This study aimed to analyze, through the morphometric method, the perimeter and length of the tongue, the collagen fibers, and the perimeter of blood vessels at different gestational ages and fetal weights.
Material and methodsTongues (n=55) of stillborns autopsied at 23–40 weeks of gestational age were macroscopically analyzed, and their length and perimeter were measured. Fifty-five tongue fragments were collected through a longitudinal section in the region that accompanies the median lingual sulcus and histologically processed. Slides were stained with picrosirius and immunolabeled with CD31 antibody. Quantification was performed on collagen fibers under polarized light, and on the perimeter of vessels with the CD31.
ResultsA positive and significant correlation of gestational age with tongue perimeter and length was found. There was a positive and significant correlation between collagen fibers and gestational age, as well as between gestational age and the perimeter of blood vessels. Between collagen fibers and fetal weight, a positive and significant increase was observed. Regarding the correlation between the perimeter of blood vessels and the fetal weight, an increase was observed.
ConclusionAs gestational age advances, there is an increase in tongue perimeter and length, in the percentage of collagen fibers, and in vascular perimeter, demonstrating that tongue formation is directly related to tongue growth and development.
Analisar, por meio do método morfométrico, o perímetro e o comprimento da língua, as fibras de colágeno, o perímetro dos vasos sanguíneos, em idades gestacionais e de acordo com o peso fetal.
Materiais e métodosLínguas (n=55) de natimortos autopsiados com 23-40 semanas de idade gestacional foram analisadas macroscopicamente, medidas em comprimento e perímetro; 55 fragmentos das línguas foram coletados por meio de uma secção longitudinal na região que acompanha o sulco lingual médio e processados histologicamente. As lâminas foram coloridas com picrosirius e imunomarcadas com o anticorpo CD31. A quantificação foi feita em fibras de colágeno examinadas com microscópio de luz polarizada e o perímetro dos vasos com o CD31.
ResultadosFoi encontrada uma correlação positiva e significativa da idade gestacional com o perímetro e o comprimento da língua. Houve uma correlação positiva e significativa entre as fibras de colágeno e a idade gestacional; bem como entre a idade gestacional e o perímetro dos vasos sanguíneos; e houve um aumento positivo e significativo entre as fibras de colágeno e o peso fetal. No que diz respeito à correlação entre o perímetro dos vasos sanguíneos e o peso fetal, houve um aumento.
ConclusãoConforme a idade gestacional avança, há um aumento no perímetro e no comprimento da língua, um aumento no percentual de fibras de colágeno e um aumento no perímetro vascular, demonstra que a formação da língua está diretamente relacionada ao crescimento e ao desenvolvimento da língua.
Pediatric autopsy is an important study of the structural and functional differences of the organs according to the time of fetal development. In the autopsy, the estimation of the gestational age (GA) is important to identify whether fetal development was occurring normally, to determine the death time in relation to the birth, to ascertain the diagnosis of the diseases that are specific to that developmental stage, and to detect those children classified as risk in the neonatal period.1
During examination, the evaluation of the tongue provides a variety of information, as it is a special organ of reception, chewing, swallowing, speaking, and tasting. Evidences from mammalian studies suggests that it is composed of muscle cells that have different arrangements in origin and insertion, and different histochemical properties in comparison with other skeletal muscles.2,3
The development of the tongue is described as a relatively rapid process, which begins between the fourth and fifth week of intrauterine life. This process has a remarkable effect on the oral cavity4; therefore, it is extremely important for the development of the stomatognathic system that the tongue develops correctly.5 There appears to be a synchrony in the formation of the orofacial complex since, from the 14th week onwards, the muscles of the oropharyngeal region are sufficiently advanced to move the tongue, coinciding with peaks in the growth of the head circumference that occur between the 15th and 17th week.6,7
Collagen is expressed in the tongue in the early stages of development, and is detectable in the mesenchyme derived from cranial neural crest cells (CNCC), adjacent to the tongue epithelium8 and in tendons of the extrinsic muscles, which connect the tongue to the mandible.9 The connective tissue and the vascular system of the tongue are derived from the CNCC, while most of its muscles originate from myoblasts that migrated from the occipital somites.10
Endothelial cells play a key role in the control of coagulation, thrombosis, vascular tone, permeability, inflammation, tissue repair, and angiogenesis.11 They constitute a heterogeneous population of cells in the human body. Functions and molecular characteristics of endothelial cells vary along the vascular tree and in the same organ between different vessels, as for example, phenotypic variations can occur in the expression of the CD31 molecule in these cells.12–14
Ultrasound examinations have indicated a highly significant correlation between fetal tongue circumference and gestational age (14 to 26 weeks).15 This data may be useful in the prenatal diagnosis of suspected congenital syndromes that include, among its manifestations, tongue growth disorders and GA estimation.12
The autopsy material is very rich for research, since through macro- and microscopic analyses it is possible to make feasible research studies with clinical diagnoses and detection of structural abnormalities. The autopsy is considered an important diagnostic method for the physician, since it allows documenting and comparing clinical and pathological cases.1 In pediatric autopsies, the hallux-calcaneus length (HCL) is a reliable parameter to establish GA in fetuses and stillbirths. The GA obtained by HCL is taken by measuring the length of the foot, from the heel to the tip of the hallux.16
The aim of this study was to analyze, through the morphometric method, the perimeter and length of the tongue, the collagen fibers, the perimeter of the blood vessels, at different GAs and in relation to the fetal weight. It can contribute as a new method to estimate GA through the tongue development.
Material and methodsThis was a retrospective study, approved by the Ethics Committee of the Federal University of Triângulo Mineiro, under protocol No. 1158.
Of the 152 pediatric autopsy reports analyzed, those of 55 stillborns autopsied by the General Pathology Discipline at the Clinical Hospital of the Federal University of Triângulo Mineiro (HC/UFTM), Uberaba, State of Minas Gerais, from 1994 to 2015 were selected. The anatomopathological examination was performed by two pathologists, and the information obtained from the autopsy reports was GA, determined using the HCL method (hallux-calcaneus length), and fetal weight.
Inclusion criteria were GA between 23 and 40 weeks, those with data of gender and fetal weight, and those in which the tongue was in good preservation condition. The exclusion criteria were stillborn infants with malformations and lack of data in reports such as GA, fetal weight and gender. Moreover, any cases with intrauterine growth restriction or another alteration were excluded.
Measurement of tongue length and perimeterThe 55 tongues analyzed were arranged on the macroscopy laboratory bench and identified individually, with the respective autopsy number, along with a ruler for later calibration in the ImageJ® Software (National Institutes of Health, USA). All photographs were taken from the same distance (30cm) using a Canon Rebel® camera (Canon, Tokyo, Japan). Morphometric analyses were performed measuring the length from the glossoepiglottic fold to the apex of the tongue; to obtain the perimeter of the tongue, the contour was measured throughout the macroscopic extension.
Sample collection and histopathological processingFifty-five tongue fragments from autopsied stillborns recovered in the archive of biological material of the discipline of General Pathology (UFTM) were used. Fragments were removed through a longitudinal section in the region that accompanies the median lingual sulcus, with a thickness of approximately 0.5cm. Serial cuts of 4μm in thickness were then performed. Slides were stained with Picrosirius (PS; saturated aqueous solution of picric acid added with 0.1g% Sirius red F3B) (Bayer, Leverkusen, Germany) with counterstaining by hematoxylin, and a slide was used for immunohistochemistry.
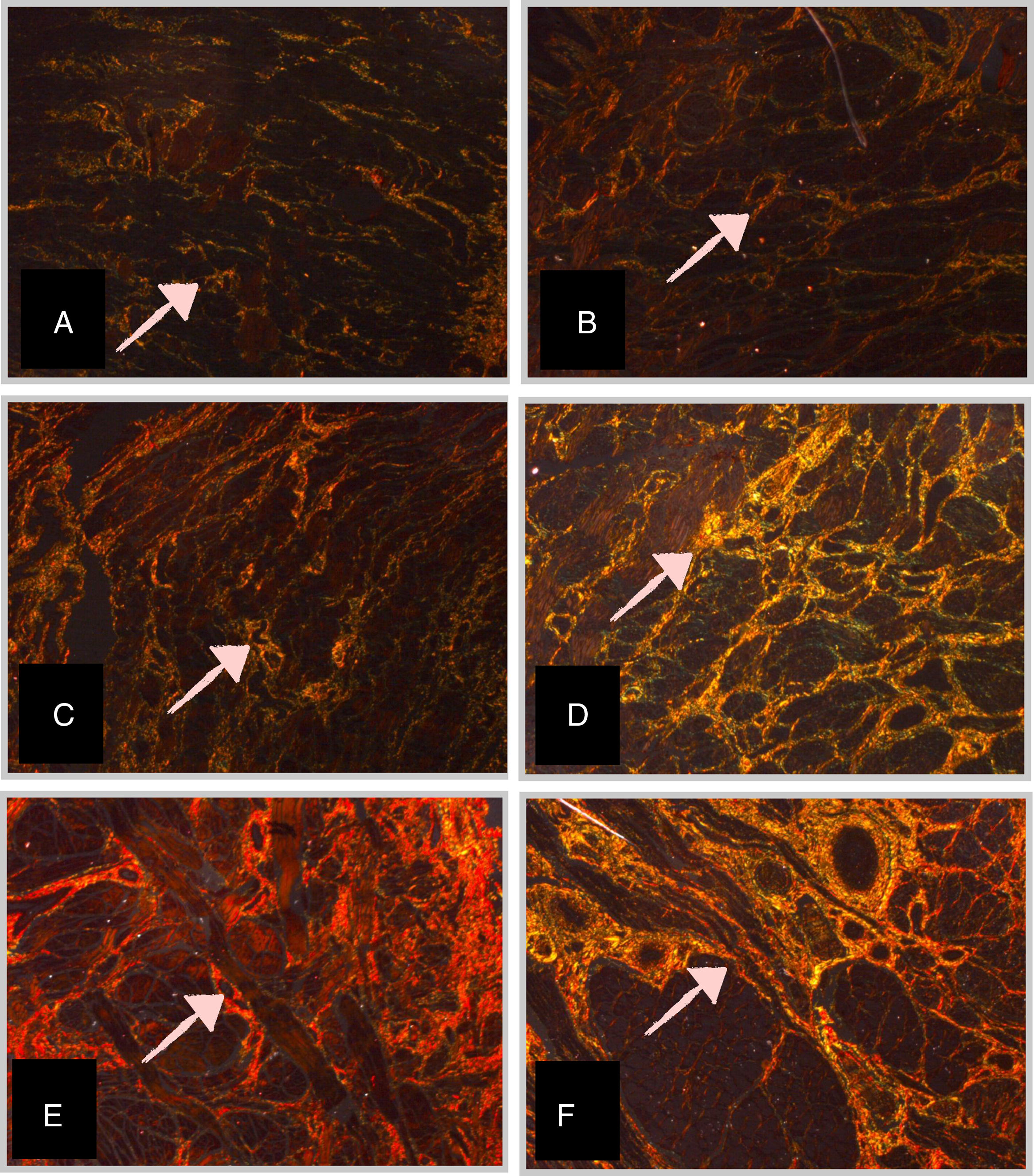
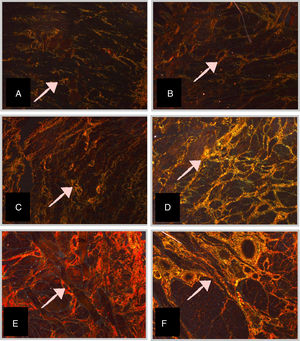
Morphometric analysis of collagen fibersThe PS-stained slide was analyzed for quantification of collagen fibers. The number of fields for evaluation and quantification of collagen fibers of the longitudinal section of the tongue, at different GAs, was defined as four quadrants and ten fields per quadrant of the histological section were analyzed. The area of collagen fibers under polarized light presented a birefringent appearance, ranging from orange to red (Fig. 1). Collagen fibers were marked by the observer to obtain the percentage of collagen per field analyzed. Thus, the field image was digitized using a camera coupled to a microscope with a Leica Qwin Plus® image analyzer (Leica Microsystems Inc, IL, USA). Morphometry was performed with Leica Qwin Plus® software image analyzer system (Leica Microsystems Inc, IL, USA), with a 10× objective lens (final magnification of 320×).
Micrographs of stillborn tongue fragments examined under polarized light, showing the birefringent collagen fibers (arrows) (Picrosirius – 10×–320× final magnification) at different gestational ages (GA).
(A) GA: 23 weeks; (B) GA: 28 weeks; (C) GA: 34 weeks; (D) GA: 37 weeks; (E) GA: 39 weeks, and (F) GA: 40 weeks.
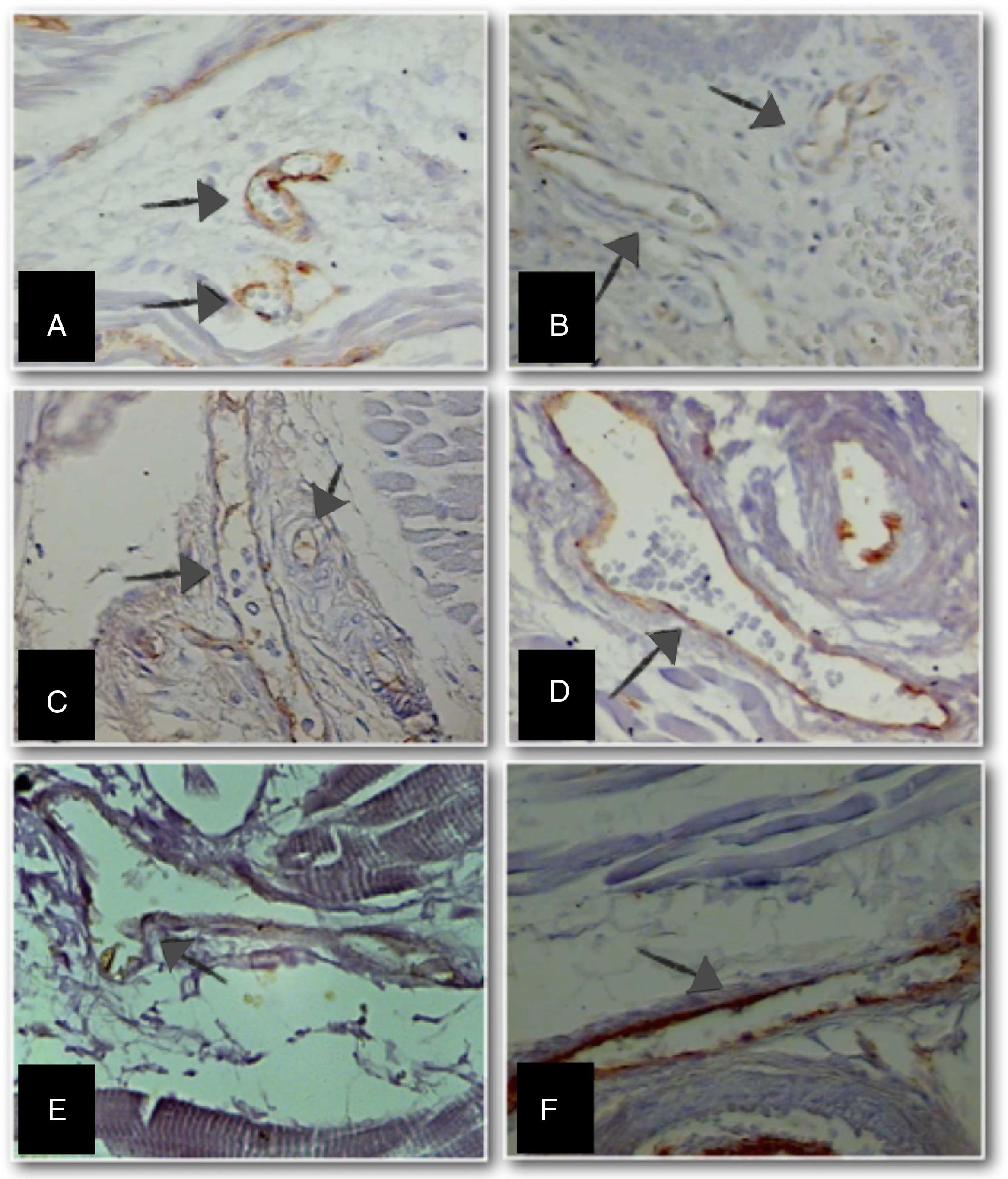
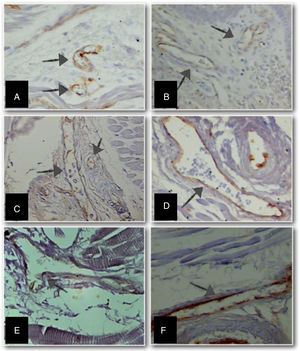
Immunohistochemistry was performed to identify anti-CD31 positivity. The number of fields for evaluation and quantification of the CD31 marker in the longitudinal section of the tongue, at different GAs, was defined as four quadrants and tend fields per quadrant of the histological section were analyzed. Measurements were taken using a video camera coupled to a light microscope, and these to a computer with the image analyzer system Axiovision SE64 Rel. 4.9.1® software. The perimeter of blood vessels was measured using ImageJ® Software (National Institutes of Health, USA), with an objective lens 100× (final magnification 3250×; Fig. 2).
Micrographs of stillborn tongue fragments examined under light microscopy, showing the increase in anti-CD31 immunolabeled blood vessels (arrows) (objective 100×–3250× final magnification) at different gestational ages (GA).
(A) GA: 23 weeks; (B) GA: 28 weeks; (C) GA: 34 weeks; (D) GA: 37 weeks; (E) GA: 39 weeks, and (F) GA: 40 weeks.
For the statistical analysis, a spreadsheet of the program Microsoft Excel® was elaborated. The information was analyzed using the electronic program GraphPad Prism® version 5.0 (GraphPad Software, Inc, CA, USA). To verify the type of distribution of the variables the statistical test of Shapiro–Wilk was applied. For correlation, the Spearman correlation coefficient (rS) was used for non-normal distribution. The differences were considered statistically significant when p was less than 5% (p<0.05).
ResultsOf the 152 reports of pediatric autopsies analyzed, 55 were selected for evaluation, with a median GA of 33 weeks, ranging from 23 to 40 weeks. Regarding gender, 34 (60.71%) were male and 22 (39.28%) were female. The analyzed data were presented in Table 1.
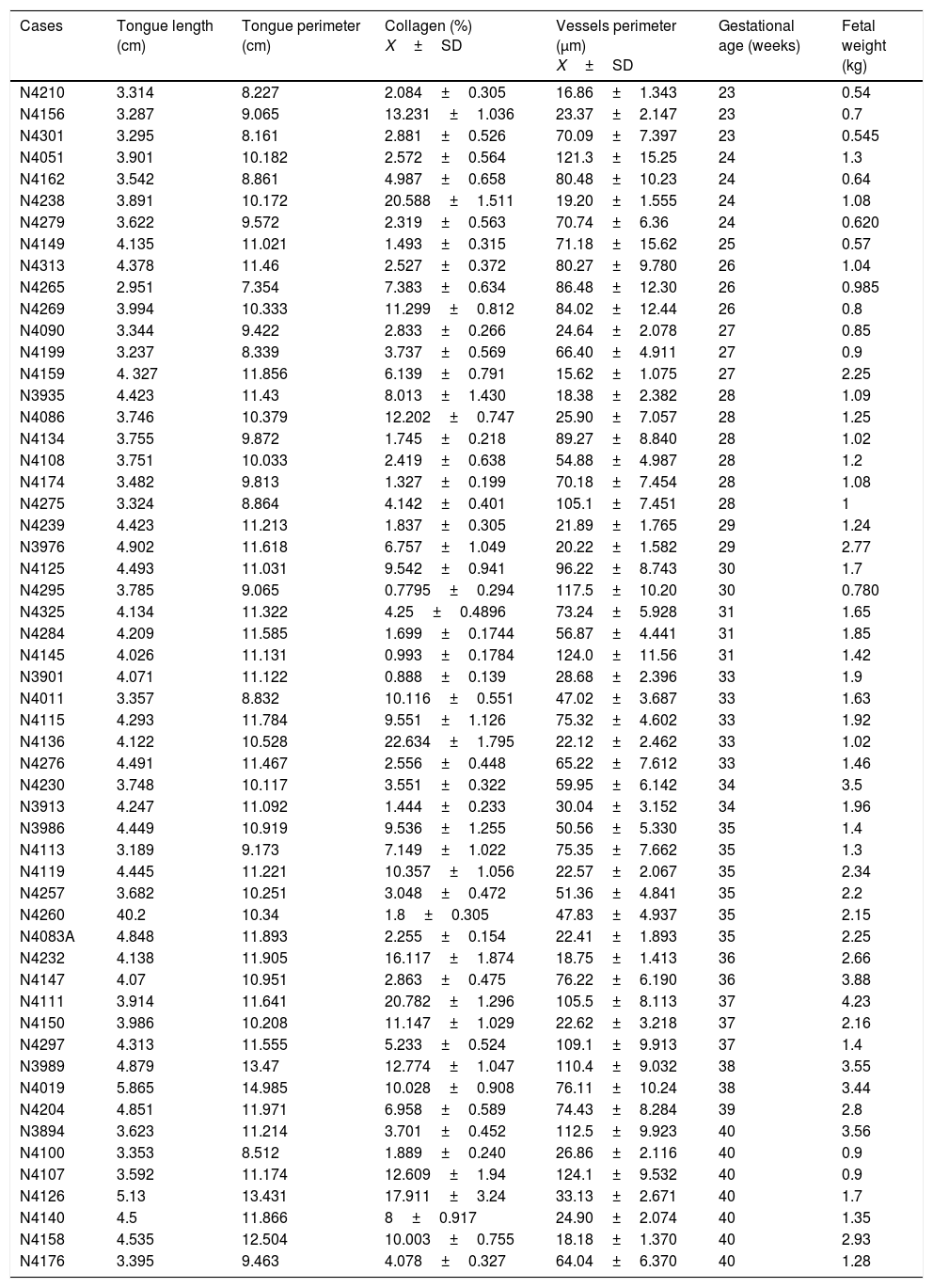
Constitutional and morphometric data of the 55 stillborns autopsied by the discipline of General Pathology at the Clinical Hospital of the Federal University of Triângulo Mineiro (HC/UFTM), Uberaba, State of Minas Gerais, Brazil, from 1994 to 2015.
| Cases | Tongue length (cm) | Tongue perimeter (cm) | Collagen (%) X±SD | Vessels perimeter (μm) X±SD | Gestational age (weeks) | Fetal weight (kg) |
|---|---|---|---|---|---|---|
| N4210 | 3.314 | 8.227 | 2.084±0.305 | 16.86±1.343 | 23 | 0.54 |
| N4156 | 3.287 | 9.065 | 13.231±1.036 | 23.37±2.147 | 23 | 0.7 |
| N4301 | 3.295 | 8.161 | 2.881±0.526 | 70.09±7.397 | 23 | 0.545 |
| N4051 | 3.901 | 10.182 | 2.572±0.564 | 121.3±15.25 | 24 | 1.3 |
| N4162 | 3.542 | 8.861 | 4.987±0.658 | 80.48±10.23 | 24 | 0.64 |
| N4238 | 3.891 | 10.172 | 20.588±1.511 | 19.20±1.555 | 24 | 1.08 |
| N4279 | 3.622 | 9.572 | 2.319±0.563 | 70.74±6.36 | 24 | 0.620 |
| N4149 | 4.135 | 11.021 | 1.493±0.315 | 71.18±15.62 | 25 | 0.57 |
| N4313 | 4.378 | 11.46 | 2.527±0.372 | 80.27±9.780 | 26 | 1.04 |
| N4265 | 2.951 | 7.354 | 7.383±0.634 | 86.48±12.30 | 26 | 0.985 |
| N4269 | 3.994 | 10.333 | 11.299±0.812 | 84.02±12.44 | 26 | 0.8 |
| N4090 | 3.344 | 9.422 | 2.833±0.266 | 24.64±2.078 | 27 | 0.85 |
| N4199 | 3.237 | 8.339 | 3.737±0.569 | 66.40±4.911 | 27 | 0.9 |
| N4159 | 4. 327 | 11.856 | 6.139±0.791 | 15.62±1.075 | 27 | 2.25 |
| N3935 | 4.423 | 11.43 | 8.013±1.430 | 18.38±2.382 | 28 | 1.09 |
| N4086 | 3.746 | 10.379 | 12.202±0.747 | 25.90±7.057 | 28 | 1.25 |
| N4134 | 3.755 | 9.872 | 1.745±0.218 | 89.27±8.840 | 28 | 1.02 |
| N4108 | 3.751 | 10.033 | 2.419±0.638 | 54.88±4.987 | 28 | 1.2 |
| N4174 | 3.482 | 9.813 | 1.327±0.199 | 70.18±7.454 | 28 | 1.08 |
| N4275 | 3.324 | 8.864 | 4.142±0.401 | 105.1±7.451 | 28 | 1 |
| N4239 | 4.423 | 11.213 | 1.837±0.305 | 21.89±1.765 | 29 | 1.24 |
| N3976 | 4.902 | 11.618 | 6.757±1.049 | 20.22±1.582 | 29 | 2.77 |
| N4125 | 4.493 | 11.031 | 9.542±0.941 | 96.22±8.743 | 30 | 1.7 |
| N4295 | 3.785 | 9.065 | 0.7795±0.294 | 117.5±10.20 | 30 | 0.780 |
| N4325 | 4.134 | 11.322 | 4.25±0.4896 | 73.24±5.928 | 31 | 1.65 |
| N4284 | 4.209 | 11.585 | 1.699±0.1744 | 56.87±4.441 | 31 | 1.85 |
| N4145 | 4.026 | 11.131 | 0.993±0.1784 | 124.0±11.56 | 31 | 1.42 |
| N3901 | 4.071 | 11.122 | 0.888±0.139 | 28.68±2.396 | 33 | 1.9 |
| N4011 | 3.357 | 8.832 | 10.116±0.551 | 47.02±3.687 | 33 | 1.63 |
| N4115 | 4.293 | 11.784 | 9.551±1.126 | 75.32±4.602 | 33 | 1.92 |
| N4136 | 4.122 | 10.528 | 22.634±1.795 | 22.12±2.462 | 33 | 1.02 |
| N4276 | 4.491 | 11.467 | 2.556±0.448 | 65.22±7.612 | 33 | 1.46 |
| N4230 | 3.748 | 10.117 | 3.551±0.322 | 59.95±6.142 | 34 | 3.5 |
| N3913 | 4.247 | 11.092 | 1.444±0.233 | 30.04±3.152 | 34 | 1.96 |
| N3986 | 4.449 | 10.919 | 9.536±1.255 | 50.56±5.330 | 35 | 1.4 |
| N4113 | 3.189 | 9.173 | 7.149±1.022 | 75.35±7.662 | 35 | 1.3 |
| N4119 | 4.445 | 11.221 | 10.357±1.056 | 22.57±2.067 | 35 | 2.34 |
| N4257 | 3.682 | 10.251 | 3.048±0.472 | 51.36±4.841 | 35 | 2.2 |
| N4260 | 40.2 | 10.34 | 1.8±0.305 | 47.83±4.937 | 35 | 2.15 |
| N4083A | 4.848 | 11.893 | 2.255±0.154 | 22.41±1.893 | 35 | 2.25 |
| N4232 | 4.138 | 11.905 | 16.117±1.874 | 18.75±1.413 | 36 | 2.66 |
| N4147 | 4.07 | 10.951 | 2.863±0.475 | 76.22±6.190 | 36 | 3.88 |
| N4111 | 3.914 | 11.641 | 20.782±1.296 | 105.5±8.113 | 37 | 4.23 |
| N4150 | 3.986 | 10.208 | 11.147±1.029 | 22.62±3.218 | 37 | 2.16 |
| N4297 | 4.313 | 11.555 | 5.233±0.524 | 109.1±9.913 | 37 | 1.4 |
| N3989 | 4.879 | 13.47 | 12.774±1.047 | 110.4±9.032 | 38 | 3.55 |
| N4019 | 5.865 | 14.985 | 10.028±0.908 | 76.11±10.24 | 38 | 3.44 |
| N4204 | 4.851 | 11.971 | 6.958±0.589 | 74.43±8.284 | 39 | 2.8 |
| N3894 | 3.623 | 11.214 | 3.701±0.452 | 112.5±9.923 | 40 | 3.56 |
| N4100 | 3.353 | 8.512 | 1.889±0.240 | 26.86±2.116 | 40 | 0.9 |
| N4107 | 3.592 | 11.174 | 12.609±1.94 | 124.1±9.532 | 40 | 0.9 |
| N4126 | 5.13 | 13.431 | 17.911±3.24 | 33.13±2.671 | 40 | 1.7 |
| N4140 | 4.5 | 11.866 | 8±0.917 | 24.90±2.074 | 40 | 1.35 |
| N4158 | 4.535 | 12.504 | 10.003±0.755 | 18.18±1.370 | 40 | 2.93 |
| N4176 | 3.395 | 9.463 | 4.078±0.327 | 64.04±6.370 | 40 | 1.28 |
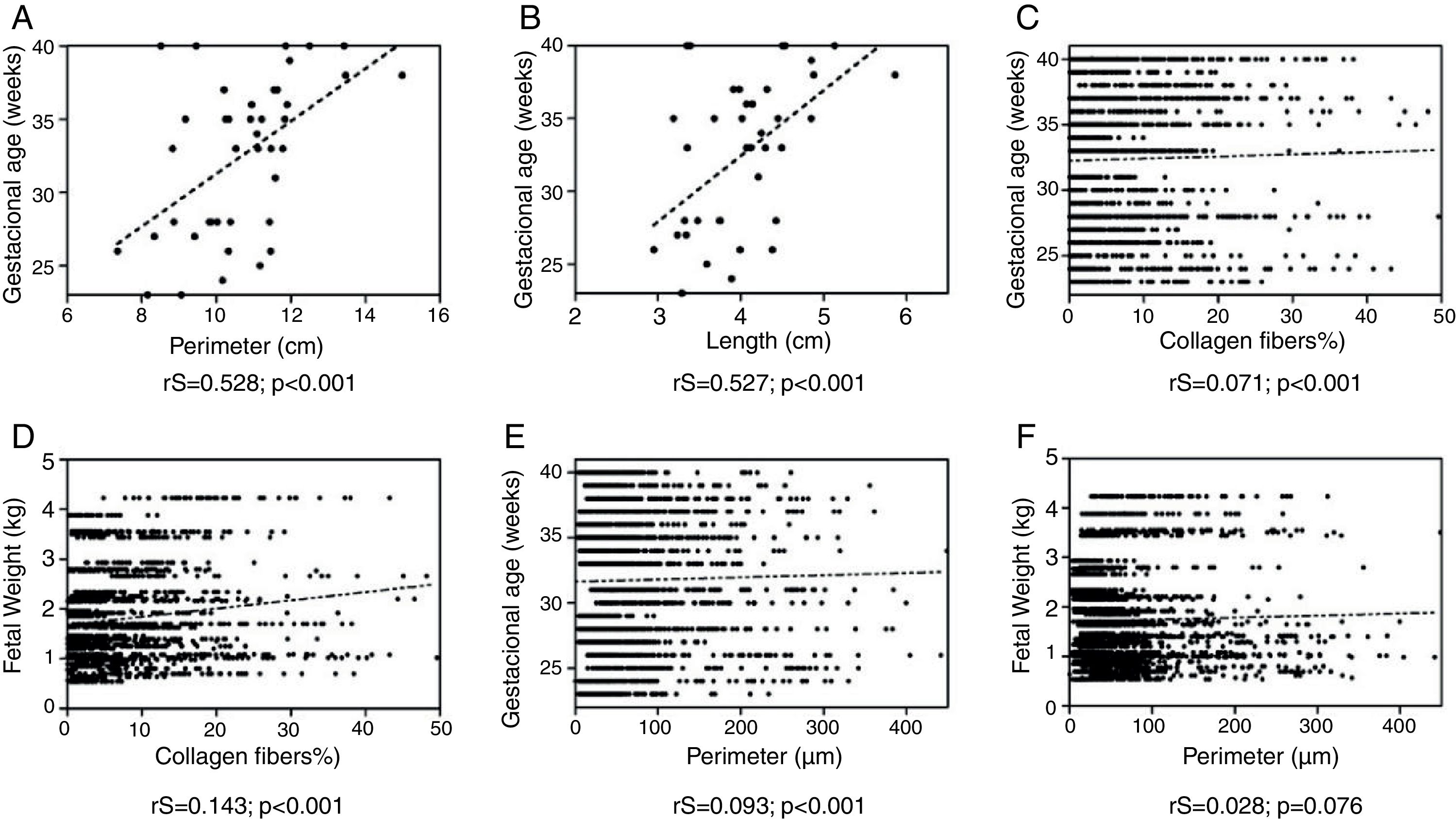
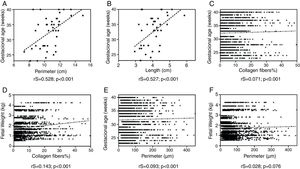
The tongue perimeter presented a positive and significant correlation with GA (rS=0.528; p<0.001; Fig. 3).
The correlation of GA and tongue length was also positive and significant (rS=0.527; p<0.001; Fig. 3).
A positive and significant increase was observed in the correlation of the GA with collagen fibers (rS=−0.071; p=0.001; Fig. 3).
Considering the relationship between collagen fibers and fetal weight, a positive and significant correlation was observed (rS=0.143; p<0.001; Fig. 3).
The correlation between the GA and the perimeter of the blood vessels was positive and significant (rS=0.093; p<0.001; Fig. 3).
A correlation was observed between the perimeter of blood vessels and the fetal weight: there was an increase in the perimeter of the vessels, in tendency significant (rS=0.028; p=0.076; Fig. 3).
DiscussionThe present study corroborates the literature, as a positive and significant increase in perimeter and length of the tongue was reported at different GAs.4,17 Fetal development is extremely important for the evaluation of the newborn, thus GA is an indispensable parameter for evaluation and survival after birth.18 Foot length is an important element for the structural assessment of the fetus at different GAs, because it is a body measurement that is closely related to GA, weight, and length.19
A positive and significant correlation was observed between collagen fibers in the tongue of stillborns and GA (23–40 weeks) and weight. This finding indicate that CNCC initiates and directly potentiates tongue development and gives rise to fibroblasts that promote the development of connective tissue.9,20
In addition, blood vessels perimeter was positively correlated with different GAs. The authors chose to use the perimeter for vessel morphometry using the CD31 marker, which has not yet been described in the literature. As observed in a study on stillborns with 20 to 40 weeks GA, anti-CD31 is a marker of vessels in relation to the development of GA.14,21
The present study corroborates the literature, in which 23 tongues of autopsied stillborns were analyzed, demonstrating that after the seventh week, the vessels (whose walls are beginning to develop) increase progressively. In the posterior region of the tongue, the blood vessels are small and form a very dense capillary network. The anterior vascularization of the tongue is greater, the vessels have smaller calibers, providing the conditions for a rapid supply of energy and nutrients to the myocytes. This capillary network of the tongue has been described in the literature as an important element against diseases.22–24
In turn, the increase of the perimeter of blood vessels correlated with the fetal weight was positive and tendency significant. There is a natural tendency for fetal growth during the different GAs, but some factors may cause changes in fetal weight, since this variable is different in each GA and depends on external factors, such as maternal nutrition. Intrauterine complications resulting in newborns with low birth weight (<2500g) are recognized as risk factors that contribute to the development of vascular diseases in adulthood.25 Fetal weight and GA should be taken into account due to the influence of other characteristics (genetic and socioeconomic factors). The increase in fetal weight may be related to a severe fetal complication, which generates a fetal systemic response characterized by edema, inflammation, and alteration of chemical mediators.26
The precise evaluation for growth in the neonatal period is important to observe if the fetus was subjected to abnormal intrauterine conditions that resulted in delayed growth acceleration.27 However, antenatal ultrasound detection and estimated fetal weight are far from straightforward, because these well-defined parameters are estimated using complex calculations that may give varying results for the same fetus. To further complicate matters, then obtained results can then be plotted on a number of different antenatal reference charts generated from local, national, or international cohorts, some of which are customized for maternal factors, such as parity, height, weight, and ethnicity. These variations contribute to large differences in antenatal detection abnormalities.28,29
Therefore, with the advance of GA, there is an increase in the perimeter and length of the tongue, an increase in the percentage of collagen fibers and an increase in the vascular perimeter, demonstrating that tongue formation is directly linked to fetal growth and development. Therefore, tongue embryogenesis would be a valid parameter to estimate GA in the pediatric autopsy, in conjunction with traditional methods.
FundingConselho Nacional de Desenvolvimento Cientifico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG), and Fundação de Ensino e Pesquisa de Uberaba (FUNEPU).
Conflicts of interestThe authors declare no conflicts of interest.
The present study was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG), and Fundação de Ensino e Pesquisa de Uberaba (FUNEPU).
Please cite this article as: Aguiar LS, Juliano GR, Silveira LA, Oliveira MS, Torquato BG, Juliano GR, et al. Tongue development in stillborns autopsied at different gestational ages. J Pediatr (Rio J). 2018;94:616–23.
Study conducted at Universidade Federal do Triângulo Mineiro (UFTM), Instituto de Ciências Biológicas e Naturais, Disciplina de Patologia Geral, Uberaba, MG, Brazil.