Autism spectrum disorders are lifelong and often devastating conditions that severely affect social functioning and self-sufficiency. The etiopathogenesis is presumably multifactorial, resulting from a very complex interaction between genetic and environmental factors. The dramatic increase in autism spectrum disorder prevalence observed during the last decades has led to placing more emphasis on the role of environmental factors in the etiopathogenesis. The objective of this narrative biomedical review was to summarize and discuss the results of the most recent and relevant studies about the environmental factors hypothetically involved in autism spectrum disorder etiopathogenesis.
SourcesA search was performed in PubMed (United States National Library of Medicine) about the environmental factors hypothetically involved in the non-syndromic autism spectrum disorder etiopathogenesis, including: air pollutants, pesticides and other endocrine-disrupting chemicals, electromagnetic pollution, vaccinations, and diet modifications.
Summary of the findingsWhile the association between air pollutants, pesticides and other endocrine-disrupting chemicals, and risk for autism spectrum disorder is receiving increasing confirmation, the hypothesis of a real causal relation between them needs further data. The possible pathogenic mechanisms by which environmental factors can lead to autism spectrum disorder in genetically predisposed individuals were summarized, giving particular emphasis to the increasingly important role of epigenetics.
ConclusionsFuture research should investigate whether there is a significant difference in the prevalence of autism spectrum disorder among nations with high and low levels of the various types of pollution. A very important goal of the research concerning the interactions between genetic and environmental factors in autism spectrum disorder etiopathogenesis is the identification of vulnerable populations, also in view of proper prevention.
Os transtornos do espectro autista (TEAs) são vitalícios e normalmente são doenças devastadoras que afetam gravemente o funcionamento social e a autossuficiência. A etiopatogenia é presumivelmente multifatorial, resultante de uma interação muito complexa entre fatores genéticos e ambientais. O aumento drástico na prevalência de TEAs observado nas últimas décadas levou à maior ênfase no papel dos fatores ambientais na etiopatogenia. O objetivo desta análise da narrativa biomédica foi resumir e discutir os resultados dos estudos mais recentes e relevantes sobre os fatores ambientais hipoteticamente envolvidos na etiopatogenia dos TEAs.
FontesFoi realizada uma pesquisa na Biblioteca Nacional de Medicina dos Estados Unidos (PubMed) sobre os fatores ambientais hipoteticamente envolvidos na etiopatogenia dos TEAs não sindrômicos, incluindo: poluentes atmosféricos, pesticidas e outros desreguladores endócrinos, poluição eletromagnética, vacinas e alterações na dieta.
Resumo dos achadosEmbora a associação entre poluentes atmosféricos, pesticidas e outros desreguladores endócrinos e o risco de TEA esteja recebendo cada vez mais confirmações, a hipótese de uma relação causal real entre eles ainda precisa de mais dados. Os possíveis mecanismos patogênicos por meio dos quais os fatores ambientais podem causar TEA em indivíduos geneticamente predispostos foram resumidos, com ênfase especial no papel cada vez mais importante da epigenética.
ConclusõesFuturas pesquisas devem investigar se há uma diferença significativa na prevalência de TEA entre nações com níveis altos e baixos de vários tipos de poluição. Um objetivo muito importante da pesquisa a respeito das interações entre fatores genéticos e ambientais na etiopatogenia do TEA é a identificação de populações vulneráveis, também em virtude da prevenção adequada.
Autism spectrum disorders (ASDs) are lifelong and often devastating conditions that severely affect social functioning and self-sufficiency, having a very negative impact on the lives of the entire family of the affected individuals. According to the criteria of the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-5), ASDs are defined by persisting deficits in social communication and interaction, as well as by restricted and repetitive behaviors, interests, and activities.1 ASDs presumably have a multifactorial etiopathogenesis, resulting from a very complex interaction between genetic and environmental factors.2,3 Only in a minority of cases is the presence of a defined medical condition demonstrable.
Epidemiological studies during the last decades have shown a dramatic increase in ASD prevalence, which has reached as much as 1–2% of children in recent years.4 The epidemiological study by Nevison suggests that this increase is mainly real,5 and therefore only in small part attributable to better knowledge of the problem. This phenomenon needs further investigation and possible explanatory hypotheses in terms of public health. Of course this prevalence increase cannot be explained based only on genetic factors, and the role of possible environmental factors should be carefully considered. First, it is necessary to understand what has changed in the environment and habits during the last few decades. In literature, several hypotheses have been considered. In this review, the authors carried out a synthesis of the most intriguing hypotheses as follows: all the recent (between January 1, 2013 and August 20, 2016) and relevant (preferably case–control studies involving humans) literature available on PubMed (United States National Library of Medicine) was selected, using the following keywords: “autism”, “air pollutants”, “pollution”, “pesticides”, “endocrine-disrupting chemicals”, “environmental factors”, “electromagnetic fields”, “vaccinations”, “omega-3”, and “epigenetics”.
Air pollutantsOver the last years, the etiopathogenic role of the exposure to air pollutants, mainly heavy metals and particulate matter (PM) during the pre-, peri-, and postnatal period, has been seriously considered in the literature, although definite conclusions are lacking. Here follows a brief description of some of the most important recent papers in this regard.
Becerra et al. investigated the possible effect of traffic-related air pollution exposure during pregnancy on the development of autism. The authors performed a population-based case–control study using data from air monitoring stations and a land-use regression (LUR) model to estimate exposure rates. They identified children born in Los Angeles, California, United States of America (USA), who were diagnosed with a primary autistic disorder (AD) according to the Diagnostic and Statistical Manual of Mental Disorders, 4th Edition, Text Revision (DSM-IV-TR)6 at the age of 3–5 years during the period 1998–2009. They included 7603 children with AD and 10 controls (by definition without documentation of autism) per case matched by sex, year of birth, and minimum gestational age. The authors performed conditional logistic regression, adjusting for: maternal age, birthplace, race/ethnicity, and education; birth type (single, multiple), parity; insurance type (a socioeconomic status proxy); and gestational age at birth. They calculated a 12–15% relative increase in odds of autism per interquartile range (IQR) increase for ozone and PM2.5 (PM with an aerodynamic diameter less than 2.5μm) when mutually adjusting for both pollutants. Moreover, they calculated a 3–9% relative increase in the odds of AD per IQR increase for LUR-derived estimates of exposure to nitric oxide and nitrogen dioxide. LUR-derived associations were most robust for children born to mothers who had less than high school education. The authors concluded by suggesting the presence of associations between prenatal exposure to mostly traffic-related air pollution and autism.7
Volk et al. examined the possible association between air pollution and autism. They performed a population-based case–control study in California (USA) including 279 preschool children with autism (according to both the Autism Diagnostic Observation Schedule [ADOS] and the Autism Diagnostic Interview-Revised [ADI-R]) and 245 normal controls, frequency matched by sex, age, and broad geographic area. Odds ratios for autism were adjusted for children's sex and ethnicity, parents’ education level, maternal age, and prenatal smoking. Children with autism, during gestation and the first year of life, were more likely to live at residences with the highest quartile of exposure to traffic-related air pollution compared with controls. In the same periods, the exposure to nitrogen dioxide, PM2.5, and PM10 (PM with an aerodynamic diameter less than 10μm) were also associated with autism. The authors concluded that during pregnancy and the first year of life, the exposure to traffic-related air pollution, nitrogen dioxide, PM2.5, and PM10 is associated with autism.8
In Taiwan, Jung et al. studied the possible association between children's long-term postnatal exposure to air pollution and newly diagnosed ASD. From 2000 through 2010, they performed a prospective population-based cohort study considering 49073 individuals aged less than 3 years. Within this cohort, 342 children developed ASD according to the criteria of the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM).9 Hazard ratios were adjusted for age, anxiety, gender, intellectual disability, preterm birth, and socioeconomic status. The risk of new diagnoses of ASD augmented according to increasing levels of ozone, carbon monoxide, nitrogen dioxide, and sulfur dioxide. The results of the study suggest that children's exposure to these four pollutants in the preceding 1–4 years may increase the risk of developing ASD. No association has been found between PM10 and the risk of new diagnoses of ASD.10
In the USA, Roberts et al. studied the possible association between perinatal exposure to air pollutants and ASD. They estimated associations between levels of hazardous air pollutants at the birth time/place and ASD in the children (325 cases vs. 22101 controls) of participants in the Nurses’ Health Study II, a prospective longitudinal cohort of female nurses recruited in 1989. The authors accounted for possible biases by adjusting for family-level and census tract-level socioeconomic status, maternal age at birth, and birth year. Perinatal exposures to the highest vs. lowest quintile of diesel particulate, lead, manganese, nickel, and cadmium – as well as an overall measure of metals – were significantly associated with ASD. For most pollutants, the association was stronger for males than for females. The authors concluded that perinatal exposure to air pollutants may increase ASD risk, suggesting further studies for the possible sex-specific biological pathways associating perinatal exposure to air pollutants with ASD.11
Von Ehrenstein et al. evaluated the risks for autism related to the exposure to monitored ambient air pollutants during pregnancy in Los Angeles County, California. Among the cohort of children born between 1995 and 2006, the authors considered 148722 individuals whose mothers were living in a 5-km buffer radius around air pollution monitoring stations during pregnancy. The authors included 768 children diagnosed with AD according to DSM-IV-TR criteria6 between 1998 and 2009. The risks for autism were heightened per interquartile range increase in average concentrations of several pollutants during pregnancy, including 1,3-butadiene, meta/para-xylene, other aromatic solvents, lead, perchloroethylene, and formaldehyde, adjusting for maternal age, race/ethnicity, nativity, education, insurance type, parity, child sex, and birth year. The authors concluded that autism risks in children may increase following in utero exposure to traffic- and industry-related ambient air toxics.12
Talbott et al. studied the possible association between prenatal and early childhood exposure to PM2.5 and risk for ASD. ASD diagnosis was made if a child scored ≥15 on the Social Communication Questionnaire (SCQ) and had written documentation, including the ADOS or other diagnostic tools results, of an ASD diagnosis from a child psychologist or psychiatrist. The authors performed a population-based case–control study, considering 217 ASD children, born in Southwestern Pennsylvania (USA) between 2005 and 2009, in comparison with 226 controls without ASD, frequency matched for age, sex, and race. Odds ratios (adjusted for maternal age, education, race, and smoking) were high, but not significant, for specific prenatal and postnatal intervals (pre-pregnancy, pregnancy, and year one), while postnatal year two was significant. The authors also evaluated the effect of cumulative pregnancy periods; starting three months before pregnancy through pregnancy, the adjusted odds ratios were significant for pre-pregnancy through year two. The authors concluded that both prenatal and postnatal PM2.5 exposures are associated with increased ASD risk, suggesting that future studies should consider multiple pollutant models as well as the elucidation of the PM2.5 involvement in the etiopathogenesis of ASD.13
Again in Southwestern Pennsylvania, Talbott et al. performed a population-based case–control study to estimate the possible association between exposure to 30 environmental neurotoxicants and ASD.13 The authors studied 217 children with ASD born between 2005 and 2009 in comparison with 224 controls, frequency matched by birth year, sex, and race, and 5007 controls from a random sample of birth certificates using birth residence. The authors calculated odds ratios, adjusted for mother's age, education, race, smoking status, child's year of birth, and sex. They found that living during pregnancy in areas with higher styrene and chromium levels was associated with increased ASD risk, while borderline effects were found for polycyclic aromatic hydrocarbons (PAHs) and methylene chloride. However, based on these findings, it is unclear whether the mentioned chemicals represent risk factors in themselves or reflect the effect of a pollutant mixture.14
Considering that in the USA children with ASD appear to live in spatial clusters and that the reason for this clustering is hard to determine due to boundless variations in healthcare access and diagnostic practices, Schelly et al. explored ASD diffusion in Costa Rica, a small setting where no variations in healthcare access or diagnostic practices are present. In addition, in Costa Rica the potential for exposure to mercury from the source hypothetically implicated in ASD (see coal-fired power plants) is absent, and areas with high levels of air pollution are spatially concentrated. The study included 118 children with ASD, diagnosed according to ADOS, assessed in the period of 2010–2013. The authors identified spatial clusters suggesting a mechanism that does not depend on factors such as information about ASD, healthcare access, diagnostic practices, or environmental toxicants. The results that emerged from the study do not support the most likely environmental clustering cause, which is air pollution.15
Dickerson et al. performed an ecologic study in five sites located in the USA: in Arizona, Maryland, New Jersey, South Carolina, and Utah, respectively. They examined the association during the 1990s between prevalence of ASD (diagnosed according to DSM-IV-TR criteria6), at the level of census tract, and nearness of tract centroids to the closest industrial facilities emitting arsenic, lead, or mercury. They analyzed 2489 census tracts with 4486 ASD cases, adjusting for demographic and socio-economic area-based characteristics. The authors found that the prevalence of ASD was increased in census tracts located in the closest 10th percentile compared to those located in the furthest 50th percentile. The authors concluded that these findings suggest an association between residential proximity to industrial facilities that emit air pollutants and increased ASD prevalence.16
Dickerson et al., again considering the same sample of 4486 children with ASD living in 2489 census tracts, using multi-level negative binomial models, studied the possible association between lead, mercury, and arsenic air concentrations and ASD prevalence. When adjusting for demographic and socio-economic factors, tracts with lead concentrations in the highest quartile had significantly increased ASD prevalence compared to tracts with concentrations of lead in the lowest quartile. Furthermore, tracts with mercury concentrations over the 75th percentile and arsenic concentrations under the 75th percentile had a significantly increased ASD prevalence compared to tracts with concentrations of arsenic, lead, and mercury under the 75th percentile. The authors concluded by suggesting a possible association between air lead concentrations and ASD prevalence, and sustaining that multiple metal exposure may produce synergistic effects on the development of ASD.17
In the USA, Kalkbrenner et al. examined the exposure to PM10 at the birth address in 979 children with ASD, diagnosed according to DSM-IV-TR,6 born from 1994 to 2000 (645 in North Carolina and 334 in California), compared with 14666 randomly sampled controls born in the same counties and years (12434 in North Carolina and 2232 in California, respectively). The authors calculated odds ratios of autism for a 10μg/m3 increase in PM10 concentration within periods of three months from the preconceptional period through the child's first birthday. Odds ratios were adjusted for year, state, mother's education and age, race/ethnicity, and neighborhood-level urbanization and median household income. In addition, a nonparametric term for week of birth was included to account for seasonal trends. The authors found that PM10 exposure during the third-trimester, but not in earlier pregnancy, was associated with a higher risk of autism. The authors pointed out that their data did not allow them to know the PM composition (PM10 arises from traffic, wood smoke, and power plants), but further research in this field is also important in order to facilitate prevention efforts against the different sources from which the PM can arise.18
In the USA, Raz et al. examined the possible association between mother's PM exposure and odds of ASD in her child, performing a nested case–control study of the Nurses’ Health Study II participants.11 They included 245 children with a diagnosis of ASD, confirmed by ADI-R and Social Responsiveness Scale (SRS), and 1522 randomly selected children without ASD. During pregnancy the exposure to PM2.5 was associated with higher odds of ASD, with an adjusted (for child sex, year of birth, month of birth, maternal age at birth, paternal age at birth, and census income) odds ratio for ASD of 1.57 per IQR increase in PM2.5, considering only the women who had maintained the same address before and after pregnancy (160 cases and 986 controls). The association between PM2.5 exposure and ASD was stronger in the third trimester than in the first two trimesters of pregnancy when mutually adjusted. Little association was found between PM10-2.5 (PM with an aerodynamic diameter between 10μm and 2.5μm) and ASD. The authors concluded that during pregnancy, especially in the third trimester, a higher maternal exposure to PM2.5 is associated with increased odds of a child with ASD.19
Guxens et al. performed a study on four European population-based birth/child cohorts. They assessed whether prenatal exposure to nitrogen oxides and PM, estimated between 2008 and 2011, was associated with autistic traits. The authors included 8079 children aged 4–10 years in the study. Autistic traits were assessed using respectively: the ASD module of the Autism–Tics, Attention Deficit and Hyperactivity Disorders, and Other Comorbidities (A-TAC) inventory in the Swedish cohort; the Pervasive Developmental Problems subscale of the Child Behavior Checklist for Toddlers (CBCL1½–5) in the Dutch and in the Italian cohort; an adapted 18-item version of the SRS in the Dutch cohort; and the Childhood Autism Spectrum Test (CAST) in the Spanish cohorts. Prenatal exposure to nitrogen oxides and PM was not associated with autistic traits. The authors hypothesized that prenatal air pollution exposure could be related to ASD but not to broad autistic traits. They suggested further research, including: European studies on children diagnosed with ASD; comparing manifestation and detection of ASD between the USA and Europe; and exploring the mechanisms underlying the association between air pollutants and ASD in order to better understand the different results compared to the previous studies.20
Pesticides and other endocrine-disrupting chemicalsEndocrine disrupting chemicals (EDCs) are generally manmade substances that can interfere with the endocrine system, and are present in a large number of home and industrial products. The possible association between EDCs, in particular pesticides, and autism has been discussed at length in recent years, so far without reaching definite conclusions.
In the context of the population-based case–control study called Childhood Autism Risks from Genetics and Environment (CHARGE), Shelton et al. explored, in California, the possible association between proximity of residence to agricultural pesticides during gestation and ASD or developmental delay (DD). For 970 participants, aged 2–5 years, commercial pesticide application data collected using the California Pesticide Use Report (from 1997 to 2008) were connected with the addresses during gestation. The pounds of active ingredient applied regarding organophophates, organochlorines, pyrethroids, and carbamates (four pesticide families selected by the authors) were aggregated within buffer distances of 1.25-km, 1.5-km, and 1.75-km from the residence. The authors used multinomial logistic regression to calculate the exposure odds ratio comparing cases affected by ASD (486, diagnosed according to both ADI-R and ADOS), or by DD (168, assessed by the Mullen Scales of Early Learning and the Vineland Adaptive Behavioral Scale), with normal controls (316) frequency matched to the ASD cases by sex, age, and the regional catchment area. Data were adjusted for education of the father, home ownership, birthplace of the mother, child race/ethnicity, maternal prenatal vitamin intake, and year of birth. During pregnancy, about one-third of the mothers lived within 1.5km of an application of agricultural pesticides. Living near organophosphates at some time during pregnancy was associated with 60% increased ASD risk, higher for third-trimester exposure to organophosphates overall, and for second-trimester applications of chlorpyrifos (an organophosphate explored independently). Children of mothers living near applications of pyrethroids during the preconception period or pregnancy at third trimester were at higher risk of both ASD and DD. DD risk was higher in those living near applications of carbamates, with no specific vulnerable periods. The authors concluded that their study supports the association between pesticide exposure during pregnancy, particularly organophosphates, and neurodevelopmental disorders, as well as providing novel findings about the associations of pyrethroids and carbamates with ASD and DD.21
In California, Keil et al. examined the possible association between mother-reported use of imidacloprid, which is a common household pesticide utilized for the treatment of fleas and ticks for pets, and ASD. Participants were enrolled as part of the aforementioned CHARGE study.21 The analytic dataset included complete information, collected before September 2011, for 407 children with ASD (assessed using ADI-R and ADOS) and 262 normal controls, frequency matched by sex, age at interview, and region of birth. The authors used Bayesian logistic models to evaluate the association between imidacloprid and ASD as well as to correct for potential differential exposure misclassification because of recall in a case–control study of ASD. At interview, control cases were slightly younger than ASD children (mean: 3 years 7 months and 3 years 10 months, respectively). Data were adjusted for: child's gender, birth regional center, and age; mother's education, race/ethnicity, and parity; and ownership of pets during the prenatal period. The odds of prenatal exposure to imidacloprid among cases with ASD were only slightly higher than among controls. A susceptibility window analysis showed higher odds ratios for exposures during gestation than for exposures during early life, and while considering only consistent users of imidacloprid, the odds ratio raised to 2.0. The authors concluded that the association between exposure to imidacloprid and ASD could result from exposure misclassification alone, due to recall bias. However, they suggested further investigation about this association and emphasized the need for validation studies concerning prenatal imidacloprid exposure in patients with ASD.22
Unfortunately, both these studies21,22 were based on data collected retrospectively, without biological samples acquired during pregnancy. In this regard, the work of Braun et al. represents a step forward. In this study on the gestational exposure to EDCs and autistic behaviors, the authors considered, besides six organochlorine pesticides, eight phthalate metabolites (phthalates are mainly utilized as plasticizers), bisphenol A (used to produce some plastics and epoxy resins), 25 polychlorinated biphenyls (PCBs) (for example, once widely utilized in electrical equipment as dielectric and coolant fluids), eight brominated flame retardants (widely used for example in electronic products), and four perfluoroalkyl substances (utilized in plastic laboratory materials and pipes). In the context of the study called Health Outcomes and Measures of the Environment (HOME), the authors measured the blood or urine concentrations of the mentioned substances in 175 pregnant women living in the Cincinnati metropolitan area (Ohio, USA), enrolled in the period of 2003–2006 (prospective birth cohort study). When the children were aged 4 and 5 years, mothers were administered the SRS. Data were adjusted for: maternal age at delivery, race, marital status, education, parity, insurance status, employment, household income, prenatal vitamin use, mother's depressive symptoms and IQ, caregiving environment, and gestational exposure to tobacco smoke. The authors explored confounder-adjusted associations between the mentioned EDCs and autistic behaviors assessed by SRS utilizing a two-stage hierarchical regression model to account for repeated measures and confounding by correlated EDCs. Most of the EDCs were associated with unimportant differences in SRS scores. Each two-standard deviations increase in polybrominated diphenyl ether-28 (PBDE-28) (a brominated flame retardant) or trans-nonachlor (an organochlorine pesticide) serum concentration was associated with more autistic behaviors. Conversely, the authors found fewer autistic behaviors in the children of women with detectable vs. non-detectable PCB-178 (a polychlorinated biphenyl), β-hexachlorocyclohexane (an organochlorine pesticide), or PBDE-85 (a brominated flame retardant) concentrations. Increasing perfluorooctanoate (PFOA) (a perfluoroalkyl substance) concentrations were also associated with fewer autistic behaviors. These latter associations with fewer autistic behaviors may suggest a possible protective action of the involved substances. The authors concluded that some EDCs were associated with greater or fewer childhood autistic behaviors, but the modest size of the sample did not allow them to reach definite conclusions.23
The intriguing hypothesis that EDCs may modify the axes of endogenous hormones, interfering with steroid-dependent neurodevelopment and increasing the ASD risk,23,24 still requires experimental confirmation.
Other environmental factorsGao et al. investigated the association between prenatal environmental risk factors and autism in Tianjin (China). They performed a case–control study including 193 children with autism and 733 typically developed controls matched by age and sex from 2007 to 2012 by using a questionnaire. The case group consisted of children diagnosed by a pediatrician according to the criteria of DSM-IV-TR6 and those who scored ≥30 on the Childhood Autism Rating Scale (CARS). Statistical analysis was performed using Quick Unbiased Efficient Statistical Tree (QUEST) and logistic regression analysis in order to calculate the odds ratio of each risk factor, adjusted for socioeconomic factors. By combining a classification tree and logistic regression analysis, the authors found that mother's depression during pregnancy and neonatal complications (anoxia, jaundice, and aspiration pneumonia) were associated with increased risk for child autism, while mother's air conditioner use and father's freshwater fish diet before pregnancy were associated with reduced risk for child autism. The authors mentioned that air conditioner use might decrease the concentration of air pollutants resulting from lower air PM by filters. Another hypothesis is that a better controlled indoor environment could reduce dampness and conditions that favor microbe growth. The fish diet of fathers might help to reduce or prevent paternal obesity; alternatively, fish oil might improve the quality of sperm.25
In the present authors’ opinion, the possible involvement of electromagnetic pollution in ASD etiopathogenesis is one of the most intriguing hypotheses, but at the same time one of the least studied.26 The great increase in electromagnetic pollution, related to the huge deployment of wireless technologies, seems to overlap chronologically with the increase in autism prevalence detected over the last decades. A number of studies in the literature have suggested possible biological and health effects, including carcinogenicity, attributable to electromagnetic exposure, probably at least in part mediated by damages to the DNA.27–29 In particular with regard to ASD, the hypothesized pathogenic mechanisms of electromagnetic pollution include: damages to the DNA, oxidative stress, intracellular calcium increase, dysfunction of the immune system, and disruption of the blood–brain barrier.28,29 The observational case–control study performed by Pino-López and Romero-Ayuso in Spain, involving 70 cases with ASD and 136 controls (aged 16–36 months), suggested the presence of a correlation between job-related electromagnetic exposure of the parents, in particular the father, and ASD in their children.30 However, the study shows some limitations in terms of methodology, including the fact that the authors used data from a single center (Ciudad Real) and employed a tool (Modified Checklist for Autism in Toddlers: M-CHAT) not entirely reliable for the diagnosis of ASD. A systematic epidemiological study involving multiple centers located in different geographical areas with diverse electromagnetic exposure levels is needed to evaluate the hypothesis of an association between the electromagnetic pollution extent and autism prevalence.26 But unfortunately, until now these kinds of studies on this topic have been lacking.
The possible role of childhood vaccinations in the ASD etiopathogenesis has been much discussed over the years and the debate is still ongoing. In their evidence-based meta-analysis, Taylor et al. considered five cohort and five case–control studies on this topic, involving 1256407 and 9920 children, respectively. The cohort data did not show any relations between vaccination and autism or ASD, nor between multiple vaccines (MMR), thimerosal, or mercury and autism or ASD. Similarly, the case–control data did not show an increased autism or ASD risk following exposure to MMR, mercury, or thimerosal when grouped by condition or by type of exposure. The authors concluded suggesting that vaccinations as well as the vaccine components (thimerosal or mercury) or MMR are not associated with an increased risk for autism or ASD.31 It is however to be noted that, according to the observation of Turville and Golden, the meta-analysis of Taylor et al. confirmed previous studies showing that ASD incidence is similar in groups of children who have been differently vaccinated, but it did not compare the ASD incidence in vaccinated and unvaccinated children.32 Therefore, there is still some uncertainty concerning this matter and, according to Sealey et al., this topic should be studied further and the scientific community should still be vigilant concerning the possible association between vaccines and ASD.33 However, at present foregoing vaccinations appears to be an unjustified and dangerous choice for public health.
Van Elst et al. developed an enticing theory according to which there may be a link between polyunsaturated fatty acids (PUFA) and ASD. The authors have pointed out that over the last decades the increase in ASD prevalence seems to have been concomitant with modifications in dietary fatty acid composition, namely the substitution of cholesterol with omega-6 in many foodstuffs, which has caused a remarkable increase in the omega-6/omega-3 ratio. The authors hypothesized that in particular during the life's earliest stages, an omega-3 deficit may lead to alterations of: neurogenesis, synaptogenesis, myelination; neurotransmitter synthesis and turnover; brain connectivity; expression of peroxisome proliferator-activated receptors; responses of inflammation; and cognitive functioning and behavior. All of the foregoing factors appear to be related to ASD.34 Unfortunately, to date randomized clinical trials as a whole have shown no significant effects of supplementation with omega-3 in children affected by ASD, even though one cannot exclude the existence of a subgroup of patients with ASD who may respond to this treatment.35
DiscussionToday it is widely accepted that both genetic and environmental factors are implicated in the etiopathogenesis of the so-called non-syndromic ASD, which is not secondary to a well-known genetic condition (such as tuberous sclerosis complex or fragile X syndrome). For a long time, the weight of the genetic component was considered clearly prevalent, but in recent years, given the dramatic increase in the prevalence of ASD, the role of environmental factors has gained relatively greater importance. In addition to these obvious epidemiological considerations, as suggested by Wong et al., the hypothesis that the exposure to environmental factors may considerably contribute to the ASD development is supported by the presence of different subsets of candidate genes in each subject with ASD and by the large phenotypical variability of these individuals.36 However, there is still much to discover about the complex interaction between these causal factors. The relatively new concept of epigenetics can be very helpful in this regard, at least in a subpopulation of cases with ASD. Epigenetics represents a fundamental gene regulation mechanism that is based on chemical modifications of DNA and histone proteins, without changing the DNA sequence. It has been proposed that environmental factors, such as heavy metals and EDCs, can modify epigenetic status and gene expression, causing ASD.37,38
However, the etiopathogenic role of environmental factors has still to be considered carefully due to the lack of conclusive data. While the association between, for example, air pollutants and the risk for ASD is receiving increasing confirmation, the hypothesis of a real causal relation between them needs further data, as well as the fact that possible pathogenic mechanisms of the air pollutants involved in ASD remain hypothetical. In humans, air pollutant exposure has been shown to favor oxidative stress and inflammation,16 which may contribute to ASD pathogenesis.39 Furthermore, lead, mercury, and arsenic are known neurotoxicants that can cross the blood-brain barrier and impair neurodevelopment.16 Epigenetic mechanisms may be hypothesized based on the results of the experimental study on animals carried out by Hill et al.37 Also to be taken into consideration is the possibility that mixtures of air pollutants, and not single pollutants, are related to ASD, due to synergistic effects12,17; of course, this would make the interpretation of data emerging from studies about pollutants and ASD much more difficult. According to Guxens et al., the father's air pollution exposure during the preconception period also may have a role in ASD etiopathogenesis,20 but studies about this topic are scanty. Unfortunately, the current knowledge in this field is still at the level of assumptions or only a little further developed.
On the other hand, the importance of the genetic component in the etiopathogenesis of ASD should not be forgotten, demonstrated by an impressive amount of data in the literature over the years, and also confirmed today by a simple observation: while the prevalence of the disorder has been increasing over the last decades, the large disproportion between males and females in favor of the former has been confirmed.4 If the etiopathogenesis of ASD was related only to environmental factors, with which both males and females are inevitably in contact, there should be no reason for this persisting difference in prevalence between the sexes.
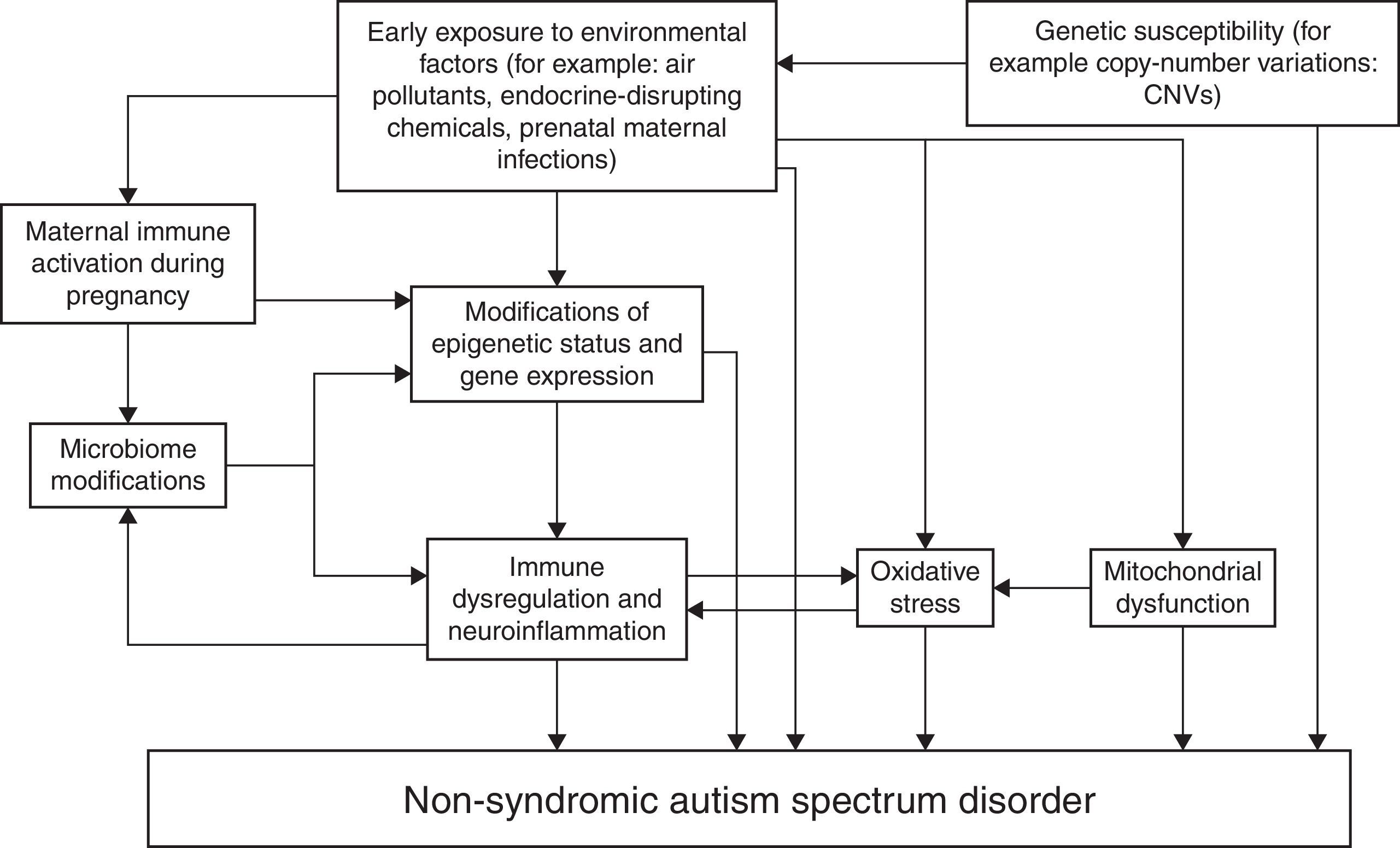
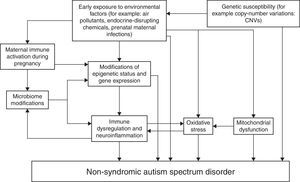
Fig. 1 represents an attempt to summarize various possible interactions among genetic and environmental factors involved in the etiopathogenesis of non-syndromic ASD. The schema shows that in genetically predisposed individuals, environmental factors may lead to autism directly or through epigenetic mechanisms, alterations of the immune function of both the mother during pregnancy (maternal immune activation) and the affected individual, modifications of the microbiome (represented by the totality of symbiotic micro-organisms harbored by the human body) that in turn reciprocally interacts with the immune function, oxidative stress, and mitochondrial dysfunction.37–45 This schema does not claim to be exhaustive and certainly will need updates.
However, despite the lack of conclusive data, the possible impact of environmental pollutants on public health should be kept in mind and therefore the precautionary principle, according to Suades-González et al., should be applied in order to protect children from possible pathogenic factors.46 First of all, there are obvious ethical and moral reasons to prompt a precautionary approach to the pollutants in the event that this can reduce the occurrence of new cases of ASD. However, even apart from these essential considerations, there are also reasons related to the possibility of obtaining, with adequate prevention, a savings of human and financial resources due to the high costs to society caused by the management of individuals with ASD over a lifetime.
ConclusionsDespite the great efforts performed during the last decades in medical research, involving considerable human and financial resources, today many aspects of ASD etiopathogenesis are still unknown, while the prevalence of this heterogeneous condition has increased greatly without satisfactory explanations. This led to develop several hypotheses, which are often divergent and still need confirmation. Thanks to the huge amount of data that has emerged from the recent research on autism, much more information is now known about the brain functioning of these subjects, but also about the typically-developed individuals. However, unfortunately, even today there is no specific treatment that can cure. In 2016, it is still observed that this disabling disorder is more and more frequent, without a known etiopathogenesis nor decisive treatments. This situation has indirectly favored, in the families involved, the use of treatments from complementary and alternative medicine,47 a phenomenon that is not always without risks for the patients.
Future research should investigate whether there is a significant difference in the prevalence of ASD among nations with high and low levels of the various types of pollution. So far, the vast majority of studies that have looked for a correlation between pollutants and autism have been conducted in the USA. This may be a limiting factor from an epidemiological point of view, especially because most of the few studies conducted elsewhere have not confirmed the results obtained in the USA. It is unclear whether this is due to methodological problems or to real differences between the geographical areas considered. According to Shelton et al.,21 a very important goal of the research concerning the interactions between genetics and environment is the identification of vulnerable populations, also in view of proper prevention. Finally, another aspect that the authors consider very important for research in this field is the uniformity of diagnostic criteria and assessment tools for ASD, in order to make the results of the studies performed across the world comparable.
Conflicts of interestThe authors declare no conflicts of interest.
The authors would like to thank Cecilia Baroncini for English revision.
Please cite this article as: Posar A, Visconti P. Autism in 2016: the need for answers. J Pediatr (Rio J). 2017;93:111–9.