To characterize the influence of birthplace on outcomes of patients with gastroschisis admitted to three hospitals in a state in Brazil’s southeastern region, according to condition inborn (born in a reference center) or outborn (born outside the reference center).
MethodsRetrospective multicenter cohort study of patients with gastroschisis. The sample size utilized was of patients admitted in three hospitals with a diagnosis of gastroschisis ICD 10 Q79.3 between January 2000 to December 2018. Patients were divided into two groups, inborn and outborn. Characteristics of prenatal, perinatal and postoperative were compared using statistical tests. The level of significance adopted was P-value < 0.05.
ResultsIn total, 144 cases of gastroschisis were investigated. The outborn patients group had higher rates of absence of antenatal diagnosis (p = 0.001), vaginal delivery (p = 0.001), longer time between birth and abdominal wall closure surgery (p = 0.001), to silo removal (p = 0.001), to first enteral feeding (p = 0.008), for weaning from mechanical ventilation (p = 0.034), used less peripherally inserted central catheter (PICC) and required more venous dissections (p = 0.001), and lower mean of serum sodium (p = 0.015). There were no differences in mortality rates and length of hospital stay between the inborn and outborn groups.
ConclusionAlthough outborn patients with gastroschisis were less likely to have an antenatal diagnosis and were more prone to a longer time to undergo surgical and feeding procedures, and to spend more time in mechanical ventilation, these disadvantages seemed not to reflect on the death rate and the length of hospital stay of patients from this group.
Gastroschisis is a congenital anomaly that consists of a defect between the insertion of the umbilical cord and abdominal wall located on right, in most cases, with prolapse of intestines, rarely liver, and without covering of membranes.1 Adequate care inthemanagement of newborns with gastroschisis requires delivery in a tertiary care center, with the availability of neonatal surgery, multidisciplinary team in the neonatal intensive care unit (NICU).2 In this scenario, access to highly complex care decreased gastroschisis mortality to less than 5% in cohorts from high-income countries.3,4 In contrast, these rates remain high, ranging from 39.1% to 58%, according to data from recent studies, in low middle-income countries.5–7 Delivery outside the referral center, which is more likely to happen in those countries, has been described as a risk factor associated with death.5–7
Three previous studies on inborn and outborn patients with gastroschisis performed in high-income countries found different results.8–10 In Brazil, studies carried out in hospitals in the north and northeast regions reported similar gastroschisis mortality rates of 51.2% and 51.6%, respectively. In both studies most patients were outborn.11,12 In the southeast region, two recent studies reported mortality rates of 4 and 10.6%, wherein most of the entire population consisted of inborn, respectively.13,14
The state of Espírito Santo (ES) is located in southeast Brazil, a region that has the highest human development indexes (HDI) in the country.15 The ES provides a regionalized, structured health system with NICUs beds concentrated in the Metropolitan Region of Grande Vitória – ES (MRGV-ES).16 In this context, the objective of this study was to characterize the influence of birthplace on outcomes of patients with gastroschisis admitted to three NICUs in ES, according to inborn or outborn condition.
MethodsMulticenter retrospective cohort study carried out in three NICUs of public hospitals located in MRGV-ES, Brazil.
This study enrolled patients who were admitted in three NICUs from January 2000 to December 2018 with the diagnosis of gastroschisis (ICD 10 Q79.3) as registered in their surgical act reports. Periods of data collection varied according to availability of medical records of each hospital: from 2000 to 2018 at NICU A (where were treated most gastroschisis’s patients before opening NICUs B and C), from 02/23/2013 to 12/31/2018 at NICU B (data collected since the beginning of NICU operation from 2013) and between 11/11/2010 to 12/31/2018 at NICU C (although it was opened in 2002, data was only available in the system as of 2011). The exclusion criteria were individuals with genetic syndromes or other major congenital malformations. Patients were treated by pediatric surgeons and other members of a multidisciplinary team at each study place. Two patients from NICU B, who were referred from hospitals other than the ones participating in this study, were included, since their perinatal, delivery, and birth data were fully available. This project was approved by Research Ethics Committee — Opinion No. 2671249/CEP – CIAS/Unimed-Vitória (CAAE 87878918.1.0000.5061). The terms of adherence to this project by study hospitals were obtained through the Secretaria de Estado da Saúde-ES (SESA-ES).
The research population was divided into two groups, inborn or outborn. The inborn was defined as a patient whose delivery, surgical and clinical treatment were performed at the same place (referral tertiary center). All patients not designated as inborn were considered outborn.10
Demographic variables were the place of origin (birthplace - municipalities in central, northern, and southern regions of ES and neighboring states), place of admission (UTIN A, UTIN B, UTIN C). Prenatal and perinatal variables were maternal age (years), parity (primiparous or multiparous), antenatal diagnosis (yes or no), ultrasound (US) result (normal, gastroschisis, other malformation, not performed), number of prenatal care consultations (≤5 or >5), route of delivery (cesarean section or vaginal delivery), APGAR bulletin (score 0–10), gestational age (weeks) by CAPURRO method (most used in term newborns or borderline preterm infants), birth weight (grams), gender (female or male), the time between birth and first repair surgery (hours). Variables of postoperative assistance were simple or complex gastroschisis, classified according to aspect of loops as reported by surgeon;17,18 simple (normal intestinal loops with or without malrotation) and complex (high degree of abdominovisceral disproportion, atresias, strictures, volvulus, necrosis), time of mechanical ventilation (MV) (time and percentage of days to extubation/ ≤ or >15 days (the cutoff point was based in Miranda et al.),19 time to first enteral feeding (mean), time to full enteral feedings (mean), time on parenteral nutrition (PN) (mean), use of vasoactive substances (yes or no), use of antimicrobials (1, 2 or more regimens), type of venous access (peripherally inserted central catheter — PICC, deep venipuncture, venous dissection or peripheral venoclysis), use of blood products, serum sodium in milliequivalent per liter (mEq/L) first dosage in first week of life (normal reference values between 135 to 145 mE/L-, serum albumin in grams per deciliter (g/dL) -first dosage in first week of life, normal reference values between 3.5 to 4.8 g/dL), neonatal clinical and/or comproved sepsis diagnosed by attending physician (yes or no), short bowel syndrome (yes or no), length of hospital stay (days) and discharge outcome (death or alive).
The statistical analysis was performed using Statistical Package for Social Sciences (SPSS) version 22.0 for Windows. Categorical variables were compared using the chi-square or Fisher's exact tests. For quantitative variables, normality tests were performed. In case of normality, Student’s t-test was performed and results presented with means and standard deviations. In the case of non-normality, Mann–Whitney non-parametric test was performed and results were presented with median and interquartile range (IQR). Level of significance adopted was 5% (P-value <0.05).
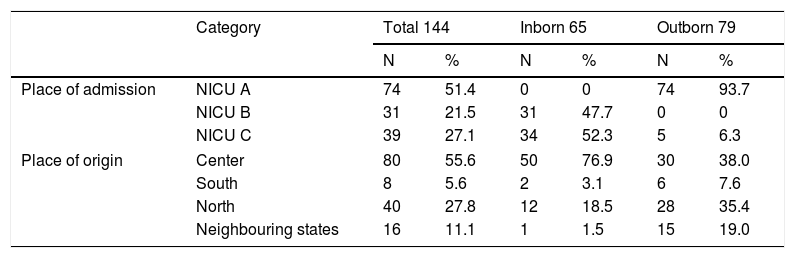
ResultsIn total, 144 cases of gastroschisis were investigated between January 2000 to December 2018. Table 1 shows the distribution of patients by place of admission and region of origin, according to inborn or outborn groups. Most cases from regions outside MRGV-ES, (located in the central region) or from neighboring states were treated at NICU A, which admits only outborn patients. On the other hand, most cases of inborn patients came from the central region of ES.
Place of admission and region of origin of patients with gastroschisis treated in three NICUs of RMGV-ES, BR.
| Category | Total 144 | Inborn 65 | Outborn 79 | ||||
|---|---|---|---|---|---|---|---|
| N | % | N | % | N | % | ||
| Place of admission | NICU A | 74 | 51.4 | 0 | 0 | 74 | 93.7 |
| NICU B | 31 | 21.5 | 31 | 47.7 | 0 | 0 | |
| NICU C | 39 | 27.1 | 34 | 52.3 | 5 | 6.3 | |
| Place of origin | Center | 80 | 55.6 | 50 | 76.9 | 30 | 38.0 |
| South | 8 | 5.6 | 2 | 3.1 | 6 | 7.6 | |
| North | 40 | 27.8 | 12 | 18.5 | 28 | 35.4 | |
| Neighbouring states | 16 | 11.1 | 1 | 1.5 | 15 | 19.0 | |
NICUs, Neonatal intensive care unit; RMGV-ES, Metropolitan Region of Grande Vitória - Espírito Santo.
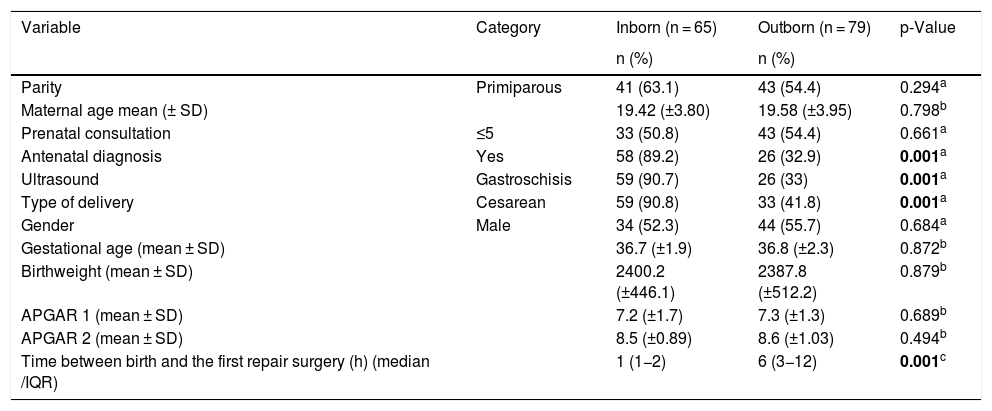
Table 2 shows variables of prenatal and perinatal. Diagnosis of gastroschisis during pregnancy was more frequent in the inborn group (p = 0.001). Although the obstetric US was performed in 100% of inborn and 85% of outborn, the percentage of positivity for gastroschisis was 90.7% in inborn and only 33% in outborn patients. Cesarean section was more frequent in the inborn patients (p = 0.001) and the time between birth and first repair surgery was longer in the outborn group (p = 0.001). There were no differences between the two groups regarding the mean of maternal age, parity, number of prenatal consultations, gender, gestational age, mean of birth weight, APGAR bulletin.
Prenatal and perinatal variables of patients with gastroschisis treated in three NICUs in RMGV-ES, BR.
| Variable | Category | Inborn (n = 65) | Outborn (n = 79) | p-Value |
|---|---|---|---|---|
| n (%) | n (%) | |||
| Parity | Primiparous | 41 (63.1) | 43 (54.4) | 0.294a |
| Maternal age mean (± SD) | 19.42 (±3.80) | 19.58 (±3.95) | 0.798b | |
| Prenatal consultation | ≤5 | 33 (50.8) | 43 (54.4) | 0.661a |
| Antenatal diagnosis | Yes | 58 (89.2) | 26 (32.9) | 0.001a |
| Ultrasound | Gastroschisis | 59 (90.7) | 26 (33) | 0.001a |
| Type of delivery | Cesarean | 59 (90.8) | 33 (41.8) | 0.001a |
| Gender | Male | 34 (52.3) | 44 (55.7) | 0.684a |
| Gestational age (mean ± SD) | 36.7 (±1.9) | 36.8 (±2.3) | 0.872b | |
| Birthweight (mean ± SD) | 2400.2 (±446.1) | 2387.8 (±512.2) | 0.879b | |
| APGAR 1 (mean ± SD) | 7.2 (±1.7) | 7.3 (±1.3) | 0.689b | |
| APGAR 2 (mean ± SD) | 8.5 (±0.89) | 8.6 (±1.03) | 0.494b | |
| Time between birth and the first repair surgery (h) (median /IQR) | 1 (1−2) | 6 (3−12) | 0.001c |
NICUs, Neonatal intensive care units; RMGV-ES, Metropolitan Region of Grande Vitória – Espírito Santo; SD, standart deviation; h, hours; IQR, interquartile range.
Bold values signifies p-Values < 0,05.
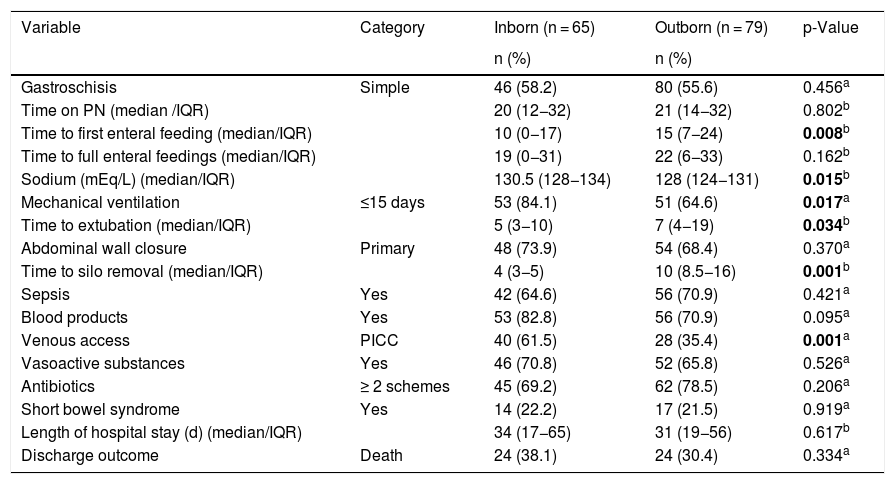
Table 3 shows the results of postoperative care variables. The mean time to first enteral feeding was lower in the inborn group (p = 0.008), the mean time to full enteral feedings and time on PN was similar in both groups. Serum sodium had a lower mean value in outborn patients (p = 0.015). Since serum albumin values were available in only 71 (49%) patients, the statistical analysis of this variable could not be performed. In all cases in which this protein was measured, its level was below the normal value (mean- 2.2 g/dL); moreover, it was slightly lower amongst outborn than inborn patients, respectively 2.1 g/dL and 2.3 g/dL. Use of PICC was higher in the inborn group (p = 0.001), and outborn patients had a greater need for dissections or venous punctures. Inborn group had higher proportion of individuals with MV use time below 15 days (p = 0.017) and a lower mean time to extubation (p = 0.034). The method of closing the abdominal wall was similar in both groups. For those patients who underwent secondary closure, the time for silo removal was shorter in the inborn group (p = 0.001). There were no significant differences between the two groups regarding simple or complex gastroschisis, the number of antimicrobial regimens, use of vasoactive substances, sepsis, use of blood products, short bowel syndrome, length of hospital stay, and rate of deaths (p = 0.334). The total mortality rate was 33.8%.
Variables of hospital care in the postoperative period of patients with gastroschisis treated in three NICUs in RMGV-ES, BR.
| Variable | Category | Inborn (n = 65) | Outborn (n = 79) | p-Value |
|---|---|---|---|---|
| n (%) | n (%) | |||
| Gastroschisis | Simple | 46 (58.2) | 80 (55.6) | 0.456a |
| Time on PN (median /IQR) | 20 (12−32) | 21 (14−32) | 0.802b | |
| Time to first enteral feeding (median/IQR) | 10 (0−17) | 15 (7−24) | 0.008b | |
| Time to full enteral feedings (median/IQR) | 19 (0−31) | 22 (6−33) | 0.162b | |
| Sodium (mEq/L) (median/IQR) | 130.5 (128−134) | 128 (124−131) | 0.015b | |
| Mechanical ventilation | ≤15 days | 53 (84.1) | 51 (64.6) | 0.017a |
| Time to extubation (median/IQR) | 5 (3−10) | 7 (4−19) | 0.034b | |
| Abdominal wall closure | Primary | 48 (73.9) | 54 (68.4) | 0.370a |
| Time to silo removal (median/IQR) | 4 (3−5) | 10 (8.5−16) | 0.001b | |
| Sepsis | Yes | 42 (64.6) | 56 (70.9) | 0.421a |
| Blood products | Yes | 53 (82.8) | 56 (70.9) | 0.095a |
| Venous access | PICC | 40 (61.5) | 28 (35.4) | 0.001a |
| Vasoactive substances | Yes | 46 (70.8) | 52 (65.8) | 0.526a |
| Antibiotics | ≥ 2 schemes | 45 (69.2) | 62 (78.5) | 0.206a |
| Short bowel syndrome | Yes | 14 (22.2) | 17 (21.5) | 0.919a |
| Length of hospital stay (d) (median/IQR) | 34 (17−65) | 31 (19−56) | 0.617b | |
| Discharge outcome | Death | 24 (38.1) | 24 (30.4) | 0.334a |
NICUs, Neonatal intensive care units; RMGV-ES, Metropolitan Region of Grande Vitória – Espírito Santo; PN, parenteral nutrition; SD, standart deviation; d, days; IQR, interquartile range.
Bold values signifies p-Values < 0,05.
This is the first multicenter study carried out in Brazil that assessed outcomes of patients with gastroschisis, according to inborn or outborn condition. In this study, there were no significant differences in mortality rates between the two groups. A similar result was reported in two studies carried out in high-income countries.8,10 Perhaps, this finding may reflect the fact that once outborn patients are referred to tertiary reference centers, they may benefit in time from high technology and specialized neonatal care. Hence, the disadvantages faced by this group could be offset by their referral to a specialized center.
The mortality rate of 33.8% observed in this study is higher than studies conducted in other states of the southeast region of Brazil (4%–14.9%);13,14,20 lower than the ones reported by the study carried out in the north (51.2%) and northeast part of the country (51.6%),11,12 and close to a study also conducted in southeastern Brazil that showed a total mortality rate from gastroschisis of 26.9%.19 However, Miranda et al. compared patients before and after implantation of protocols for monitoring pregnant women of fetuses with gastroschisis from antenatal diagnosis to the evolution of newborns in NICU at a regional center. They observed a decrease in gastroschisis mortality rate from 34.3% to 24.8%, despite the increase in rate of complex gastroschisis from 11.4% to 15.7% in the same period. This result corroborates the importance of monitoring gastroschisis cases in tertiary reference centers, with the implementation of protocols for improving quality of care.19
In a study carried out in a high-income country, the rate of antenatal diagnosis of gastroschisis was higher in inborn patients with statistical significance.10 In this study, the prenatal diagnosis rate of gastroschisis in outborn patients was low (33%), notwithstanding the fact that 85% of their mothers underwent this imaging exam. Since most patients in this group were from the state countryside or neighboring states, the failure to detect malformation in gestational US may likely be related to aspects associated with qualification of prenatal care outside the MRGV-ES.
In this study, the rate of cesarean section performed in the inborn patients was higher than outborn, probably related to a low rate of antenatal diagnosis in the outborn group. In a high-income country study, there were no differences in the delivery route between both patients, however, scheduled delivery was higher (p < 0.01) in inborn patients.10 Delivery time and birth route are a controversial issue in gastroschisis cases. Possible benefits of elective preterm delivery include decreased bowel exposure to amniotic fluid, reduction of necrosis, atresia, and its sequelae. However, the risks of elective premature delivery include comorbidities, such as respiratory distress syndrome, which can complicate the postoperative period of these patients.2
In an outborn group, the time between birth and the first repair surgery was longer, and the mean values of serum sodium and albumin were lower. These findings are interconnected since a more prolonged exposure time of intestinal loops increases the loss of those nutrients. Moreover, the aforementioned metabolic disorders seem to be related to increased days of MV in this group as suggested by a Brazilian multicenter study. According to its authors, edema of intestinal loops predispose to depletion of albumin and sodium, and, consequently, to low hydrostatic and oncotic pressures, which, can involve the lungs and ventilation, which is already hampered by the abdominal pressure. This chain of events could explain the increase in ventilation time observed in newborns with hyponatremia and hypoalbuminemia.21
In this study rate of complex gastroschisis was similar in both groups. This data was similar to that found in a study carried out in a high-income country.10 In low-income countries, delays in access to neonatal surgical care and inadequate prehospital management have been reported to be causes of hypothermia, hypovolemia, sepsis, and hydroelectrolytic disorders in patients with gastroschisis.22 These complications may reflect on postnatal factors, such as contamination and torsion of the vascular pedicle, and result in ischemia and intestinal necrosis, which are lesions typified as complex gastroschisis.5,22 Such serious intestinal injuries were associated with death in patients with gastroschisis in a study carried out in the northern region of the country, whose population was mostly outborn. According to the authors, high mortality rates observed by infection and sepsis are probably due to inadequate prenatal care, causing a lack of antenatal diagnosis and leading to inadequate delivery planning, delay in surgical correction (found a mean time of 18.7 h between delivery and wall closure), consequently, increased risk of postoperative infection and sepsis.11
Delayed closure of gastroschisis has been associated with a longer length of hospital stay and duration of TPN.23 In this study, among the patients who underwent secondary closure of the abdominal wall, those in the outborn group had a longer time for silo removal. Furthermore, the mean time to first enteral feeding was higher in this group. Nonetheless, the mean time to parenteral nutrition suspension and to reach full enteral feedings were similar in both groups.
Inborn patients had more placement of PICC, and consequently, less need for venous dissections than outborn patients. This difference may be explained by the fact that the older cohort was a compound of outborn patients and because PICC was introduced at this facility in 2007. A study in Mexico found a significant reduction of deaths in patients with gastroschisis after implantation of a protocol for improving the quality of care, which included PICC on admission to NICU.24
The current concept of short bowel syndrome is a requirement for parenteral nutrition greater than 60 days after intestinal resection or a bowel length of less than 25% of expected.25 The total rate of short bowel syndrome in this study of 21.8% was greater than recent studies carried out in southeastern Brazil (6% and 13%).13,14 This data may be related to surgical complications and the high mortality rate found.
In this study, there was no difference in the mean length of hospital stay between inborn and outborn patients. Two studies carried out in high-income countries showed different results; in the first, the outborn group had a longer hospital stay;10 in the second there was no statistically significant difference between outborn and inborn patients.8 A study in an upper-middle-income country found a longer mean of length of stay in an outborn group.5
Since this is a retrospective study, there may be some information bias. The studied outborn population consisted of cases that survived and had access to a tertiary hospital, so there may have been some selection bias. In addition, the treatments used in reference centers depended on the experience of multi-professional teams and protocols for approaching gastroschisis of each NICU. These limitations can be corrected in future multicenter studies with prospective randomized controlled study design.
Our study showed that patients born in a tertiary center had different outcomes than those born in peripheral maternity hospitals, but with no statistically significant difference in mortality rates in both groups. Although this result suggests that not all neonates with gastroschisis should be born in a tertiary referral center, the authors pointed out the need for faster and specialized transport for outborn patients, with the guarantee of beds in tertiary centers; planned delivery with strict protocols for inborn, to always achieve the shortest time between delivery and the first surgical intervention, with reduction of postoperative complications.
Conflicts of interestThe authors declare no conflicts of interest.
The authors are grateful to Dr. Marcelo Ramos Muniz for the text review.
This research is linked to the Graduate Program in Collective Health, Universidade Federal do Espírito Santo (UFES), Vitória, ES, Brazil.