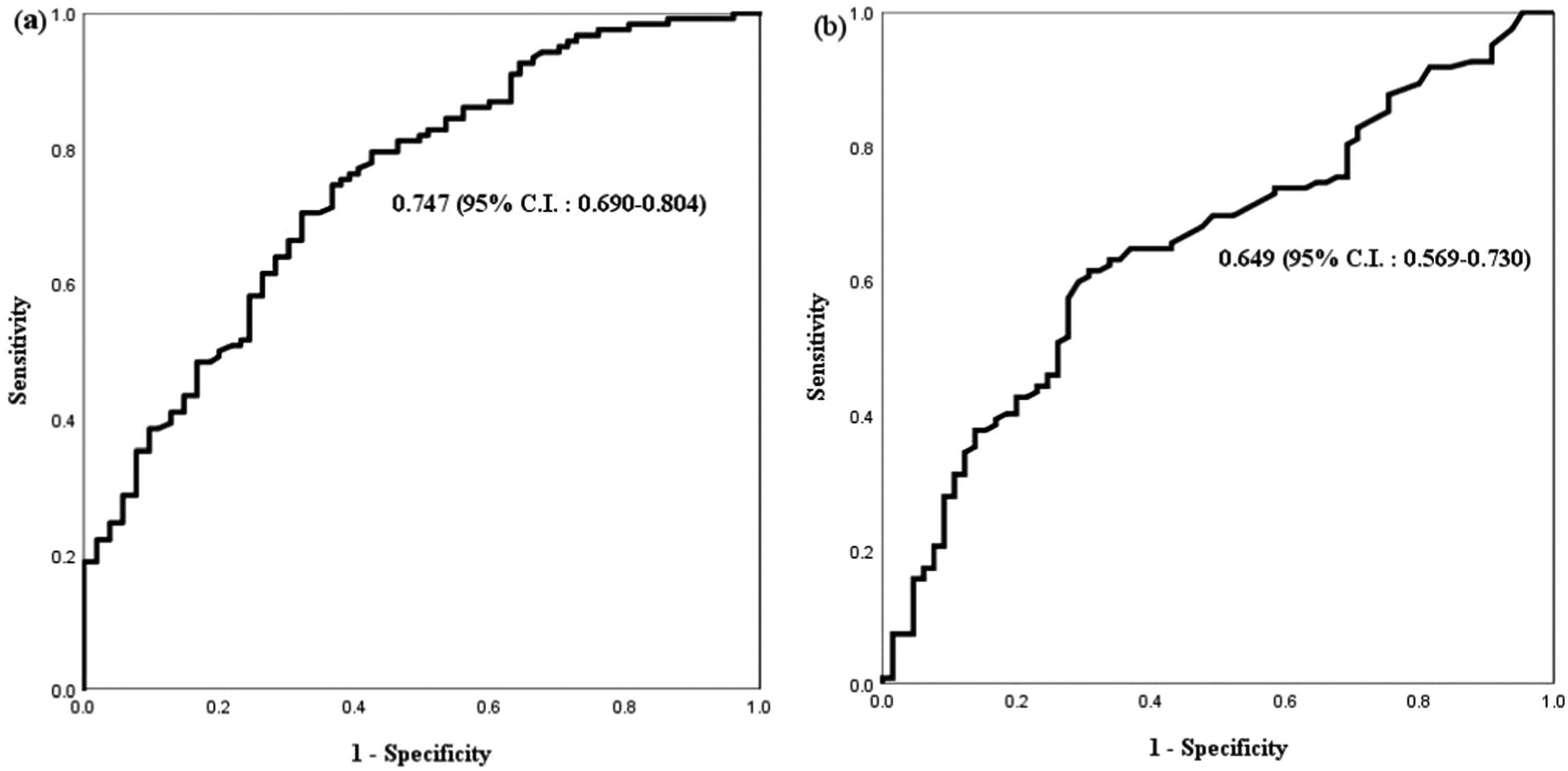
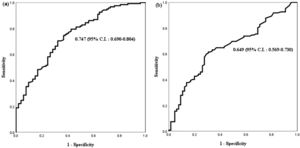
Diagnostic accuracy of serum ASCA IgG and IgA titersThe diagnostic accuracy of serum ASCA IgG and IgA titers for pediatric CD was evaluated by performing ROC analysis and calculating the area under the ROC curve (AUC) (Figure 1). By ROC analysis, the optimal cutoff value for serum ASCA IgG was 32.7 U/mL for pediatric CD with a sensitivity of 50.0%, a specificity of 80.0%, a positive predictive value (PPV) of 80.7%, and a negative predictive value (NPV) of 58.9%. The optimal cutoff value for serum ASCA IgA was 11.9 U/mL for pediatric CD with a sensitivity of 42.6%, a specificity of 80.0%, PPV of 82.8%, and NPV of 38.8%.
Receiver operating characteristic curve (ROC) for serum ASCA IgG and IgA in CD vs. non-CD patients (UC and IBD-U) (A) ROC curve for serum ASCA IgG titer in CD vs. non-CD patients. The cut-off value for serum ASCA IgG was 32.7 (U/mL) for pediatric CD with a sensitivity of 50.0% and a specificity of 80.0%, for pediatric CD. (B) ROC curve for serum ASCA IgA titer in CD vs. non-CD patients (UC, IBD-U). The cut-off value for serum ASCA IgA was 11.9 (U/mL) with a sensitivity of 42.6% and a specificity of 80.0%, for pediatric CD.
1 ASCA, anti-Saccharomyces cerevisiae antibody; CD, Crohn's disease; UC, ulcerative colitis; IBD-U, inflammatory bowel disease unclassified.
In this single-center retrospective study of pediatric IBD patients, the authors investigated reliable serological markers to differentiate pediatric IBD subgroups (CD, UC, and IBD-U) and reflect disease activity and clinical phenotype of pediatric IBD. Our study revealed that compared with pediatric UC patients, pediatric CD and IBD-U patients had higher serum ASCA IgG and IgA titers as well as higher seropositivity rates for both ASCA IgG and IgA. Furthermore, in the present study, PR3-ANCA-positive rates were the highest in UC, followed by IBD-U, whereas pANCA-positive rates were the highest in IBD-U, followed by UC. MPO-ANCA was negative in all study subjects.
In a previous report by Birimberg-Schwartz et al.,14 serum ANCA positivity was most prevalent in UC, followed by IBD-U, and then CD (64%, 43%, and 30%, respectively; p < 0.001), when pANCA was tested with IIF method. In our study using the same testing method for pANCA, pANCA was most frequently positive in IBD-U (33.6%), followed by UC (28.0%); and when serum PR3-ANCA was tested by ELISA method, it was most frequently positive in UC. Therefore, even the same ANCA test may have different positive rates depending on the test method. Chandradevan et al.2 reported that IBD-U patients were given another classification after two years of follow-up, with a 2:1 ratio of UC (n = 35) to CD (n = 19). Upon follow-up of IBD-U, the pANCA-positive patients were more likely to be reclassified as UC than CD. Therefore, a positive pANCA may predict that IBD-U is more similar to the characteristics of UC than CD.
As for PR3-ANCA, a recent study in pediatric patients with CD and UC, excluding IBD-U, revealed that the positive rate of PR3-ANCA was significantly higher in UC (57.6%) than in CD (7.1%),5 and similarly, in our study, seropositivity rate of PR3-ANCA was the highest in UC and followed by IBD-U and none of the pediatric CD patients were positive for PR3-ANCA. The positive rate of MPO-ANCA in both UC and IBD-U was 0% in our study, which is similar to previous studies reporting a very low positivity rate of MPO-ANCA in UC.9,15
Regarding the association of serological markers and disease phenotypes of IBD, Chandrakumar et al.16 examined the associations between ASCA-positive status and disease location and behavior in a pediatric cohort and suggested that ASCA-positive patients did not have a higher risk of perianal disease. In contrast, the authors found that perianal lesions were associated with greater ASCA IgG quantitative values, but not with ASCA IgG or IgA seropositivity, in our study. The difference may be because a previous study focused only on seropositivity instead of serological titers of ASCAs. Therefore, our results suggest the clinical utility of measuring serum ASCA IgG titers in pediatric CD-related perianal disease. In UC, Mahler et al.17 reported that PR3-ANCA reactivity was associated with more extended disease location in adults. In the present study, pancolitis, the most severe type of UC, was found in six of the nine patients with positive PR3-ANCA, reflecting the degree of disease extent.
Regarding serum titers of ASCA and disease activity of IBD, Canani et al.18 reported a relationship between serum ASCA titers and only PCDAI. However, in our study, serum ASCA IgG titers were correlated positively with PCDAI as well as fecal calprotectin, CRP, ESR, WBC, ANC, and platelet count. The negative relationship between serum albumin levels and ASCA IgG and IgA titers indicates that these antibodies tend to increase in response to severe inflammatory responses and protein loss. This may indicate that greater quantitative levels of serum ASCA titers reflect more severe disease, similar to earlier studies demonstrating that serum albumin might indicate disease severity.19 In the correlation analysis, when the log value was applied to ASCA IgG and IgA titer and variables, the effect size of CRP and ESR was found to be larger, showing that serum ASCA IgG and IgA titer are more related to inflammatory responses than other variables. Based on our results, serum ASCA IgG and IgA titers may be useful as serological biomarkers in evaluating the disease activity of pediatric IBD.
As for diagnostic accuracy of serum ASCA, Mizuochi et al.20 found that ASCA had 26.7% sensitivity and 95.0% specificity using cutoff values provided by only seropositivity instead of serological titers. In the present study, serum ASCA IgG titers could distinguish pediatric CD from UC and IBD-U with a sensitivity of 50.0% and specificity of 80.0% at an optimal cutoff of 32.7 U/mL, and serum ASCA IgA titers with a sensitivity of 42.6% and specificity of 80.0% with a cutoff of 11.9 U/mL. When additional internal verification with 2/3 and 1/3 random data from the original study subjects was conducted to validate the results of this study, similar ASCA IgG and IgA titer results (internal validation set 1: 30.70 U/mL and 11.45 U/mL, set 2: 32.75 U/mL and 11.95 U/mL, respectively) were shown. Beyond confirming seropositivity for ASCA, measuring serological titers of ASCA IgG and IgA may additionally help differentiate CD from non-CD in practice, providing noteworthy insight.
Nevertheless, several limitations were present in this study. First, to compensate for the small sample size limitation, the difference was estimated for the two groups separately. Regarding effect size (difference), there was a greater difference in the 95% CI in CD vs. UC (57.9%) than in UC vs. IBDU (42.5%) in ASCA IgG. For pANCA, the difference in the 95% CI between CD and UC (26.6%) was comparable to that between CD and IBD-U (26.1%). Second, all patients included in this study were East Asian children, and data were collected from a single center. Further prospective multicenter studies of serological markers for pediatric IBD should be conducted in multiple ethnic groups and countries.
In conclusion, the authors found serological markers, including ASCA IgG, ASCA IgA, pANCA, and PR3-ANCA, excluding MPO-ANCA, to be valuable biomarkers of pediatric IBD. Furthermore, measuring serological titers of ASCA IgG and IgA may additionally help evaluate the disease activity and phenotype of pediatric IBD in practice. This is the first study to comprehensively cover the clinical values and the measurement of serum ASCA titers as well as serum ANCAs, including PR3-ANCA and MPO-ANCA, along with existing pANCA for the diagnosis and differentiation of pediatric IBD and its subgroups such as CD, UC, and IBD-U.